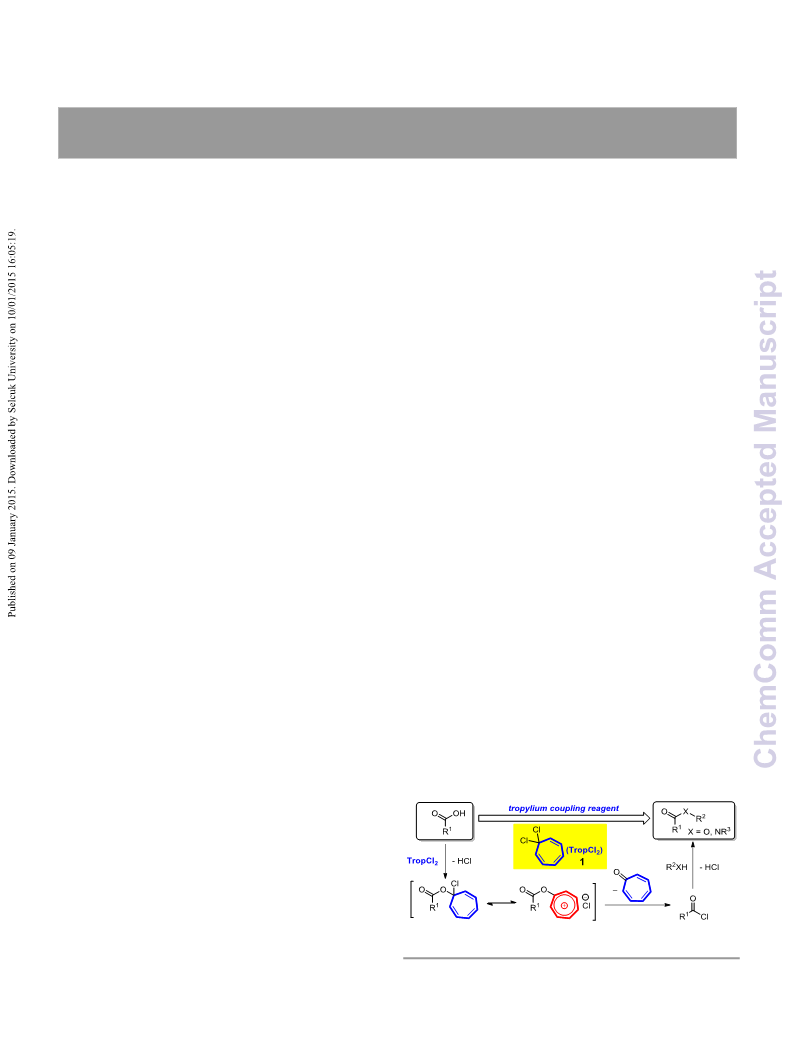
DOI: 10.1039/C4CC09539A
ChemComm
COMMUNICATION
Chemical Communication
suppressants such as HOBt to bioactive macropeptide and
polypeptide coupling reactions is currently underway in our
group and will be reported shortly.
We thank Curtin University for funding the work. Generous
laboratory support from Dr. Alan Payne (Curtin) is gratefully
acknowledged.
Notes and references
a
Department of Chemistry, Curtin University, Perth, WA 6102,
Australia. Tel.: +61-8-9266-3102; Fax: +61-8-9266-2300; E-mail:
†
Electronic Supplementary Information (ESI) available: [Experimental
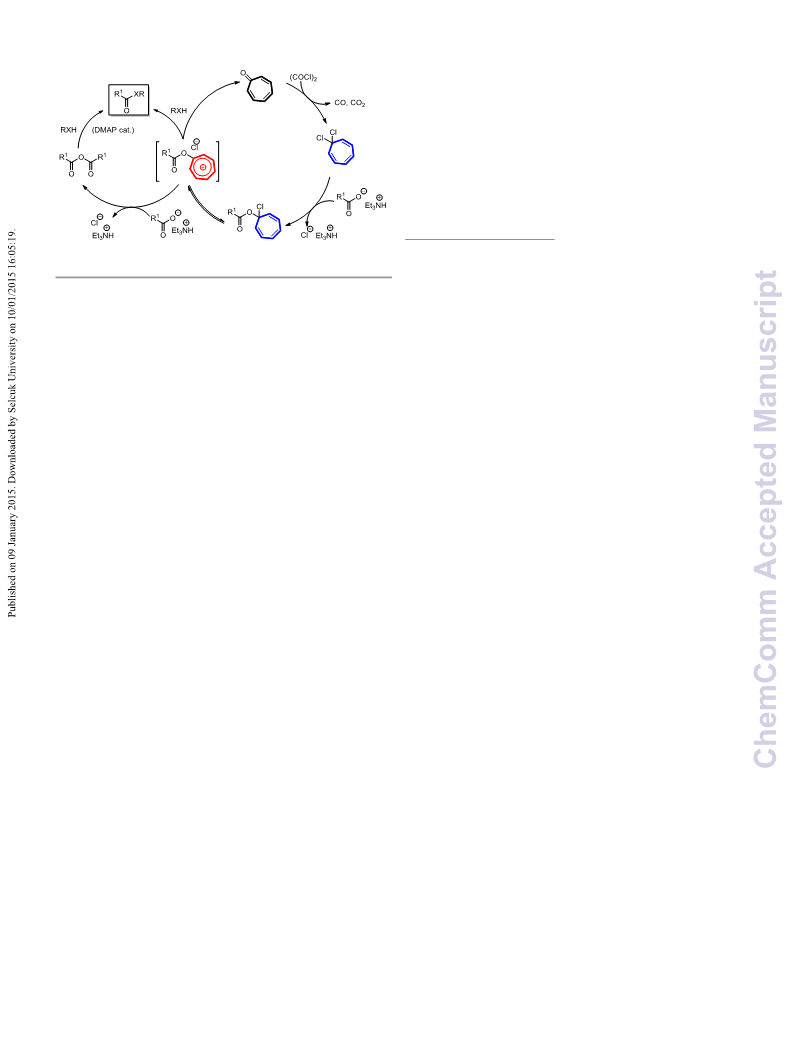
Scheme 2. Proposed mechanism for the tropone-catalyzed coupling
reaction of carboxylic acids
procedures and characterization data]. See DOI: 10.1039/c000000x/
more nucleophilic than the alcohol substrates and quickly
reacted with the newly formed tropylium ester intermediates to
form the anhydrides. The formation of acid chlorides from the
tropylium ester intermediates, however, cannot be exclusively
ruled out at the moment. Presumably, the alcohols reacted with
these tropylium ester intermediates and as well as the newly
formed anhydrides to afford the ester products. For aliphatic
acids, the anhydrides were possibly reactive enough to directly
react with the alcohol substrate to form the esters. On the other
hand, the benzoic and cinnamic anhydrides were probably less
reactive and reacted sluggishly with the alcohols if DMAP was
not present. We propose a reaction mechanism to illustrate all
these possible pathways (Scheme 2).
1
2
I. Dhimitruka, J. SantaLucia Jr., Org. Lett., 2006, 8, 47.
K. Bahrami, M. M. Khodaei, H. Tafghan, M. S. Arabi, Tetrahedron
Lett., 2013, 54, 5064.
For reviews on esterification reactions, see: (a) E. Haslam,
Tetrahedron, 1980, 36, 2409; (b) R. C. Larock, Comprehensive
Organic Transformations, 2nd ed.; John Wiley & Sons: New York,
1999; p1932. (c) R. Shelkov, M. Nahmany, A. Melman, Org. Biomol.
3
Chem., 2004,
2, 397; (d) J. Otera, J. Nishikido, Esterification,
Methods, Reactions, and Applications, 2nd ed.; Wiley-VCH:
Weinheim, 2010.
4
5
For reviews on amidation reactions, see: (a) S. Han, Y. Kim,
Tetrahedron, 2004, 60, 2447; (b) C. A. G. N. Montalbetti, V. Falque,
Tetrahedron, 2005, 61, 10827. (c) A. El-Faham, F. Albericio, Chem.
Rev., 2011, 111, 6557; (d) E. Valeur, M. Bradley, Chem. Soc. Rev.,
2009, 38, 606.
For recent developments of new coupling reagents, see: C. Salomé,
H. Kohn, Tetrahedron, 2009, 65, 456; J. K. Twibanire, T. B.
Grindley, Org. Lett., 2011, 13, 2988; J. Tian, W-C. Gao, D-M. Zhou,
C. Zhang, Org. Lett., 2012, 14, 3020; H. Chen, X. Xu, L. Liu, G.
The catalytic amidation coupling reactions, employing
10 mol% tropone and the slow addition of oxalyl chloride, also
proceeded smoothly, affording the products in good to high
yields. The addition of a catalytic amount of DMAP was still
Tang, Y. Zhao, RSC Adv., 2013, 3, 16247; D. Dev, N. B. Palakurthy,
K. Thalluri, J. Chandra, B. Mandal, J. Org. Chem., 2014, 79, 5420.
H. Chen, X. Xu, L. Liu, G. Tang, Y. Zhao, RSC Advances, 2013,
essential for improved yields of benzamides (entries 15 and 16
Table 3). Bulkier amines (entries 16 18 19, Table 3) seemed to
serve as better substrates for the coupling reactions than less
hindered amines (entries 14 12
15 17, Table 3). Traces of
,
6
,
,
3
, 16247.
7
8
T. V. Nguyen, A. Bekensir, Org. Lett., 2014, 16, 1720.
For recent developments in the use of another aromatic cation,
the cyclopropenium ion, to promote chemical transformation, see:
(a) B. D. Kelly, T. H. Lambert, J. Am. Chem. Soc., 2009, 131, 13930;
(b) D. J. Hardee, L. Kovalchuke, T. H. Lambert, J. Am. Chem.
Soc., 2010, 132, 5002; (c) V. P. Srivastava, R. Patel, Garima, L. D. S.
Yadav, Chem. Commun., 2010, 46, 5808; (d) C. M. Vanos, T. H.
,
,
cycloheptatrienimine by-products in these reaction mixtures
suggested that the amines, being relatively more nucleophilic
than the alcohols, competed with the carboxylate anions to react
with tropylium chloride (1), leading to unwanted consumption
Lambert, Chem. Sci., 2010, 1, 705; (e) B. D. Kelly, T. H. Lambert
of the reagents. Bulkier amines are less nucleophilic, hence less
likely to proceed through this side-reaction.
Org. Lett., 2011, 13, 740; (f) C. M. Vanos, T. H. Lambert, Angew.
Chem. Int. Ed., 2011, 50, 12222; (g) J. M. Nogueira, S. H. Nguyen,
C. S. Bennet, Org. Lett., 2011, 13, 2814; (h) J. M. Nogueira, J. P.
Issa, A. H. A. Chu, J. A. Sisel, R. S. Schum, C. S. Bennet, Eur. J.
Org. Chem. 2012, 4927; (i) J. S. Bandar, T. H. Lambert, J. Am.
Chem. Soc., 2012, 134, 5552; (j) E. D. Nacsa, T. H. Lambert, Org.
Lett., 2013, 15, 38; (k) J. S. Bandar, T. H. Lambert, Synthesis, 2013,
45, 2485; (l) A. Rai, L. D. S. Yadav, Eur. J. Org. Chem., 2013,
Conclusions
In summary, we have developed a new coupling reagent system
based on a very simple chlorinated hydrocarbon compound,
namely 1,1-dichlorocycloheptatriene. Due to its unique
property to equilibrate between its cycloheptratriene neutral
form and the aromatic tropylium ion form, this reagent can
provide the driving force for the esterification and amidation
reactions of carboxylic acids. Using this newly developed
reagent, several series of esters, lactones, amides and peptides
1889; (m) J. S. Bandar, T. H. Lambert, J. Am. Chem. Soc., 2013, 135
,
11799; (n) N. P. Panguluri, M. Samarasimhareddy, C. Madhu, V. V.
Sureshbabu, Synlett, 2014, 25, 1001.
For recent developments in the use of troyplium ions to promote
chemical transformation, see ref. 6 and: (a) J. M. Allen, T. H.
Lambert, J. Am. Chem. Soc., 2011, 133, 1260; (b) T. V. Nguyen, M.
Hall, Tetrahedron Lett., 2014, 55, 6895.
9
have been produced in good to excellent yields under mild 10 In solution, 1,1-dichlorocycloheptatriene equilibrates with its
chlorotropylium chloride form, see: M. Feigel, H. Kessler,
Tetrahedron, 1976, 32, 1575.
aromatic cation mediated chemistry and offers a new alternative
11 See the Supporting Information for more details.
reaction conditions. This work has expanded the scope of
method for carboxylic acid coupling reaction. The application
of this new method in combination with racemization
12 Ammonia gas from liquid cylinder was bubbled into the reaction
mixture as source of nucleophile.
4 | Chem. Commun., 2014, 50, 1-4
This journal is © The Royal Society of Chemistry 2014

 Nguyen, Thanh V.
Nguyen, Thanh V.
 Lyons, Demelza J.M.
Lyons, Demelza J.M.