H. Sharghi, M. H. Sarvari / Tetrahedron 58 (2002) 10323–10328
10327
and a quick process, and the yields of the products were
high.
7.10–7.27 (5H, m, Ph), 4.33 (1H, m, CHMe), 1.70 (3H, d,
Me); dC (250 MHz, CDCl3) 20.47, 28.79, 120.75, 126.22,
126.78, 129.33, 132.8; MS: Mþ (131).
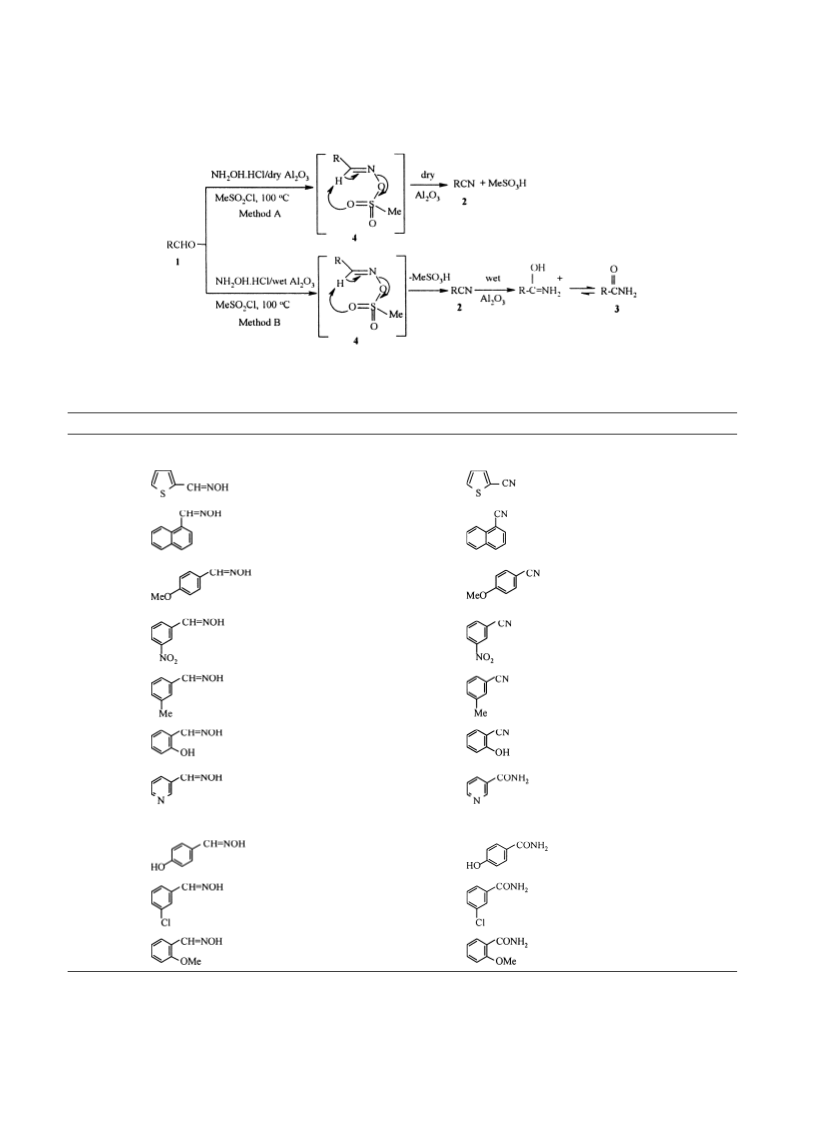
The effect of dry and wet alumina was evaluated in these
reactions. As shown in Fig. 2, the first step, in both
reactions, involves the formation of an aldoxime followed
by its reaction with MeSO2Cl to generate compound 4, as
the key intermediate. In the presence of dry-alumina
(method A) compound 4 subsequently undergoes elimin-
ation thermally to produce nitrile 2 with the liberation of
MeSO3H. On the other hand, when wet-alumina was used
(method B), the nitrile 2 underwent rapid hydration to
produce amide 3.
2.2. Typical procedure for synthesis of amides in the
presence of wet-alumina (Table 2)
Aldehyde (1 mmol), NH2OH·HCl (0.3 g, 0.4 mmol),
MeSO2Cl (0.075 mL, 1 mmol) and wet Alumina (0.5 g,
4.9 mmol, Fluka type 405 acidic, to make it wet only one
drop of water was added) were thoroughly mixed with a
mechanical stirrer. The resulting fine powder was trans-
ferred to a round-bottom flask (5 mL) and heated in an oil
bath at 1008C for appropriate time (Table 2). Then ethyl
acetate was added to the reaction mixture and alumina was
removed by filtration. The filtrate was extracted with water
(2£10 mL), dried over Na2SO4 and the solvent evaporated
in vaccuo to give the crude product, which was purified by
recrystallization from EtOH.
According to Fig. 2, we found that our new methods also be
useful for conversion of aldoximes in the presence of dry-
alumina to produce nitriles and in the presence of wet-
alumina to produce amides in high yields. Some of these
results are summarized in Table 3.
In summary, this paper shows a simple and efficient general
access to pure nitriles and amides. These two important
materials could be synthesized in overall yields of 85–97%
from their corresponding aldehydes. The catalysts, dry and
wet alumina, which have been used here for the first time for
such conversions are inexpensive and commercially avail-
able. The reaction was carried out in dry media and no
solvents were used. The work-ups were easy and the
experimental procedures are environmentally benign.
2.3. Typical procedure for synthesis of nitriles or amides
from oximes in the presence of dry or wet alumina
(Table 3)
A mixture of aldoxime (1 mmol), CH3SO2Cl (1 mmol,
0.075 mL) and dry or wet alumina (0.5 g, 4.9 mmol) was
heated in an oil bath at 1008C. The progress of the reaction
was monitored by TLC. After the reaction was complete,
ethyl acetate was added to the reaction mixture and alumina
was removed by filtration. It was then washed with water
(2£10 mL) and dried over anhydrous Na2SO4. After
removal of the solvent, the crude product was obtained
and purified by recrystallization from EtOH.
2. Experimental
2.1. Typical procedure for synthesis of nitriles in the
presence of dry-alumina (Table 1)
Acknowledgements
Aldehyde (1 mmol), NH2OH·HCl (0.3 g, 0.4 mmol),
MeSO2Cl (0.075 mL, 1 mmol) and dry Alumina (0.5 g,
4.9 mmol, Fluka type 405 acidic, dried in an oven for 5 h)
were thoroughly mixed with a mechanical stirrer. The
resulting fine powder was transferred to a round-bottom
flask (5 mL) and heated in an oil bath at 1008C for
appropriate time (Table 1). Then diethyl ether (10 mL) was
added to the reaction mixture and alumina was removed by
filtration. The filtrate was extracted with water (2£10 mL),
dried over Na2SO4 and the solvent evaporated in vaccuo to
give the crude product, which solids were purified by
recrystallization from EtOH and liquids by distillation.
We gratefully acknowledge the support of this work by the
Shiraz University Research Council.
References
1. Pagni, R. M.; Kobalka, G. W.; Boothe, R.; Gaetano, K.;
Stewart, L. J.; Conawaya, R. J. Org. Chem. 1998, 53, 4477.
2. For review on surface organic chemistry, see: (a) Posner, G. H.
Angew Chem., Imt. Ed. Engl. 1978, 17, 487. (b) McKillop, A.;
Young, D. W. Synthesis 1979, 401–408. (c) Cornelis, A.;
Laszlo, P. Synthesis 1985, 909. (d) Laszlo, P. Acc. Chem. Res.
1986, 19, 121. (e) Cornelis, A.; Laszlo, P. In Chemical
Reactions in Organic and Inorganic Constrained Systems.
Setton, R., Ed.; Reider: Dordrecht, 1986; p 212.
2.1.1. 1-Phenylethyl cyanide (entry h, Table 1). Hydra-
tropaldehyde (0.13 g, 1 mmol), NH2OH·HCl (0.3 g,
0.4 mmol), MeSO2Cl (0.075 mL, 1 mmol) and dry Alumina
(0.5 g, 4.9 mmol, Fluka type 405 acidic) were thoroughly
mixed with a mechanical stirrer. The resulting fine powder
was transferred to a round-bottom flask (5 mL) and heated
in an oil bath at 1008C for 40 min. Then diethyl ether
(10 mL) was added to the reaction mixture and alumina was
removed by filtration. The filtrate was extracted with water
(2£10 mL), dried over Na2SO4 and the solvent evaporated
in vaccuo. Purification of the crude product by recrystalliza-
tion from EtOH gave the title compound (95%) as a white
solid. Mp 2108C; nmax (KBr) 3146, 2243, 1683, 1496, 1454,
1078, 1029, 835, 765, 703 cm21; dH (250 MHz, CDCl3)
3. Friedrich, K.; Wallenfels, K. In The Chemistry of the Cyano
Group. Rappoport, Z., Ed.; Interscience: New York, 1970;
pp 92–93.
4. Olah, G. A.; Vankar, Y. D. Synthesis 1978, 702.
5. Olah, G. A.; Narang, S. C.; Garcia, L. A. Synthesis 1980, 659.
6. Meshram, H. M. Synthesis 1992, 943.
7. Ali, S. L.; Nikalje, M. D.; Dewkar, G. K.; Paraskar, A. S.;
Nikalje, M. D.; Sudalai, A. J. Chem. Res. (S) 2000, 30.
8. (a) Miller, C. P.; Kaufman, D. H. Synlett 2000, 8, 1169.
(b) Srinivas, K. V. N. S.; Reddy, E. B.; Das, B. Synlett 2002, 4,

 Sharghi, Hashem
Sharghi, Hashem
 Sarvari, Mona Hosseini
Sarvari, Mona Hosseini