1
762 Journal of Chemical and Engineering Data, Vol. 51, No. 5, 2006
Figure 2. Surfactants synthesized and used in this study. (1) DBSS: n )
, sodium dibutyl sulfosuccinate. (2) DPSS: n ) 4, sodium dipentyl
sulfosuccinate. (3) DHSS: n ) 5, sodium dihexyl sulfosuccinate. (4)
DOSS: n ) 7, sodium dioctyl sulfosuccinate.
3
a
b
c
d
(
1) Sodium dibutyl sulfosuccinate. CH3 CH2 CH2 CH2 OO-
e
f
g
h
i
j
1
CCH2 CH (SO3Na)COOCH2 CH2 CH2 CH3 (yield, 90.4 %). H
NMR (CDCl3, δ): 0.93 (a and j, t, J ) 2.96 Hz, 6H), 1.36 (b and
i, t, J ) 7.68 Hz, 4H), 1.58 (c and h, t, J ) 7.55 Hz, 4H), 3.18
e, t, J ) 9.64 Hz, 2H), 4.05 (d, t, J ) 6.73 Hz, 2H), 4.18 (g,
t, J ) 6.56 Hz, 2H), 4.33 (f, t, J ) 5.12 Hz, 1H). Anal. Calcd:
C, 43.37; S, 9.65; H, 6.37. Found: C, 43.31; S, 10.07; H, 6.41.
(
Figure 1. Schematic diagram of experimental setup: 1, carbon dioxide
cylinder; 2, ISCO model 260D syringe pump; 3, magnetic stir device; 4,
SF-400 high-pressure vessel; 5, pressure transducer; 6, thermocouple
assembly; 7, intake valve; 8, back pressure valve; 9, sample vial; 10,
reclaimer vase; 11, wet-type gas meter.
a
b
c
d
e
(
2) Sodium dipentyl sulfosuccinate. CH3 CH2 CH2 CH2 CH2 -
OOCCH2 CH (SO3Na)COOCH2 CH2 CH2 CH2 CH3 (yield, 95.1
f
g
h
i
j
k
l
1
%
6
(
2
). H NMR (CDCl3, δ): 0.91-0.87 (a and l, t, J ) 3.70 Hz,
H), 1.30-1.35 (b, c, j, and k, m, J ) 3.09 Hz, 8H), 1.67-1.55
d and i, m, J ) 6.89 Hz, 4H), 3.23-3.08 (f, m, J ) 11.76 Hz,
H), 4.04 (e, t, J ) 6.84 Hz, 2H), 4.14-4.18 (h, m, J ) 5.05
Hz, 2H), 4.26-4.31 (g, m, J ) 4.95 Hz, 1H). Anal. Calcd: C,
6.65; S, 8.90; H, 6.99. Found: C, 46.14; S, 8.42; H, 6.71.
Co., and 1,1,1,2-tetrafluoroethane (HFC-134a) (99.9 %) was ob-
tained from Xi’an Jinzhu Modern Chemical Industry Co., Ltd. The
chemical reagents used in this study were of analytically pure
grade.
The schematic diagram of experimental apparatus for solubil-
ity measurement is given in Figure 1. The solubility measure-
ment of surfactants in scCO2 and HFC-134a was investigated
by using a high-pressure vessel (SF-400, Beijing, Sihe Chua-
ngzhi Keji Corporation) with a maximum pressure of 40 MPa,
4
a
b
c
d
(3) Sodium dihexyl sulfosuccinate. CH3 CH2 CH2 CH2 -
CH2 CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 CH2 CH2 CH2 -
e
f
g
h
i
j
k
l
m
n
1
CH3 (yield, 84.7 %). H NMR (CDCl3, δ): 0.90 (a and n, t, J
) 4.59 Hz, 6H), 1.29 (b, c, d, k, l, and m, t, J ) 1.38 Hz, 8H),
1.59 (e and j, t, J ) 6.87 Hz, 4H), 3.15 (g, t, J ) 9.33 Hz, 2H),
4.05 (f, t, J ) 6.87 Hz, 2H), 4.14 (i, t, J ) 6.6 Hz, 2H), 4.33
(h, t, J ) 5.16 Hz, 1H). Anal. Calcd: C, 49.47; S, 8.25; H,
7.52. Found: C, 49.57; S, 8.22; H, 7.41.
a maximum temperature of 353 K, and an internal volume of
3
6
0 cm. The vessel was equipped with two sapphire windows
with a diameter of 25 mm and a thickness of 20 mm. The
windows were sealed on both sides with poly(ether-ether-
ketone) (PEEK) seals. The ISCO model 260D syringe pump
was used to charge CO2 and HFC-134a fluids into the high-
pressure vessel.
a
b
c
d
e
(4) Sodium dioctyl sulfosuccinate. CH3 CH2 CH2 CH2 CH2 -
CH2 CH2 CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 CH2 CH2 -
f
g
h
i
j
k
l
m
n
p
q
r
1
CH2°CH2 CH2 CH3 (yield, 86.6 %). H NMR (CDCl3, δ): 0.88
(a and r, t, J ) 6.75 Hz, 6H), 1.27 (b, c, d, e, f, m, n, o, p, and
q, m, J ) 12.84 Hz, 20H), 1.59 (g and l, m, J ) 5.76 Hz, 4H),
3.14-3.17 (i, m, J ) 4.57 Hz, 2H), 4.02 (h, t, J ) 6.87 Hz,
2H), 4.13-4.18 (k, m, J ) 6.51 Hz, 2H), 4.27-4.32 (j, m, J )
4.04, 1H). Anal. Calcd: C, 54.03; S, 7.21; H, 8.39. Found: C,
54.97; S, 6.99; H, 8.33.
1
The H NMR spectra were recorded on a Superconducting
Fourier digital NMR spectrometer (Bruker, AVANCF 300MHZ).
The elemental analysis of the samples was done by elemental
analyzer (Germany, Vario EL III).
Surfactant Synthesis. The modified synthesis procedures of
surfactants were conducted according to the methods given by
4
2
Liu and Erkey. The surfactants of sodium dibutyl sulfosuc-
cinate, sodium dipentyl sulfosuccinate, sodium dihexyl sulfo-
succinate, and sodium dioctyl sulfosuccinate were synthesized
in an unclosed system and without any extra phase transfer
catalyst. The process was carried out by esterification and
sulfonation reactions.
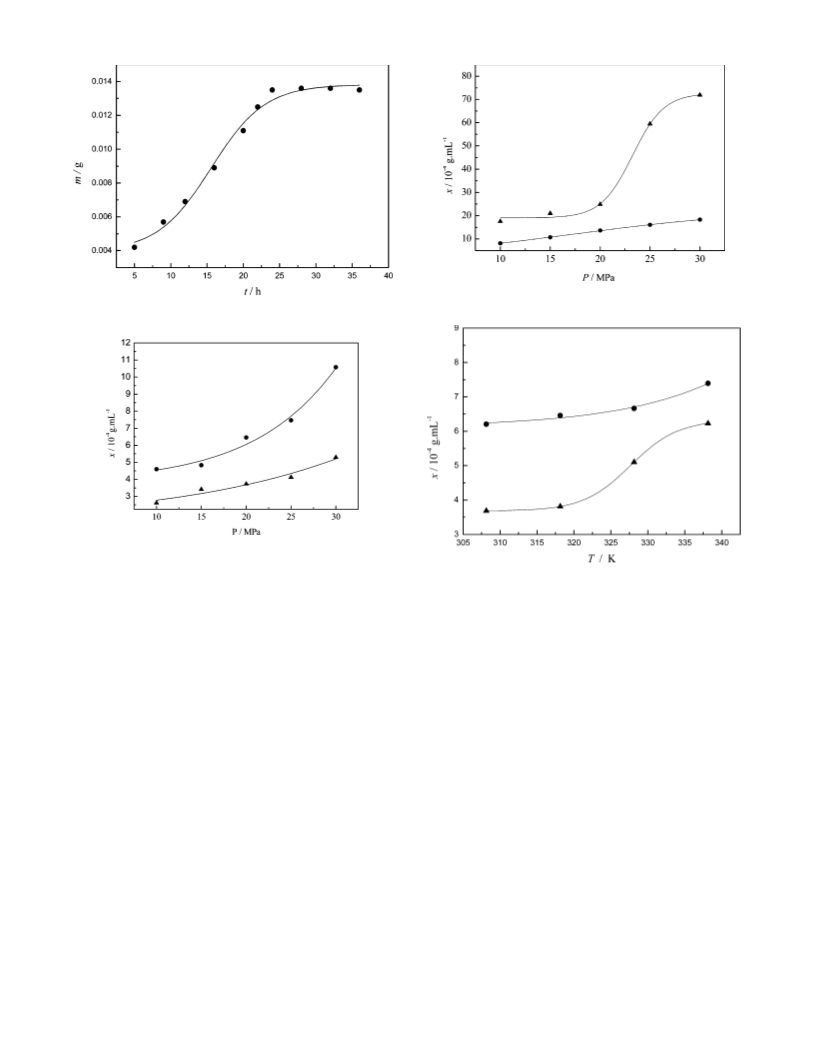
Solubility Measurement. The structures of surfactants syn-
thesized and used to measure solubility are listed in Figure 2.
A variety of methods have been developed to measure the
solubility of solutes in scCO2, which can be classified as
dynamic or static methods. In the dynamic methods, the solute
is loaded into a high pressure extraction vessel that is continu-
ously swept with scCO2, and the solubility is calculated from
an analysis of the solute concentration in the effluent stream.
The static methods may be divided into two categories on the
basis of the type of vessel used. A variable-volume view cell
enables one to determine the solubility by visual inspection of
a cloud point. The use of a fixed-volume cell requires an analysis
of the fluid phase to determine the solubility. Alternatively, one
can couple a fixed volume cell with a suitable high-pressure
spectroscopic method that directly measures the solute concen-
trations in the fluid phase. However, high-pressure absorption
spectroscopy requires special equipment that is usually very
A mixture of maleic anhydride, hydrocarbon alcohol, and
p-toluenesulfonic acid monohydrate as catalyst was refluxed
under stirring; liberated water was removed azeotropically from
the reaction system to shift equilibrium of esterification. Water
created in this reaction was collected in the trap. The reaction
was stopped when about 94 % of the theoretical amount of water
was collected in the trap. Subsequently, the esterifiable product
was neutralized to pH 7 by using aqueous sodium hydroxide
(
30 %), and floc were observed in the system. Then an aqueous
sodium hydrogen sulfite in distilled water and ethanol as
cosolvent were added into the system. The mixture was refluxed
under stirring at a fixed temperature for several hours. The
reaction was stopped if no oily matter floated on the water
surface, and the pH of the reaction system was neutral. A white
solid was obtained after purification, recrystallization, and drying
at 313 K under vacuum overnight.
23
expensive. Sherman et al. reported a simple static method using
a fixed-volume vessel combined with gravimetric analysis for
determining the solubility of solids in scCO2.
The solubility of these surfactants was measured by modifica-
2
3
tion of the procedures given by Sherman et al. In the process
of solubility measurement, an excess amount of surfactant and
a small magnetic stir bar were packed in a 12 mL (25 mm ×
1
The surfactants were characterized with H NMR spectros-
copy and elemental analysis as follows.

 Liu, Zhao-Tie
Liu, Zhao-Tie
 Wu, Jin
Wu, Jin
 Liu, Ling
Liu, Ling
 Sun, Changan
Sun, Changan
 Song, Liping
Song, Liping
 Gao, Ziwei
Gao, Ziwei
 Dong, Wensheng
Dong, Wensheng
 Lu, Jian
Lu, Jian