J. Y. Kim, J. C. Park, A. Kim, A. Y. Kim, H. J. Lee, H. Song, K. H. Park
SHORT COMMUNICATION
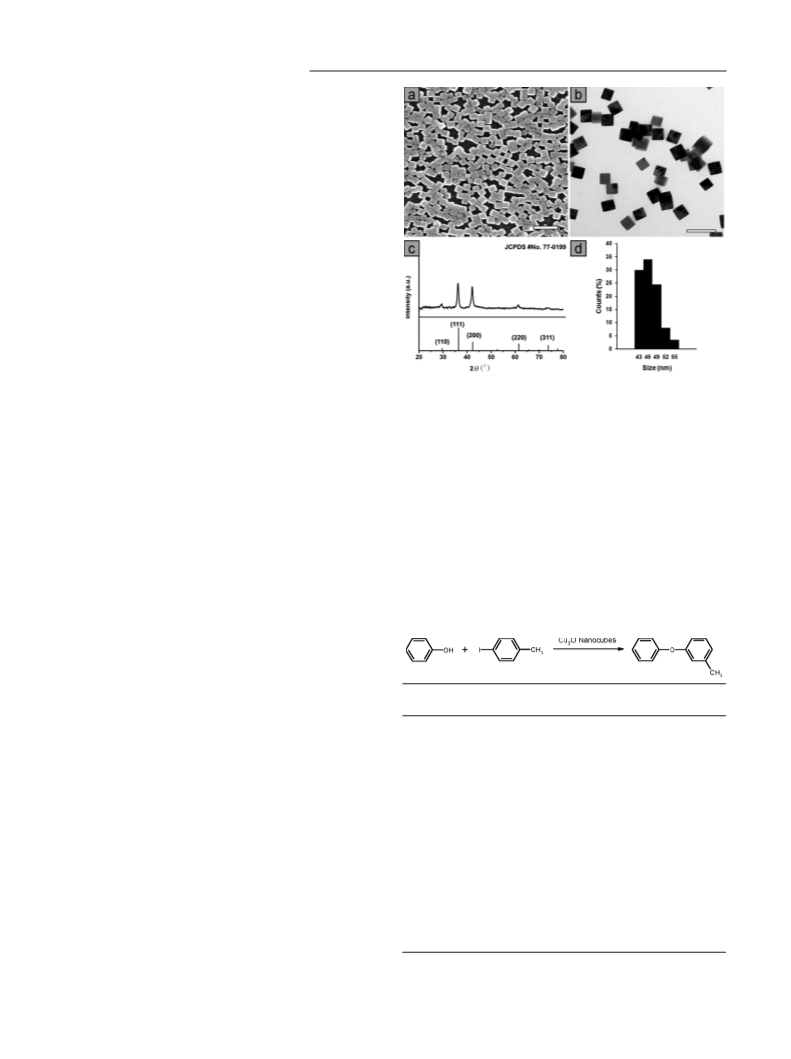
Experimental Section
[
1] a) D. Ma, Q. Cai, Acc. Chem. Res. 2008, 41, 1450–1460; b) J.
Hassan, M. Sévignon, C. Gozzi, E. Schulz, M. Lemaire, Chem.
Rev. 2002, 102, 1359–1470; c) L. Kurti, B. Czako, Strategic
Applications of Named Reactions in Organic Synthesis, Elsevier
Academic Press, California, USA, 2005; d) S. V. Ley, A. W.
Thomas, Angew. Chem. Int. Ed. 2003, 42, 5400–5449. For Pd-
catalyzed C–O bond formation, see: e) Q. Shelby, N. Kataoka,
G. Mann, J. F. Hartwig, J. Am. Chem. Soc. 2000, 122, 10718–
Preparation of Cu
dissolved in PD (Aldrich, 96%; 45 mL) was heated to 240 °C under
an inert atmosphere. Then, Cu(acac) (STREM, 98%; 4.0 mmol)
dissolved in PD (15 mL) was injected into the hot PVP solution at
40 °C, and the mixture was stirred for 15 min at the same tempera-
2
O Nanocubes: PVP (Aldrich, Mw 55,000; 5.3 g)
2
2
ture. The colloidal dispersion was cooled to room temperature, and
the product was separated by adding acetone (150 mL) and centri-
fuging at 8000 rpm for 20 min. The precipitates were washed with
ethanol several times and re-dispersed in ethanol (50 mL).
10719; f) K. E. Torraca, X. Huang, C. A. Parrish, S. L. Buch-
wald, J. Am. Chem. Soc. 2001, 123, 10770–10771; g) C. A. Par-
rish, S. L. Buchwald, J. Org. Chem. 2001, 66, 2498–2500; h) N.
Kataoka, Q. Shelby, J. P. Stambuli, J. F. Hartwig, J. Org. Chem.
Catalytic Reactions: To a 25 mL stainless steel reactor were added
2
002, 67, 5553–5566; i) A. V. Vorogushin, X. Huang, S. L.
Cu
2
O
nanocubes (2 mg, 0.1 mol-%), iodobenzene (1.6 mL,
CO (9.1 g, 0.028 mol),
Buchwald, J. Am. Chem. Soc. 2005, 127, 8146–8149; j) M. C.
Willis, D. Taylor, A. T. Gillmore, Tetrahedron 2006, 62, 11513–
0.014 mol), phenol (1.3 g, 0.014 mol), Cs
2
3
and THF (10 mL). The reaction mixture was stirred at 150 °C in
air. After three hours, the reaction mixture was poured into satu-
1
1520. For Cu-catalyzed C–O bond formation, see: k) J. Zhu,
B. A. Price, S. X. Zhao, P. M. Skonezny, Tetrahedron Lett.
2000, 41, 4011; l) H. L. Aalten, G. van Koten, D. M. Grove, T.
Kuilman, O. G. Piekstra, L. A. Hulshof, R. A. Sheldon, Tetra-
hedron 1989, 45, 5556; m) R. G. R. Bacon, S. C. Rennison, J.
Chem. Soc. C 1969, 312; n) C. E. Castro, R. Havlin, V. K. Hon-
wad, A. Malte, S. Moje, J. Am. Chem. Soc. 1969, 91, 6464.
rated aqueous NaHCO
tracted with dichloromethane (3ϫ20 mL) and dried with MgSO
The Cu O nanocubes were recovered by centrifugation, and the
3
solution (20 mL). The product was ex-
4
.
2
clean solution was analyzed by 300 MHz NMR spectroscopy.
1H NMR Spectroscopic Data
[2] a) C. H. Burgos, T. E. Barder, X. Huang, S. L. Buchwald, An-
gew. Chem. Int. Ed. 2006, 45, 4321–4326; b) M. C. Willis, J.
Chauhan, W. G. Whittinham, Org. Biomol. Chem. 2005, 3,
[
10a]
2A,[10a] 3A,[10b] 4A,[10c] 5A,[10d] 6A,[10e] and
Compounds 1A,
[
10a]
7A
are known compounds.
3094–3095; c) N. Marion, E. C. Ecarnot, O. Navarro, D. Amo-
roso, A. Bell, S. P. Nolan, J. Org. Chem. 2006, 71, 3816–3821;
d) D. Zim, S. L. Buchwald, Org. Lett. 2003, 5, 2413–2415; e)
C. M. So, Z. Zhou, C. P. Lau, F. Y. Kwong, Angew. Chem. Int.
Ed. 2008, 47, 6402–6406.
1
Diphenyl ether (1A): H NMR (CDCl
(
3
, 300 MHz): δ = 7.10–7.17
m, 4 H), 7.19–7.22 (m, 2 H); 7.36–7.44 (m, 4 H) ppm.
1
1
-Methyl-4-phenoxybenzene (2A): H NMR (CDCl
2.43 (s, 3 H), 7.09 (d, J = 8.4 Hz, 2 H), 7.14–7.19 (m, 3 H), 7.22
d, J = 8.1 Hz, 2 H), 7.40 (t, J = 7.8 Hz, 2 H) ppm.
3
, 300 MHz): δ
[
3] a) D. M. T. Chan, K. L. Monaco, R.-P. Wang, M. P. Winteres,
Tetrahedron Lett. 1998, 39, 2933–2936; b) D. A. Evans, J. L.
Katz, T. R. West, Tetrahedron Lett. 1998, 39, 2937–2940; c)
T. D. Quach, R. A. Batey, Org. Lett. 2003, 5, 1381–1384; d)
M. L. Kantam, G. T. Venkanna, C. Sridhar, B. Sreedhar, B. M.
Choudary, J. Org. Chem. 2006, 71, 9522–9524; e) B. Sreedhar,
G. T. Venkanna, K. B. S. Kumar, V. Balasubrahmanyam, Syn-
thesis 2008, 795–799.
[4] a) R. A. Altman, A. Shafir, P. A. Lichtor, S. L. Buchwald, J.
Org. Chem. 2008, 73, 284–286; b) Y.-J. Chen, H.-H. Chen, Org.
Lett. 2006, 8, 5609–5612; c) D. Ma, Q. Cai, Org. Lett. 2003,
5, 3799–3802; d) H.-J. Cristau, P. P. Cellier, S. Hamada, J.-F.
Spindler, M. Taillefer, Org. Lett. 2004, 6, 913–916; e) F. Y.
Kwong, A. Klapars, S. L. Buchwald, Org. Lett. 2002, 4, 581–
=
(
1
1
-Methoxy-4-phenoxybenzene (3A): H NMR (CDCl
δ = 3.78 (s, 3 H), 6.85–6.91 (m, 2 H), 6.94–6.99 (m, 4 H), 7.01–7.06
m, 1 H), 7.26–7.32 (m, 2 H) ppm.
3
, 300 MHz):
(
1
1
6
7
-Nitro-4-phenoxybenzene (4A): H NMR (CDCl
.81–6.83 (m, 2 H), 7.00–7.06 (m, 2 H), 7.22–7.25 (m, 1 H), 7.61–
.65 (m, 2 H), 8.06–8.09 (m, 2 H) ppm.
3
, 300 MHz): δ =
1
1
-Methyl-3-phenoxybenzene (5A): H NMR (CDCl
2.20 (s, 3 H), 6.70–6.73 (m, 2 H), 6.79 (d, J = 9.6 Hz, 1 H), 6.90
d, J = 7.8 Hz, 2 H), 6.94–6.99 (m, 1 H), 7.03–7.11 (m, 1 H), 7.17–
3
, 300 MHz): δ
=
(
5
84; f) C. Wolf, S. Liu, X. Mei, A. T. August, M. D. Casimir,
7.20 (m, 2 H) ppm.
J. Org. Chem. 2006, 71, 3270–3273; g) Y. Pan, H. Lu, Y. Fang,
X. Fang, L. Chen, J. Qian, J. Wang, C. Li, Synthesis 2007,
1242–1246; h) Y. Fang, C. Li, J. Am. Chem. Soc. 2007, 129,
8092–8093.
2
7
(
-Naphthyl phenyl ether (6A): 1H NMR (CDCl
.01–7.16 (m, 3 H), 7.25–7.27 (m, 1 H), 7.25–7.47 (m, 5 H), 7.69
d, J = 7.5 Hz, 1 H), 7.80–7.83 (m, 2 H) ppm.
3
, 300 MHz): δ =
[
[
5] Z. Liu, R. C. Larock, J. Org. Chem. 2006, 71, 3198–3209.
6] For recent reviews, see: a) C. N. R. Rao, G. U. Kulkarni, P. J.
Thomas, P. P. Edwards, Chem. Eur. J. 2002, 8, 28–35; b) H.
Schmidt, Appl. Organomet. Chem. 2001, 15, 331–343; c) H.
Bönnemann, R. M. Richards, Eur. J. Inorg. Chem. 2001, 2455–
1
1
-Methyl-2-phenoxybenzene (7A): H NMR (CDCl
7.22–7.30 (m, 3 H), 7.08–7.16 (m, 1 H), 6.92–6.99 (m, 2 H), 6.85–
.90 (m, 3 H), 2.29 (s. 3 H) ppm.
3
, 300 MHz): δ
=
6
Supporting Information (see footnote on the first page of this arti-
cle): Magnified TEM images of Cu
photo of the reaction vessel.
2
480; d) C. N. R. Rao, A. K. Cheetham, J. Mater. Chem. 2001,
2 2
O, XRD pattern of Cu O, and
11, 2887–2894; e) C. N. R. Rao, G. U. Kulkarni, P. J. Thomas,
P. P. Edwards, Chem. Soc. Rev. 2000, 29, 27–35; f) J. F. Ciebien,
R. T. Clay, B. H. Sohn, R. E. Cohen, New J. Chem. 1998, 22,
685–691; g) C. Burda, X. B. Chen, R. Narayanan, M. A. El-
Sayed, Chem. Rev. 2005, 105, 1025–1102.
7] a) D. Astruc, F. Lu, J. R. Aranzaes, Angew. Chem. Int. Ed.
[
2
005, 44, 7852–7872; b) R. J. White, R. Luque, V. L. Budarin,
J. H. Clark, D. J. Macquarrie, Chem. Soc. Rev. 2009, 38, 481–
94; c) D. Astruc, Nanoparticles and Catalysis, Wiley-VCH,
Acknowledgments
4
This research was supported by the Basic Science Research Pro-
gram through the National Research Foundation of Korea (NRF)
funded by the Ministry of Education, Science and Technology
Weinheim, Germany, 2008.
[
8] a) P. Poizot, S. Laruelle, S. Grugeon, L. Dupont, J.-M. Taras-
con, Nature 2000, 407, 496–499; b) M. Izaki, T. Shinagawa, K.
Mizuno, Y. Ida, M. Inaba, A. Tasaka, J. Phys. D 2007, 40,
3326–3329; c) J. Zhang, J. Liu, Q. Peng, X. Wang, Y. Li, Chem.
(
(
2009-0070926) and by the Korean Research Foundation Grant
KRF-2006-312-C00565).
4222
www.eurjic.org
© 2009 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Eur. J. Inorg. Chem. 2009, 4219–4223

 Kim, Jee Young
Kim, Jee Young
 Park, Ji Chan
Park, Ji Chan
 Kim, Aram
Kim, Aram
 Young Kim
Young Kim
 Lee, Hyun Ju
Lee, Hyun Ju
 Song, Hyunjoon
Song, Hyunjoon
 Park, Kang Hyun
Park, Kang Hyun