
Medicinal Chemistry Research p. 4166 - 4180 (2015)
Update date:2022-08-16
Topics:
 Ahsan, Mohamed Jawed
Ahsan, Mohamed Jawed
 Choudhary, Kavita
Choudhary, Kavita
 Jadav, Surender Singh
Jadav, Surender Singh
 Yasmin, Sabina
Yasmin, Sabina
 Ansari, Md. Yousuf
Ansari, Md. Yousuf
 Sreenivasulu, Reddymasu
Sreenivasulu, Reddymasu
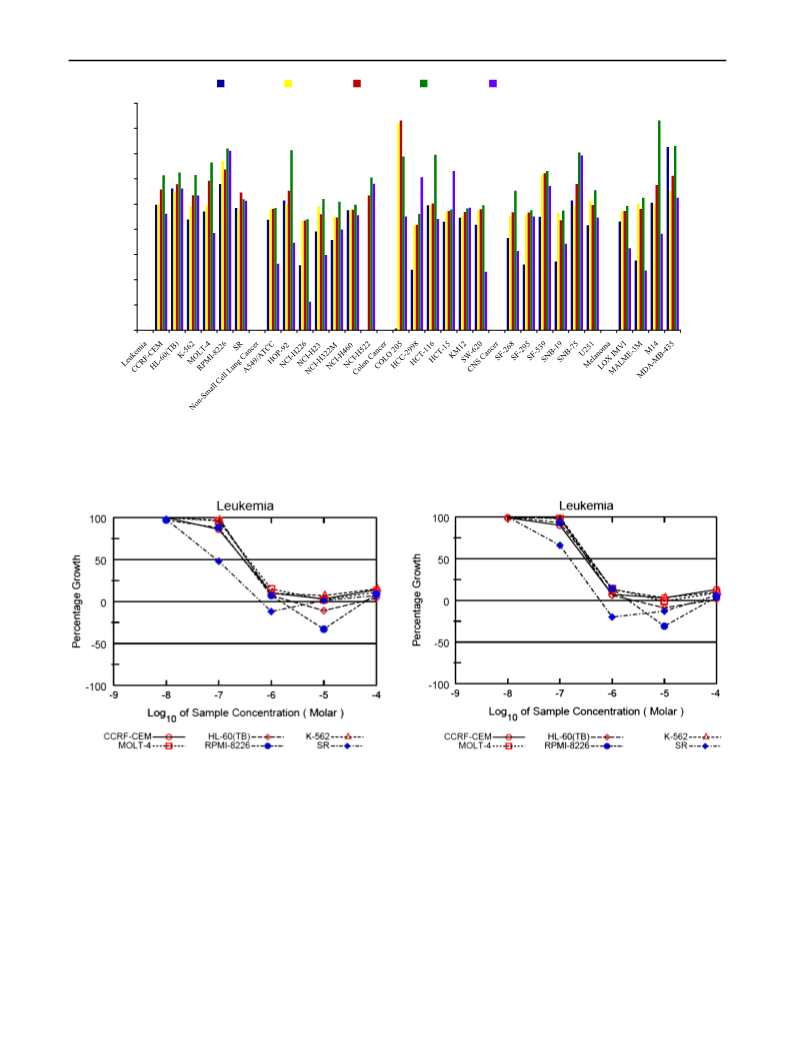
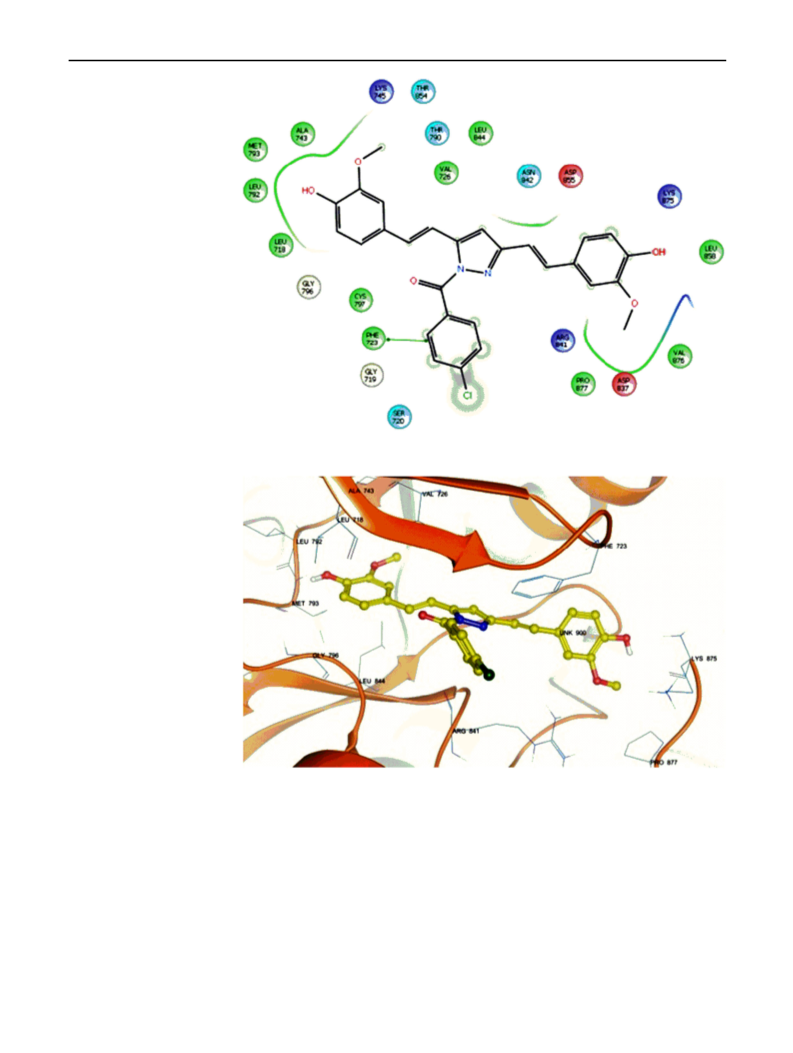
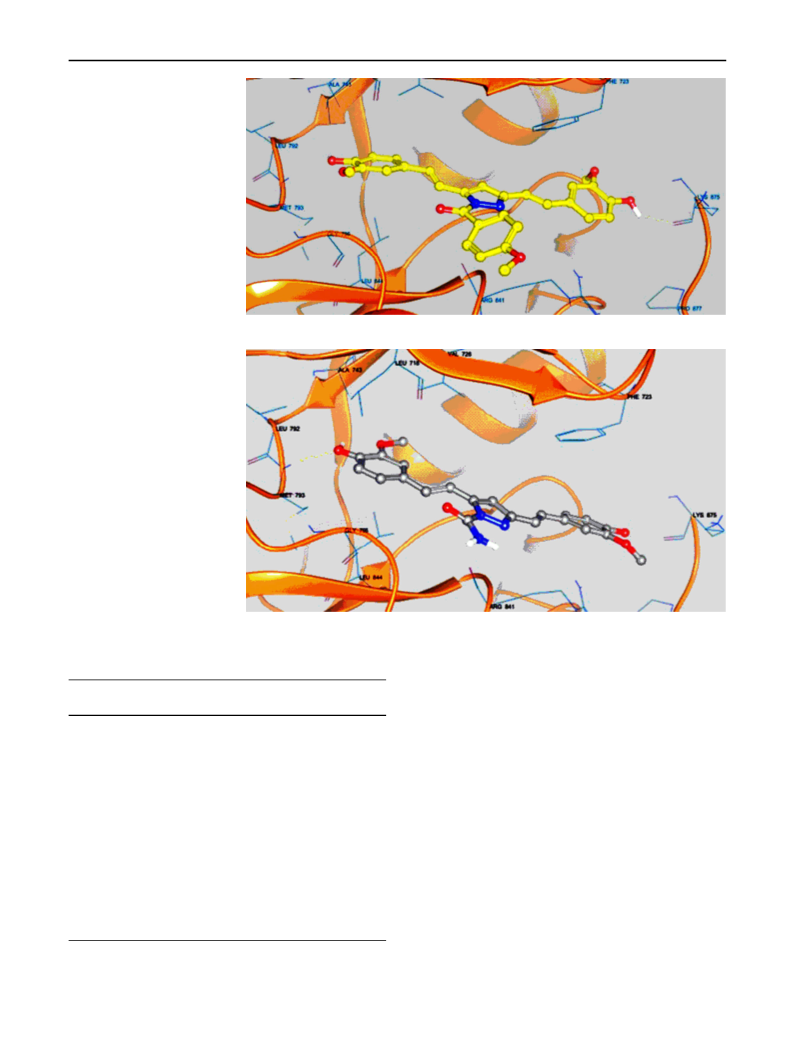
Several curcumin analogues bearing pyrazole were synthesized and characterized by IR, NMR, and mass spectral data. There were four tested compounds among 11 synthesized compounds, which were evaluated for antiproliferative activity and showed significant activity in both one-dose and five-dose assays. The antiproliferative effects were tested on a panel of 60 cell lines, according to the National Cancer Institute screening protocol. The most active compounds among the series were 3,5-bis(4-hydroxy-3-methylstyryl)-1H-pyrazole-1-carboxamide (3k) which showed mean percent growth inhibition of 116.09 in one-dose assay at 10 μM, and GI50 values were ranging between 0.0912 and 2.36 μM in five-dose assay. The best results were recorded on the leukaemia cell lines with value ranging from 0.0912 to 0.365 μM. All the tested compounds showed broad-spectrum antiproliferative activity over different cancer cell lines. When compared with the standard drug paclitaxel, the compound 3k showed superior activity on nearly 42 cell lines. The molecular docking study was performed to explore the binding interaction of these curcumin analogues with the active site of EGFR tyrosine kinase (EGFR-TK). The hydroxyl group of both phenyl rings was important for the rein-geminated hydrogen bonding by either side chain or backbone with the active site of EGFR-TK. Graphical Abstract: Four curcumin analogues were evaluated for their antiproliferative activity and showed promising results. The molecular docking studies showed that all the compounds (3a-k) were well accommodated in the EGFR tyrosine kinase.[Figure not available: see fulltext.]
View More











Contact:+1-416-493-6870
Address:Toronto, Canada
Zhejiang Tianyu Pharmaceutical Co., Ltd.
Contact:+86-576-84177669, 89189665,89189688,84168770
Address:Jiangkou Development Zone, Huangyan, Taizhou City, Zhejiang
Jiaxing Trustworthy Import And Export Co.,Ltd
Contact:+86-573-82030555
Address:Room 1202, Unit B, Charming plaza,No.1558 East Zhongshan Road , Jiaxing City, Zhejiang Province, China.
Contact:86+21-56421993
Address:3F,BUILDING 10,NO.2889 JINKE ROAD, SHANGHAI.
Shanghai birch chemical technology co.,ltd
Contact:+86-21-54096810
Address:No.2588,Jungong Road,Shanghai,China
Doi:10.1016/j.jcat.2018.05.003
(2018)Doi:10.1016/j.ica.2013.03.043
(2013)Doi:10.1039/c0gc00058b
(2010)Doi:10.1359/jbmr.2002.17.3.434
(1931)Doi:10.1021/jo00113a016
(1995)Doi:10.1023/A:1024926507658
(2003)