Y. Lei et al. / Journal of Molecular Catalysis A: Chemical 398 (2015) 164–169
167
Table 2 (Continued)
Entry
Ar-I
R2OH
EtOH
Product
Yield (%)b
1
3
4
91
93
1
EtOH
EtOH
1
5
82
1
1
6
7
EtOH
EtOH
94
82
a
◦
Reaction conditions: Pd catalyst (1.0 mol%), aryl iodide (0.5 mmol), DBU (1.5 mmol), alcohol (3 mL), 80 C, 6 h, CO (1 atm).
Isolated yield.
b
plane bending vibrations of the benzene ring, respectively [47,51].
The peaks at 1437 cm correspond to the vibrations of the P CH2
obtained was slightly inferior compared with that of 1.0 mol% cat-
alyst (Table 1, entry 16). Thus, the optimal conditions are: DBU as
the base, and 1.0 mol% catalyst.
−
1
bond [47,52]. The Pd content of Pd@KAPs(Ph-PPh ) was 0.8 wt%, as
3
measured by AAS.
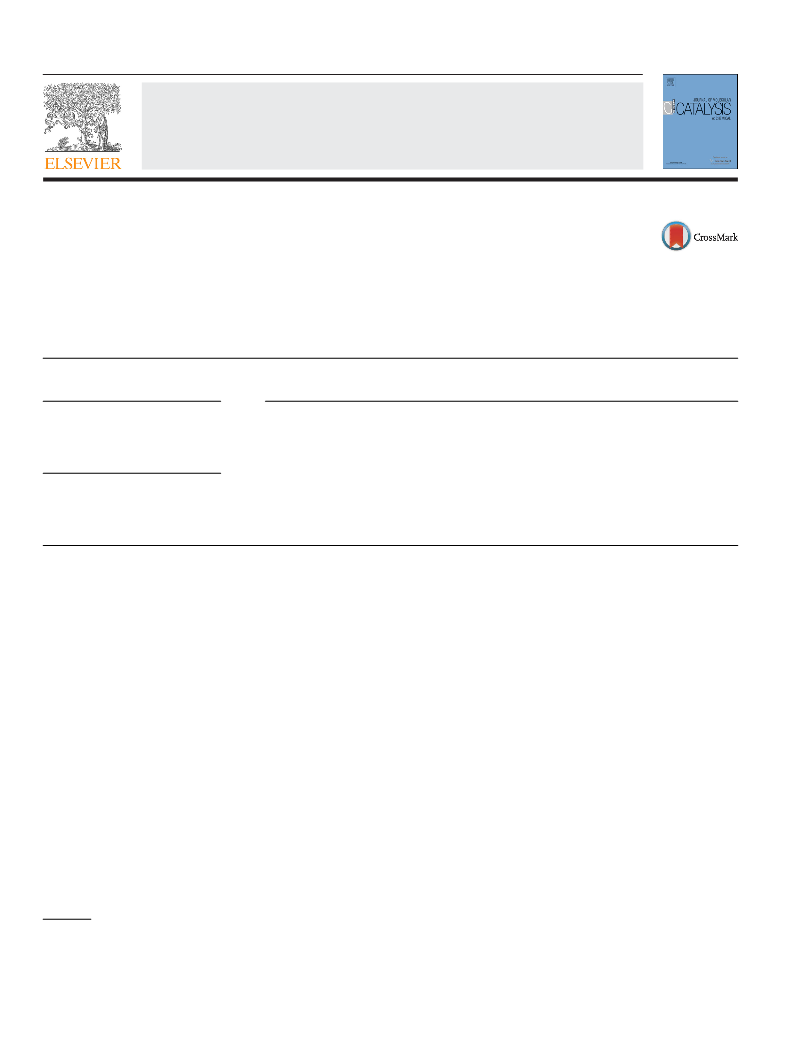
Then, we turned our focus to the scope and limitations of the sys-
tem. Firstly, the reaction of iodobenzene with different alcohols was
investigated, and the results are summarized in Table 2. Iodoben-
zene reacted efficiently with primary alcohols, such as methanol,
n-propanol, n-butanol, and benzyl alcohol, and the correspond-
ing benzoates were obtained in excellent yields (Table 2, entries
1–4). When secondary alcohols, such as isopropanol and cyclohex-
anol, were used, moderate yields were obtained (Table 2, entries
5 and 6). Unfortunately, no desired product could be detected
when t-butanol was used as a nucleophile. This might result from
the steric hindrance effect of tert-butyl group that impedes the
coordination of alcohol with the metal center (Table 2, entry 7)
[53]. Next, the alkoxycarbonylation of various aryl iodides with
ethanol was also investigated. Aryl iodides bearing both electron-
withdrawing and electron-donating groups participated in the
reactions readily, affording the corresponding benzoates in moder-
ate to excellent yields (Table 2, entries 8–15). Interestingly, when
methyl 4-iodobenzoate was used as substrate, an unsymmetrical
dialkyl terephthalate was obtained in 82% yield. Considering the
possible effect of transesterification reaction, this result is quite
remarkable (Table 2, entry 15). 3-Iodopyridine, as a representa-
tive example of aromatic heterocyclic iodide, resulted in 94% yield
of ethyl nicotinoate (Table 2, entry 16). Benzyl chloride gave the
desired product in 82% yield (Table 2, entry 17). Indeed, organic bro-
mides, such as bromobenzene, 3-bromopyridine, -bromostyrene
were also tested, unfortunately, no target products were obtained.
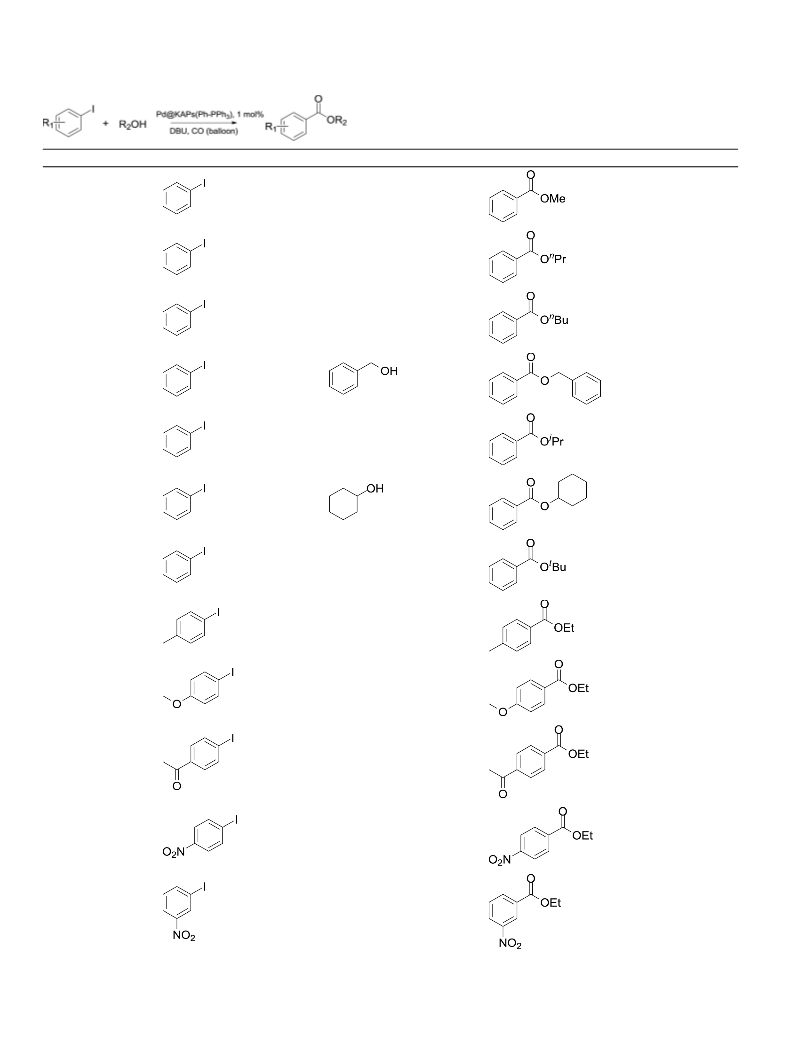
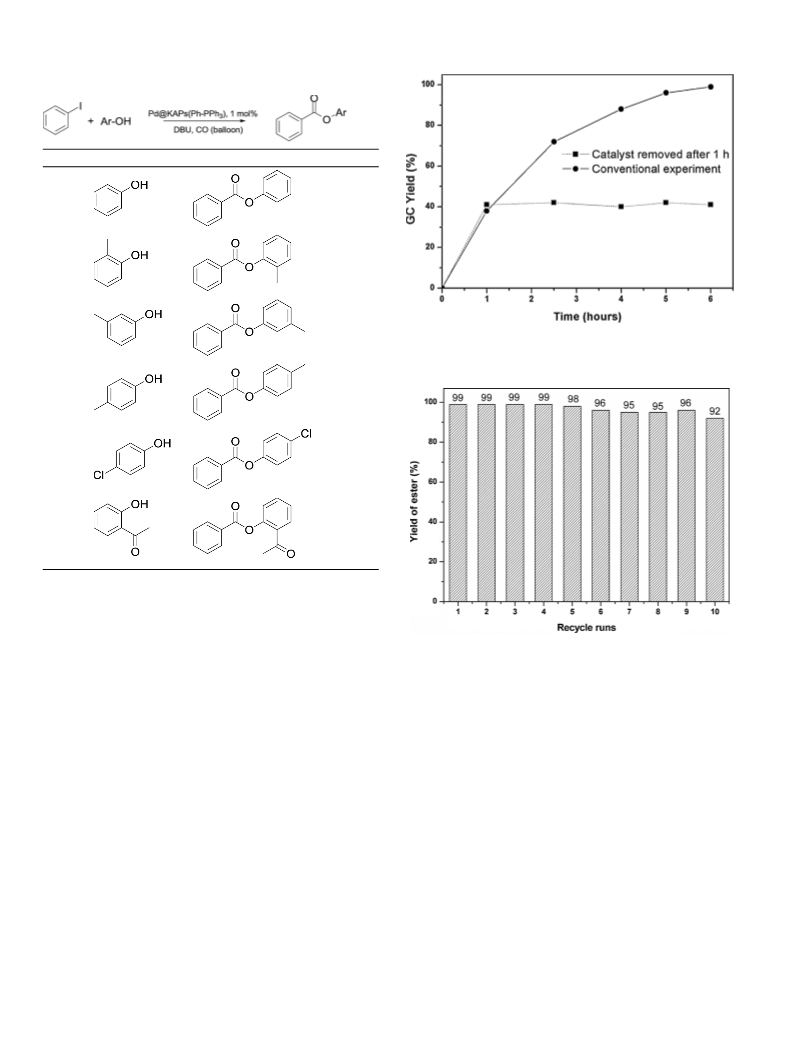
This catalyst was also used in the alkoxycarbonylation of aryl
iodides with phenols, so called phenoxycarbonylation. Consider-
ing the fact that the nucleophilicity of phenol is lower than that of
ethanol, reaction time of the phenoxycarbonylation was increased
To evaluate the catalytic activity of the thereby obtained
Pd@KAPs(Ph-PPh ) catalyst, ethoxycarbonylation of iodobenzene
3
was chosen as a model reaction. The reaction was performed in
ethanol under atmospheric CO pressure. Firstly, the influences of
catalysts and bases were examined on the model reaction, and the
obtained results are summarized in Table 1. Pd@KAPs(Ph-PPh )
gave 76% yield of ethyl benzoate within 6 h at 80 C using 1 mol%
3
◦
Pd in the presence of Et N (Table 1, entry 1). Some other hetero-
geneous Pd catalysts, such as Pd/C, Pd/Fe O , and PdCl (phen)@Y,
3
3
4
2
were also tested under the identical reaction conditions, and the
yields of ethyl benzoate reached only to 34%, 19%, and 17%, respec-
tively (Table 1, entries 2–4).
It is well known that, in the carbonylation of aryl iodide, bases
can remarkably influence the catalytic activity and selectivity
[
29–45]. To clarify the effect of base in Pd@KAPs(Ph-PPh )-
3
catalyzed reaction, various organic and inorganic bases were then
tested (Table 1, entries 5–15). It was observed that the base indeed
strongly affected the reaction selectivity and activity. When strong
inorganic bases, such as K CO , Cs CO , Na CO , and K PO ·3H O,
2
3
2
3
2
3
3
4
2
were used, significant amount of benzene that was generated
through a reductive deiodination of iodobenzene was detected
(Table 1, entries 5–8). Weak inorganic bases, such as NaHCO3 and
KHCO , worked well, and the yields of ethyl benzoate reached
3
to 86% and 91%, respectively (Table 1, entries 9 and 10). Among
all the bases screened, DBU (1,8-diazabicyclo[540]undec-7-ene)
displayed the best performance, with which 99% yield could be
obtained (Table 1, entry 13). Effect of the catalyst loading was also
studied by using DBU as a base. When 0.5 mol% of Pd@KAPs(Ph-
PPh ) was used, the reaction proceeded also well, but the yield
3

 Lei, Yizhu
Lei, Yizhu
 Wu, Linjuan
Wu, Linjuan
 Zhang, Xuefeng
Zhang, Xuefeng
 Mei, Hui
Mei, Hui
 Gu, Yanlong
Gu, Yanlong
 Li, Guangxing
Li, Guangxing