Journal of the Iranian Chemical Society
1
3
2
H) ppm; C NMR (100 MHZ, DMSO-d ): δ 125.67, 127.15,
be powerful dehydrating agent and promoter, and promising
reagent for some organic reactions [44–53]. At the begin-
ning of these investigations, pyridine (1a) was selected as the
model substrate to establish the oxygenation reaction with
H O in the presence of Tf O as a promoter in EtOH at room
6
1
39.30 ppm.
4
-Methylpyridine-1-oxide (Table 2, entry 2) (2b): orange
1
solid; H NMR (400 MHZ, DMSO-d ): δ 2.32 (s, 3H, CH ),
6
3
13
7
6
.99 (d, 2H), 8.73 (d, 2H) ppm; C NMR (100 MHZ, DMSO-
2
2
2
d ): δ 18.12, 125.15, 134.55, 147.30 ppm.
temperature. It was found that the reaction failed to proceed.
Thus, Hendrickson reagent was used as a promoter under the
same reaction conditions, and it was found that pyridine-N-
oxide (2a) was obtained in 80% yield, and confirmed by its
3
-Methylpyridine-1-oxide (Table 2, entry 4) (2d): colorless
1
oil; H NMR (400 MHZ, DMSO-d ): δ 2.22 (s, 3H, CH ), 7.27
6
3
13
(
dd, 1H), 8.04 (d, 1H), 8.07–8.13 (m, 2H) ppm; C NMR (100
1
MHZ, DMSO-d ): δ 17.99, 126.32, 126.77, 136.51, 137.34,
H NMR spectrum (Table 1, entry 1). The effect of other
6
1
39.01 ppm.
solvents such as methylene chloride, chloroform, n-hexane,
acetonitrile, ethyl acetate, acetone and water was examined
under the same reaction conditions and the desired prod-
uct was obtained in lower yields (0–35%) (Table 1, entries
4–10).
2
,4,6-Trimethylpyridine-1-oxide (Table 2, entry 5) (2e):
1
brown solid; H NMR (400 MHZ, DMSO-d ): δ 2.27 (s,
6
13
9
H, 3CH ), 7.22 (s, 1H), 8.09 (s, 1H) ; C NMR (100 MHZ,
3
DMSO-d ): δ 18.66, 19.94, 127.50, 136.36, 138.54 ppm.
6
4
-N,N-Dimethylaminopyridine-1-oxide (Table 2, entry 6)
Chloroform, dichloromethane and n-hexane as solvents
appeared to afford the worst results due to their low ability
to solve the promoter reagent affecting the reaction. The
optimum conditions for the amounts of Hendrickson reagent
and H O were 1 equi, and 4 equiv, respectively. Using less
1
(
2f): yellow solid; H NMR (400 MHZ, DMSO-d ): δ 2.95
6
13
(
s, 6, 2CH ), 6.58 (d, 2H), 8.10 (d, 2H) ppm; C NMR (100
3
MHZ, DMSO-d ): δ 40.57, 107.17, 149.73, 154.42 ppm.
6
2
,4-Dimethylpyridine-1-oxide (Table 2, entry 7) (2 g):
2
2
1
brown solid; H NMR (400 MHZ, DMSO-d ): δ 2.26 (s,
than 1 equiv of the reagent or 4 equiv of H O in the reac-
6
2 2
13
6
H, 2CH ), 6.98 (d, 1H), 7.14 (s, 1H) 7.63 (d, 1H) ppm;
C
tion led to an incomplete reaction. Application of 2 equiv
of Hendrickson reagent gave no significant increment based
on the yield of the product. It was found that temperature
increasing of the reaction shows no significant improvement
in respect to the yield. Therefore, it seems that the optimum
conditions for the reaction are pyridine (1 equiv), H O (4
3
NMR (100 MHZ, DMSO-d ): δ 20.90, 21.58, 127.01, 128.75,
6
1
34.10, 143.92, 145.37 ppm.
Quinoline-1-oxide (Table 2, entry 8) (2 h): brown solid;
1
H NMR (400 MHZ, DMSO-d ): δ 7.45 (d, 1H), 7.48–7.73
6
(
m, 2H), 7.83 (d, 1H), 8.10 (d, 1H), 8.32–8.58 (m, 2H) ppm;
2
2
1
3
C NMR (100 MHZ, DMSO-d6): δ 119.32, 122.39, 125.60,
29.11, 129.21, 130.78, 135.74, 141.29 ppm.
equiv) and Hendrickson reagent (1 equiv) in ethanol (3 mL)
at room temperature. In the absence of promoter, after 48 h,
1
Quinolin-8-ol-1-oxide (Table 2, entry 9) (2i): brown solid;
1
H NMR (400 MHZ, DMSO-d ): δ 3.33 (s, 1H, OH), 6.98
6
(
d, 1H), 7.08–8.32 (m, 3H), 8.52 (d, 1H), 8.84 (d, 1H) ppm;
1
3
Table 1 Effect of solvent, amount of promoter and H O on synthesis
2
2
C NMR (100 MHZ, DMSO-d ): δ 111.77, 114.45, 117.52,
6
of pyridine-N-oxide
Entry Solvent
1
22.20, 132.53, 135.84, 136.58, 148.56, 153.79 ppm.
Pro-
H O
2
Time (min) Yield (%)a
2
-Methylquinoline-1-oxide (Table 2, entry 10) (2j): brown
2
1
moter
(mmol)
solid; H NMR (400 MHZ, DMSO-d ): δ 2.56 (s, 3H, CH ),
6
3
(
mmol)
7
.65 (d, 1H), 7.75 (t, 1H), 7.78–7.81 (m, 2H), 7.82 (d, 1H), 7.99
1
3
(
t, 1H); C NMR (100 MHZ, DMSO-d ): δ 18.66, 119.12,
1
2
3
4
5
6
7
8
9
1
1
EtOH
EtOH
EtOH
EtOH
EtOH
H2O
CH2Cl2
MeCN
CHCl3
n-Hexane
1
0.5
2
1
1
1
1
1
1
4
4
4
2
6
4
4
4
4
4
4
70
100
70
110
65
120
120
120
120
120
120
80
50
80
67
80
35
0
20
0
0
6
1
1
7
23.91, 124.43, 128.13, 128.93, 129.45, 130.51, 141.25,
45.34 ppm. Elemental analysis: calcd (%) for C H NO: C,
10
9
5.47; H, 5.66; N, 8.80; found: C, 75.55; H, 5.72; N, 8.63.
[
2,2′-Bipyridine]-1,1′-dioxide (Table 2, entry 11) (2 k): vio-
1
let solid; H NMR (400 MHZ, DMSO-d ): δ 7.45–7.90 (m,
6
3
H), 7.96 (t, 1H), 8.01 (d, 1H), 8.10 (d, 1H), 8.74 (t, 2H) ppm;
13
C NMR (100 MHZ, DMSO-d ): δ 124.87, 125.36, 125.81,
6
1
26.65, 127.96, 136.72, 140.95, 146.60, 149.89, 150.02 ppm.
0
1
1
1
Ethylac-
35
etate
Results and discussion
1
2
Acetone
1
4
120
30
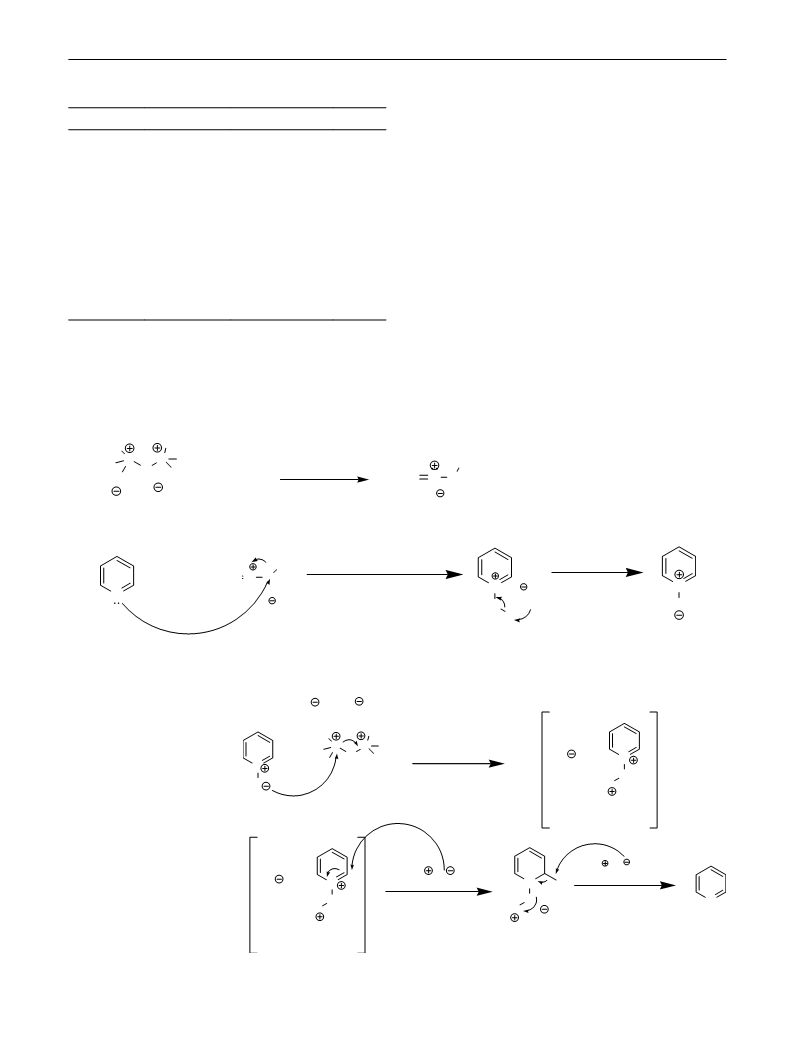
Hexaphenyloxodiphosphonium triflate, known as Hendrick-
Pyridine (1 mmol), H O (4 mmol), Hendrickson reagent (1 mmol) in
2
2
son reagent, was prepared by exothermic reaction of Tf O
EtOH at room temperature
2
a
with Ph PO in CH Cl [41–43]. This salt was indicated to
Isolated yield
3
2
2
1
3

 Khodaei, Mohammad Mehdi
Khodaei, Mohammad Mehdi
 Alizadeh, Abdolhamid
Alizadeh, Abdolhamid
 Hezarkhani, Hadis Afshar
Hezarkhani, Hadis Afshar