
Journal of Organometallic Chemistry p. 14 - 22 (2002)
Update date:2022-08-10
Topics:
 Knopf
Knopf
 Herzog
Herzog
 Roewer
Roewer
 Brendler
Brendler
 Rheinwald
Rheinwald
 Lang
Lang
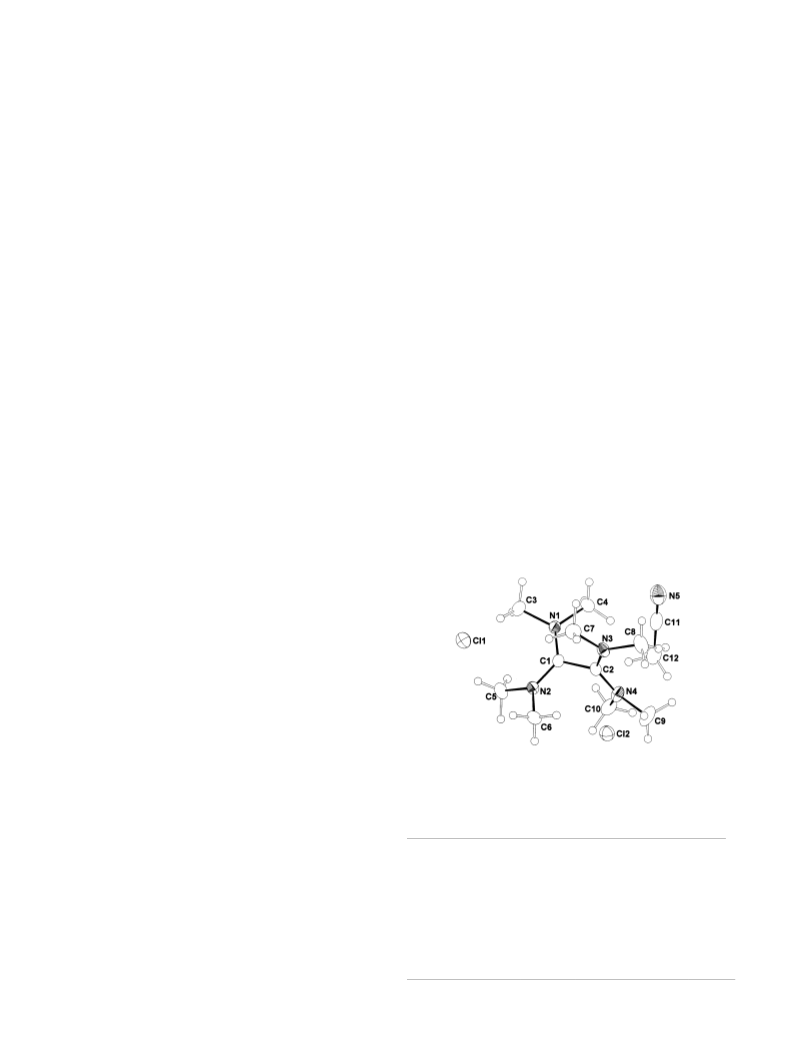
The chlorodisilanes SiClMe2-SiClMe2 (1), SiCl2Me-SiCl2Me (2), SiCl3-SiCl3 (3) and a 9:1 mixture of 2 and SiCl3-SiCl2Me (4) were reacted with the electron-rich alkene tetrakis-(dimethylamino)-ethylene (TDAE) in n-hexane as well as in polar solvents. While 1 gave no reaction at all, 3 underwent a disproportionation reaction into SiCl4 and Si(SiCl3)4. Also 2 and mixtures of 2 and 4 were disproportionated into MeSiCl3 (2a) and methylchlorooligosilanes. Additionally a crystalline mixture of Si3Me3Cl6 -TDAE (5a) plus Si3Me2Cl7 -TDAE (5b) was obtained by reaction of a 9:1 mixture of 2 and 4 with TDAE in n-hexane as well as in 1,2-dimethoxyethane. The reaction of 2 with TDAE in acetonitrile (MeCN) led to a crystalline precipitation of [TDAE]Cl2 -MeCN (6.MeCN) in addition to MeSiCl3 and methylchlorooligosilanes. The structures of 5b and 6.MeCN were determined by X-ray crystallography beside their NMR and IR spectroscopic characterization. Compound 5b crystallizes in the monoclinic space group P2/c (Z = 4), 6.MeCN in the orthorhombic space group Pna21 (Z = 4). The structure of 5b reveals a [TDAE].+ radical cation and a 1, 2-Me2Si3Cl7- anion with a pentacoordinated central silicon atom.
View More





Contact:+86-25-52346955
Address:199,JIANYE ROAD,NANJING,CHINA
website:http://www.debyesci.com
Contact:+85221376140
Address:Rm. 19C, Lockhart Ctr., 301-307 Lockhart Rd., Wan Chai
Pengchen New Material Technology Co., Ltd.
Contact:+86-512-63680537
Address:99.6 km of national road 318, Meiyan Community,Pingwang Town, Wujiang District, Suzhou 215225
Suzhou Health Chemicals Co., Ltd.
website:http://www.healthchems.com
Contact:13776257979
Address:No. 338, Jingang Avenue,
LIANYUNGANG YC FINE CHEMICAL CO., LTD
Contact:+86-518-858 99188
Address:Shangdong Modern Bldg, South Greenpark Road, Lianyungang, Jiangsu Pro. China
Doi:10.1039/c9ra10317a
(2020)Doi:10.1016/j.phytochem.2008.07.018
(2008)Doi:10.1021/jacs.9b00396
(2019)Doi:10.1016/S0040-4020(97)00989-7
(1997)Doi:10.1039/c4dt01942c
(2014)Doi:10.1248/cpb.33.2516
(1985)