
Synthetic Communications p. 805 - 812 (2020)
Update date:2022-08-10
Topics:
 Chen, Xia
Chen, Xia
 Zhou, Xiao-Yu
Zhou, Xiao-Yu
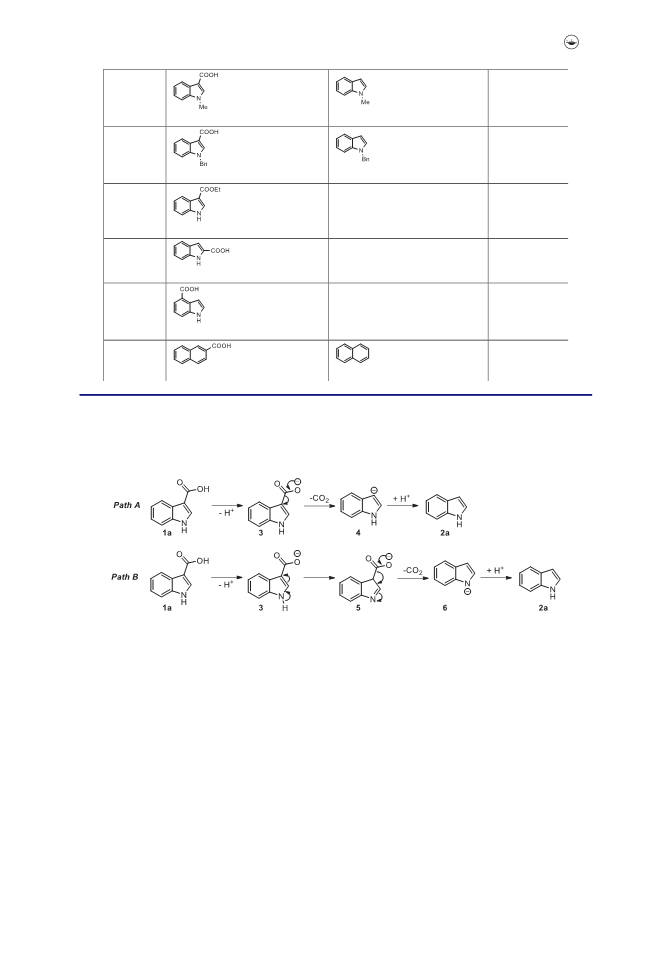
Two reaction systems have been developed for the decarboxylation of indole-3-carboxylic acids. The decarboxylation can be achieved smoothly under K2CO3-catalyzed or acetonitrile-promoted basic conditions. It provided an efficient and simple method for the transformation of indole-3-carboxylic acids and the corresponding indoles were isolated with good to excellent yields. From the experimental facts, we put forward the possible reaction mechanism.
View More







Shenzhen Hongyuan Import & Export Co., Ltd.
Contact:0755-26407171
Address:Shenzhen Hongyuan Chemical New Materials Technology Co., Ltd.
website:http://www.win-winchemical.com
Contact:0086-577-64498589
Address:6F,No.396 Xingping Road,Longwan Industrial Zone
website:http://www.win-winchemical.com
Contact:0086-577-64498589
Address:6F,No.396 Xingping Road,Longwan Industrial Zone
Tianjin Ingenochem Technology Co.,Ltd
Contact:+86-22-23677060
Address:Hitech Green Industry Park K2-9-602, Nankai district
Contact:+86-13914766747
Address:Floors 21&22, Jin Cheng Tower, No. 216 Middle Longpan Road, Nanjing
Doi:10.1021/cs501019t
(2014)Doi:10.1081/SCC-120015803
(2003)Doi:10.1021/acs.joc.6b02841
(2017)Doi:10.1039/j19690000808
(1969)Doi:10.1080/10610278.2013.844811
(2014)Doi:10.1016/S0040-4039(00)01669-5
(2000)