440
J.-C. Ruan, T. Zhang, S. Wang, J.-Q. Liu, C. Qian, and X.-Z. Chen
Vol 49
Scheme 3. Mechanism of condensation.
Table 3
Response surface experiment encoding table.
Levels
Factors
Code
-1
0
1
T (ꢀC)
n(iodine)/n(4)
n(sulfur)/n(4)
A
B
C
150
0.005
2
160
0.01
2.25
170
0.015
2.5
complete when dimethyl sulfate was completely dropped. The
reaction mixture was filtered while hot to remove solid impu-
rity. The filtrate was performed by rotary evaporator to give
crude product. 3-Methoxy-N-phenyl-benzenamine (4) (27.2 g,
90.5% yield and 99.5 GC purity) was obtained by recrystalli-
zation in a mixture solution (ethanol:water ¼ 2:1, volume ra-
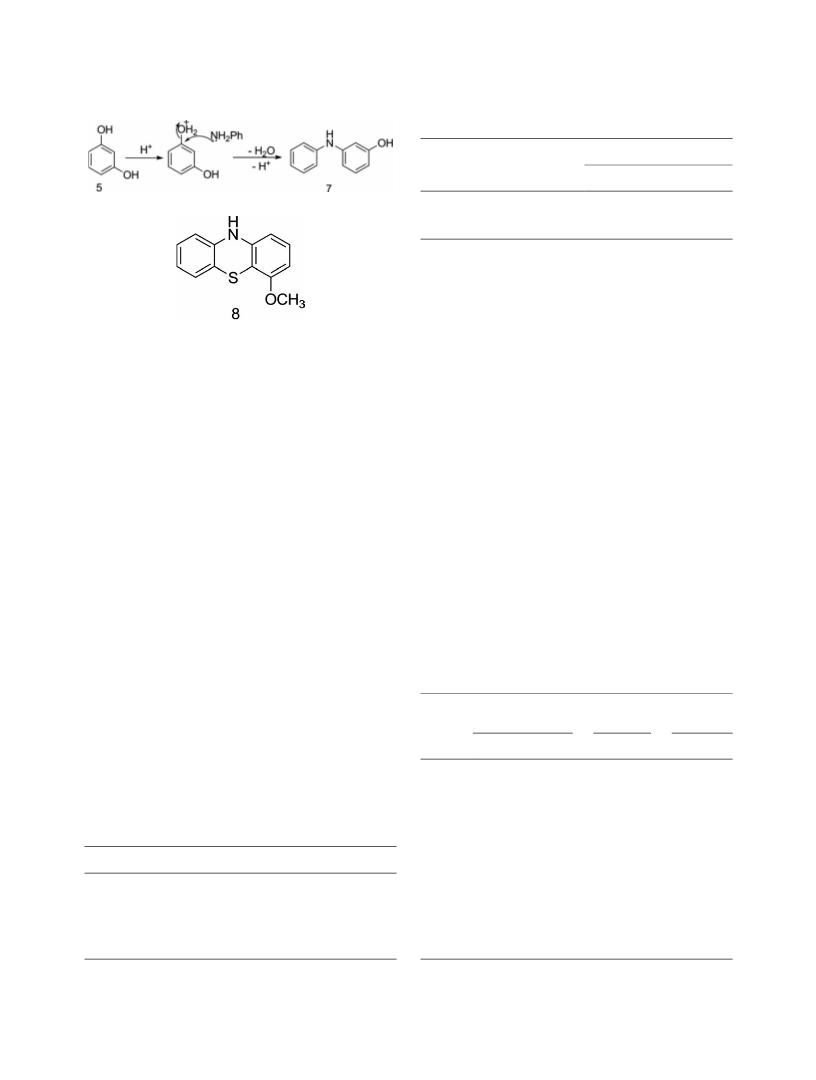
Chart 2. 4-Methoxyphenothiazine (8).
1
tio). Melting point: 69.6–70.7ꢀC (lit. 1 mp:70–71ꢀC); H NMR
(400 MHz, CDCl3) d: 3.83(s, 3H), 5.77(s, 1H), 6.54–6.56(d,
1H), 6.70–6.72(d, 2H), 6.98–7.02(t, 1H), 7.13–7.15(d, 2H),
7.20–7.25(t, 1H), 7.31–7.35(t, 2H).
analyses were performed on glass plate (30 ꢁ 100 mm2). 1H
NMR spectrums were recorded in dimethyl sulfoxide (DMSO)
or CDCl3 using 400 MHz on a NMR spectrometer (Bruker,
AV 400).
Starting materials and reagents were commercially pur-
chased and used without further purification.
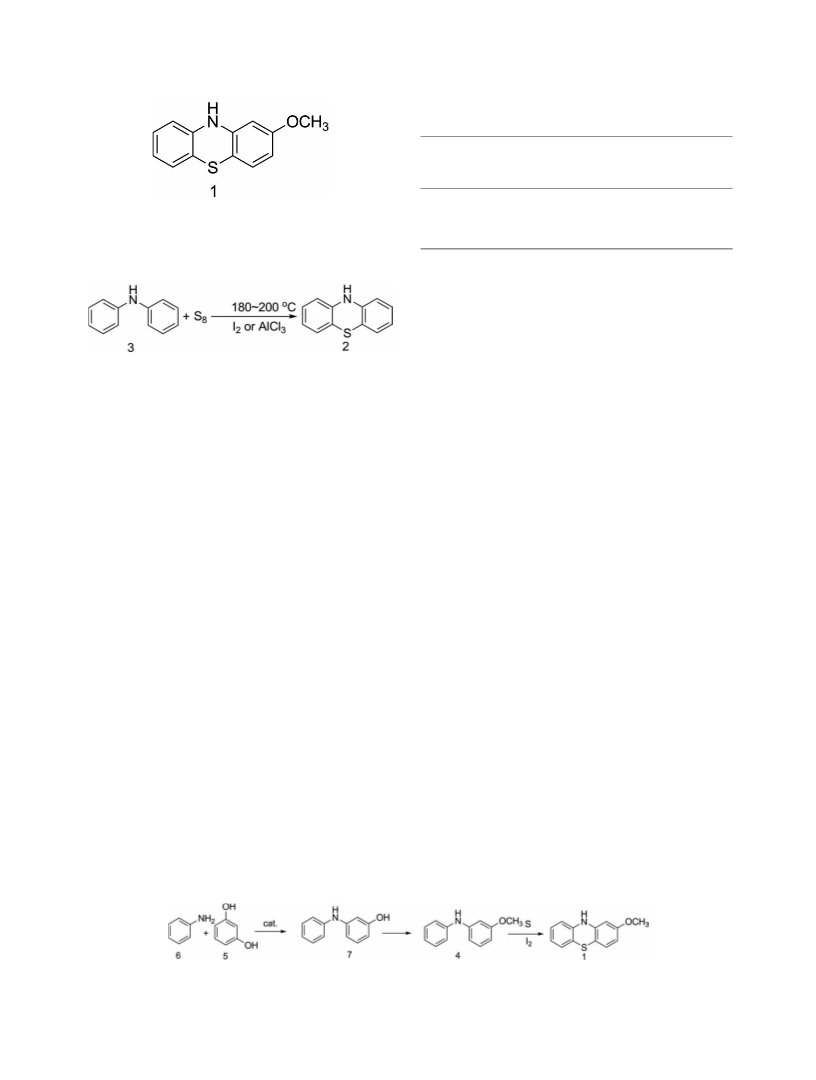
Preparation of 2-methoxyphenothiazine (1). A 500-mL
four-necked flask with a mechanic stirrer, a reflux condenser,
a thermometer and a tail gas absorber, was charged with poly-
substituted aromatics (180 mL). To the flask, 3-methoxy-N-
phenylbenzenamine (4) (50.0 g, 0.25 mol) and sulfur (17.8 g,
0.56 mol) were added under stirring. After the addition was
complete, the mixture was heated to 163ꢀC. Iodine (0.55 g)
was added after the temperature was stable. The reaction was
monitored by TLC. The reaction was complete when the 3-
methoxy-N-phenyl-benzenamine (4) disappeared. Thereafter,
the reaction mixture was cooled to 120ꢀC to give a black
layer and a yellow layer. The yellow layer was obtained and
cooled to 20ꢀC to give yellow solid. 2-Methoxyphenothiazine
(1) (50.0 g, 88.2% yield and 99.8% HPLC purity) was
obtained by recrystallization in a mixture solution (ethanol:-
water ¼ 2:1, volume ratio). Melting point: 183.9–185.4ꢀC
Preparation of 3-hydroxydiphenylamine (7). A 250-mL
four-necked flask equipped with a mechanic stirrer, a ther-
mometer, a distilling apparatus and a water separator, charged
with resorcinol (5) (16.5 g, 0.15 mol) and aniline (6) (16.7 g,
0.18 mol) was heated until the solid completely dissolved.
p-Toluenesulfonic acid (0.5 g) was added into the mixture
under stirring. The reaction was run under vigorous stirring at
190ꢀC, and the water produced was separated. The reaction
was complete until no water produced. The excess aniline (5)
was distilled under vacuum to give solid. The solid was
washed by water, then dried to give 3-hydroxydiphenylamine
(7) (25.6 g, 92.4% yield and 99.1% GC purity) as dull-red
solid. Melting point: 77.5–77.9ꢀC (lit. 16; mp: 76–79ꢀC); 1H
NMR (400 MHz, DMSO): d 6.24(d, 1H), 6.48(d, 1H), 6.51–
6.52(t, 1H), 6.77–6.81(t, 1H), 6.96–7.00(t, 1H), 7.03–7.05(d,
2H), 7.19–7.22(t, 2H), 8.01(s, 1H, NAH), 9.16(s, 1H, OAH).
Preparation of 3-methoxy-N-phenyl-benzenamine (4). To
a 250-mL three-necked flask with a mechanic stirrer, a ther-
mometer and a water separator, 3-hydroxydiphenylamine (7)
(28.0 g, 0.15 mol), toluene (100 mL), sodium hydroxide (7.2
g, 0.18 mol), and water (5 mL) were added. Yellow crystal
was precipitated when no water was separated. To this yellow
crystal, dimethyl sulfate (24.0 g, 0.19 mol) was added slowly
in 5 h, keeping the temperature below 50ꢀC. The reaction was
Table 4
Experimental conditions with corresponding measured and calculated
responses of the reaction for synthesis of 1.
Experimental
variables
Experimental
yield
Responses
No.
A
B
C
y (%)
Y (%)
1
2
3
150
170
150
170
150
170
150
170
160
160
160
160
160
160
160
0.005
0.005
0.015
0.015
0.01
2.25
2.25
2.25
2.25
2
71.5
78.7
75.9
80.4
74.5
78.4
70.6
76.5
76.1
79.9
75.1
78.3
85.2
84.5
83.6
71.6
78.2
76.2
80.2
73.8
78.1
70.7
77.0
76.5
80.1
74.7
77.7
84.3
84.3
84.3
4
5
Table 2
6
0.01
0.01
0.01
2
Results from solvent screening for the synthesis of 1.
7
8
2.5
2.5
2
Solvent
Time (h)
T (ꢀC)
Yield (%)
9
0.005
0.015
0.005
0.015
0.01
Free
1.5
1.5
1.5
1.5
1.5
1.5
140
118
140
160
125
160
64.0
72.2
74.3
86.1
71.2
85.3
10
11
12
13
14
15
2
n-Butyl alcohol
N,N-Dimethylformamide
Sulfolane
2.5
2.5
2.25
2.25
2.25
n-Octane
Polysubstituted aromatics
0.01
0.01
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Ruan, Jian-Cheng
Ruan, Jian-Cheng
 Zhang, Tao
Zhang, Tao
 Wang, Shuai
Wang, Shuai
 Liu, Jin-Qiang
Liu, Jin-Qiang
 Qian, Chao
Qian, Chao
 Chen, Xin-Zhi
Chen, Xin-Zhi