2
046 Journal of Chemical and Engineering Data, Vol. 51, No. 6, 2006
A mixture of maleic anhydride, n-pentanone (or n-octanol,
n-butanol, n-hexanol), and p-toluenesulfonic acid monohydrate
as the catalyst in a flask was refluxed under stirring at 398 K
for 2 h. The water created in this reaction was collected in a
trap. The yield of this esterification reaction was approximately
0 %.
8
Esterifiable product was neutralized to the pH of 7 using an
aqueous sodium hydroxide (30 %) solution. Floc was observed
in the system. Then an aqueous sodium hydrogen sulfite and
ethanol as cosolvent were added into the system. The mixture
was refluxed under stirring at 393 K for (3 to 4) h. A little of
the product was then taken out into water and stirred to
determine the reaction degree. The reaction was stopped if no
oily matter floated on the water surface and the pH of the
reaction system was (7 to 8). After reacting, the viscous liquid
was created in the flask. This liquid was placed in a dryer to
remove water in the system. The solid obtained was then
dissolved in anhydrous alcohol and recrystallized. A white solid
was obtained after drying at 323 K under vacuum overnight.
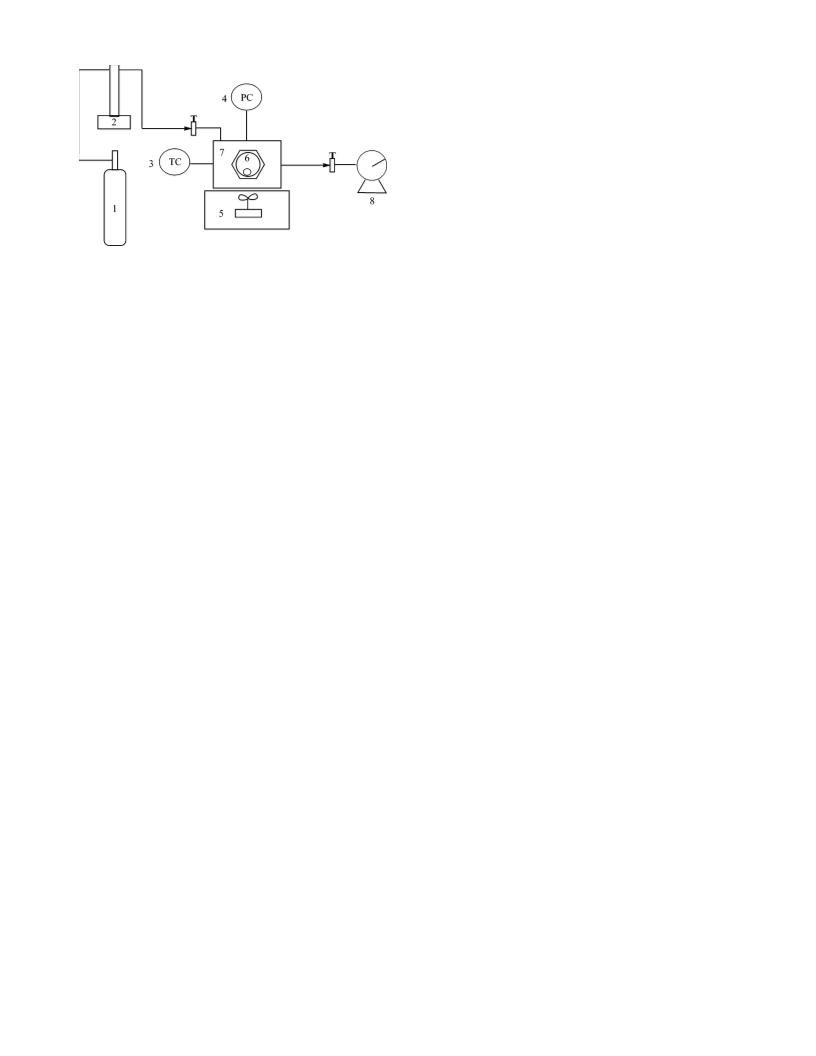
Figure 1. Schematic diagram for solubility and phase diagram measure-
ments: 1, steel cylinder; 2, syringe pump; 3, thermocouple assembly; 4,
pressure transducer; 5, magnetic stirrer; 6, glass vial; 7, high-pressure vessel;
8
, wet-type gas meter.
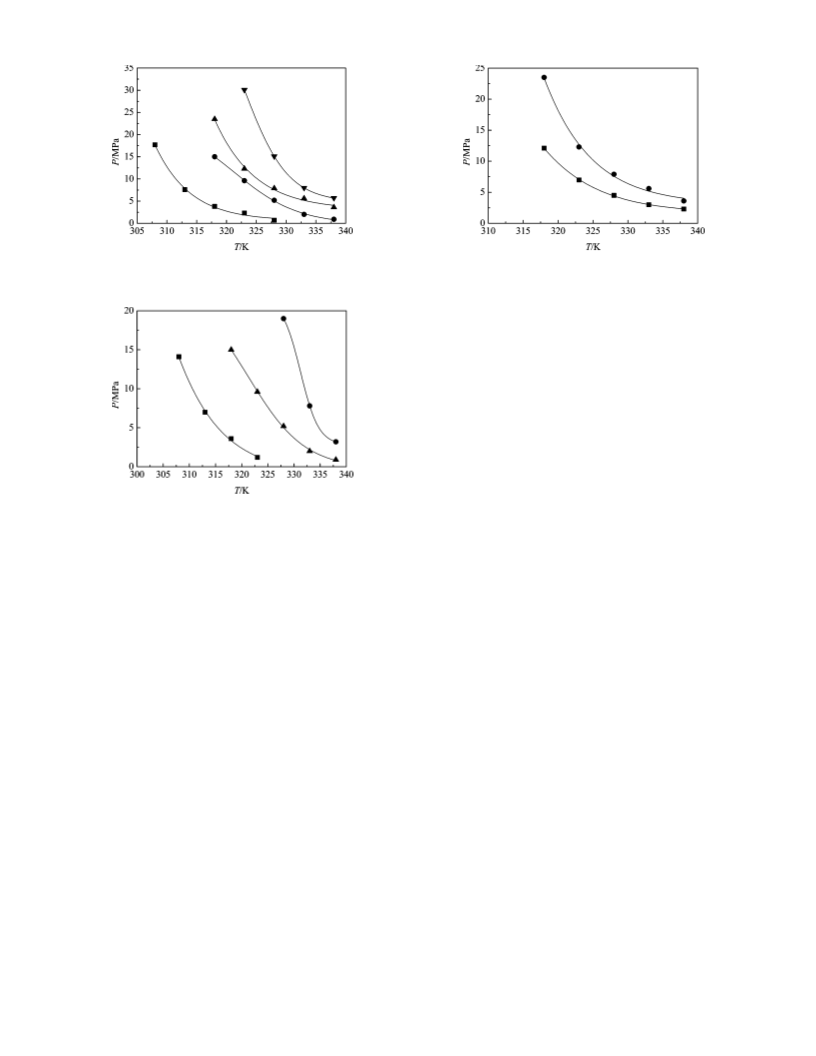
The current work describes the solubilities and pressure-
temperature phase diagrams for W/HFC-134a (water/HFC-134a)
microemulsions stabilized by the surfactants using cloud-point
1
The surfactants were characterized by H NMR spectroscopy
-
3
and elemental analysis as follows.
Sodium Salt of Dioctyl-2-sulfosuccinate. CH3 (CH2)5 CH2 -
measurements for a surfactant concentration of (1.85 × 10
a
b
c
-
3
to 5.60 × 10 ) M, temperature up to 338 K, and pressure up
to 40 MPa. These phase diagrams are important to the formation
of nanoparticales in our further studies.
d
e
f
g
h
j
CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 (CH2)5CH3 (yield,
8
6
1
6.6 %). H NMR (CDCl3, δ): 0.880 (a and j, t, J ) 6.75 Hz,
H), 1.272 (b and i, m, J ) 12.84 Hz, 20H), 1.586 (c and h, m,
Experimental Section
J ) 5.76 Hz, 4H), 3.135-3.166 (e, m, J ) 4.57 Hz, 2H), 4.024
(
4
7
d, t, J ) 6.87 Hz, 2H), 4.131-4.175 (g, m, J ) 6.51 Hz, 2H),
Materials and Instruments. Maleic anhydride (99.5 %),
p-toluenesulfonic acid monohydrate (99 %), 1,4-dioxane (99
.269-4.317 (f, m, J ) 4.04, 1H). Anal. Calcd for C, 54.03; S,
.21; H, 8.39. Found: C, 54.97; S, 6.69; H, 8.33.
%), sodium hydrogen sulfite (SO2, 65.0 %) and n-hexanol (99.0
a
b
c
Sodium Salt of Dipentyl-2-sulfosuccinate. CH3 (CH2)2 CH2 -
%) were obtained from the Sinopharm Group Chemical Reagent
d
e
f
g
h
i
j
CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 (CH2)2 CH3 (yield,
Co. Toluene (99.5 %), n-pentanone (98 %), n-octanol (99.5 %),
and sodium hydroxide (96.0 %) were obtained from the Xi’an
Chemical Reagent Factory. n-Butanol (99.0 %) was obtained
from the Tianjin No. 3 Chemical Reagent Factory. CO2 was
obtained from the Xi’an Yatai Liquid Gas Co., and 1,1,1,2-
tetrafluoroethane (HFC-134a) (99.9 %) was obtained from the
Xi’an Jinzhu Modern Chemical Industry Co., Ltd. The chemical
reagents used in this study were of analytical pure grade. All
the chemicals were used directly without further purification.
A schematic diagram of the experimental setup for solubility
measurements is given in Figure 1. The high-pressure vessel
1
9
3
1
5.1 %). H NMR (CDCl3, δ): 0.908-0.873 (a and j, t, J )
.70 Hz, 6H), 1.300-1.349 (b and i, m, J ) 3.09 Hz, 8H),
.673-1.550 (c and h, m, J ) 6.89 Hz, 4H), 3.228-3.078 (e,
m, J ) 11.76 Hz, 2H), 4.035 (d, t, J ) 6.84 Hz, 2H), 4.140-
.179 (g, m, J ) 5.05 Hz, 2H), 4.262-4.311 (f, m, J ) 4.95
Hz, 1H). Anal. Calcd for C, 46.65; S, 8.90; H, 6.99. Found: C,
6.14; S, 8.42; H, 6.71.
Sodium Salt of Dibutyl-2-sulfosuccinate. CH3 CH2 CH2 -
4
4
a
b
c
d
e
f
g
h
i
j
CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 CH2 CH3 (yield, 90.4
%
1
1
). H NMR (CDCl3, δ): 0.93 (a and j, t, J ) 2.96 Hz, 6H),
.36 (b and i, t, J ) 7.68 Hz, 4H), 1.58 (c and h, t, J ) 7.55
(Beijing, Sihe Chuangzhi Keji Corporation, SF-400) with a
Hz, 4H), 3.18 (e, t, J ) 9.64 Hz, 2H), 4.05 (d, t, J ) 6.73 Hz,
2H), 4.18 (g, t, J ) 6.56 Hz, 2H), 4.33 (f, t, J ) 5.12 Hz, 1H).
Anal. Calcd for C, 43.37; S, 9.65; H, 6.37. Found: C, 43.31;
S, 10.07; H, 6.41.
maximum pressure of 40 MPa, a maximum temperature of 353
3
K, and an internal volume of 60 cm was equipped with two
sapphire windows with a diameter of 25 mm and a thickness
of 20 mm. The windows were sealed on both sides with poly-
a
b
c
Sodium Salt of Dihexyl-2-sulfosuccinate. CH3 (CH2)3 CH2 -
(ether-ether-ketone) (PEEK) seals. The high-pressure vessel
d
e
f
g
h
i
j
CH2 OOCCH2 CH (SO3Na)COOCH2 CH2 (CH2)3 CH3 (yield,
was also fitted with a thermocouple assembly, a pressure
transducer, and a rupture disk assembly. The vessel rests on a
magnetic stirrer, and fluid is introduced into the pressure vessel
using a syringe pump (model 260D, ISCO).
1
8
6
6
4.7 %). H NMR (CDCl3, δ): 0.90 (a and j, t, J ) 4.59 Hz,
H), 1.29 (b and i, t, J ) 1.38 Hz, 8H), 1.59 (c and h, t, J )
.87 Hz, 4H), 3.15 (e, t, J ) 9.33 Hz, 2H), 4.05 (d, t, J ) 6.87
1
Hz, 2H), 4.14 (g, t, J ) 6.60 Hz, 2H), 4.33 (f, t, J ) 5.16 Hz,
The H NMR spectra were recorded on a superconducting
1
4
H). Anal. Calcd for C, 49.47; S, 8.25; H, 7.52. Found: C,
9.57; S, 8.22; H, 7.41.
Fourier digital NMR spectrometer (Bruker, AVANCF 300
MHZ). The elemental analysis of the samples was performed
by an elemental analyzer (Germany, Vario EL III) with a
variation of ( 0.5 %.
Synthesis of Surfactants. The modified synthesis procedures
of surfactants were conducted according to the methods given
by Liu and Erkey.31 The synthesis procedure of the surfactants
Measurements of Solubility. For each experiment, an excess
amount of solute and a small magnetic stirring bar was placed
in a 25 mm × 25 mm (12 mL) glass vial that was then capped
with a coarse filter paper attached to the vial with Teflon tape.
The sample vial was then weighed and placed inside the pressure
vessel.
(sodium salt of dibutyl-2-sulfosuccinate, DBSS; sodium salt of
dipentyl-2-sulfosuccinate, DPSS; sodium salt of dihexyl-2-
sulfosuccinate, DHSS; and sodium salt of dioctyl-2-sulfosuc-
cinate, DOSS) has two main steps: esterification and sulfonation
reaction.
The vessel was sealed and heated to the desired temperature
by a heater via a machined internal heating rod. Once the test
temperature was reached, stirring was initiated, and the vessel
was slowly filled with fluid until the desired pressure was

 Liu, Zhao-Tie
Liu, Zhao-Tie
 Liu, Ling
Liu, Ling
 Wu, Jin
Wu, Jin
 Song, Liping
Song, Liping
 Gao, Ziwei
Gao, Ziwei
 Dong, Wensheng
Dong, Wensheng
 Lu, Jian
Lu, Jian