Organic & Biomolecular Chemistry
Communication
V. Bizet and N. Blanchard, Eur. J. Org. Chem., 2017, 6816;
( j) F. Pan, C. Shu and L.-W. Ye, Org. Biomol. Chem., 2016,
14, 9456; (k) A. M. Cook and C. Wolf, Tetrahedron Lett.,
2015, 56, 2377; (l) G. Evano, C. Theunissen and
M. Lecomte, Aldrichimica Acta, 2015, 48, 59;
(m) X.-N. Wang, H.-S. Yeom, L.-C. Fang, S. He, Z.-X. Ma,
B. L. Kedrowski and R. P. Hsung, Acc. Chem. Res., 2014, 47,
560; (n) N. Weding and M. Hapke, Chem. Soc. Rev., 2011,
40, 4525; (o) G. Domínguez and J. Pérez-Castells, Chem.
Soc. Rev., 2011, 40, 3430; (p) C. Madelaine, V. Valerio and
R.-S. Liu, Org. Lett., 2012, 14, 5522; (f) J. Oppenheimer,
W. L. Johnson, M. R. Tracey, R. P. Hsung, P.-Y. Yao, R. Liu
and K. Zhao, Org. Lett., 2007, 9, 2361.
4 (a) H. Huang, L. Tang, Y. Xi, G. He and H. Zhu, Tetrahedron
Lett., 2016, 57, 1873; (b) S. Xu, J. Liu, D. Hu and X. Bi,
Green Chem., 2015, 17, 184; (c) N. Ghosh, S. Nayak and
A. K. Sahoo, Chem.
–
Eur. J., 2013, 19, 9428;
(d) G. Compain, K. Jouvin, A. Martin-Mingot, G. Evano,
J. Marrot and S. Thibaudeau, Chem. Commun., 2012, 48,
5196; (e) X. Jin, K. Yamaguchi and N. Mizuno, Chem. Lett.,
2012, 41, 866; (f) S. Kramer, J. L. H. Madsen, M. Rottländer
and T. Skrydstrup, Org. Lett., 2010, 12, 2758.
5 (a) A. M. Cook and C. Wolf, Angew. Chem., Int. Ed., 2016,
55, 2929; (b) L. Hu, S. Xu, Z. Zhao, Y. Yang, Z. Peng,
M. Yang, C. Wang and J. Zhao, J. Am. Chem. Soc., 2016, 138,
13135; (c) D. L. Smith, W. R. F. Goundry and H. W. Lam,
Chem. Commun., 2012, 48, 1505.
6 (a) J. A. Mulder, R. P. Hsung, M. O. Frederick, M. R. Tracey
and C. A. Zificsak, Org. Lett., 2002, 4, 1383;
(b) N. P. Grimster, D. A. A. Wilton, L. K. M. Chan,
C. R. A. Godfrey, C. Green, D. R. Owen and M. J. Gaunt,
Tetrahedron, 2010, 66, 6429; (c) M. O. Frederick,
R. P. Hsung, R. H. Lambeth, J. A. Mulder and M. R. Tracey,
Org. Lett., 2003, 5, 2663; (d) K. C. M. Kurtz, M. O. Frederick,
R. H. Lambeth, J. A. Mulder, M. R. Tracey and R. P. Hsung,
Tetrahedron, 2006, 62, 3928.
N. Maulide, Chem.
–
Asian J., 2011, 6, 2224;
(q) K. A. DeKorver, H. Li, A. G. Lohse, R. Hayashi, Z. Lu,
Y. Zhang and R. P. Hsung, Chem. Rev., 2010, 110, 5064;
(r) G. Evano, A. Coste and K. Jouvin, Angew. Chem., Int. Ed.,
2010, 49, 2840; (s) L. Ackermann and H. K. Potukuchi, Org.
Biomol. Chem., 2010, 8, 4503.
2 selected references on the oxidation of ynamides, see:
(a) T. Baker and P. W. Davies, Eur. J. Org. Chem., 2019,
5201–5204; (b) D. Zhang, J. Man, Y. Chen, L. Yin, J. Zhong
and Q.-F. Zhang, RSC Adv., 2019, 9, 12567; (c) F. Sánchez-
Cantalejo, J. D. Priest and P. W. Davies, Chem. – Eur. J.,
2018, 24, 17215; (d) X.-L. Han, X.-G. Liu, E. Lin, Y. Chen,
Z. Chen, H. Wang and Q. Li, Chem. Commun., 2018, 54,
11562; (e) C.-M. Wang, L.-J. Qi, Q. Sun, B. Zhou,
Z.-X. Zhang, Z.-F. Shi, S.-C. Lin, X. Lu, L. Gong and
L.-W. Ye, Green Chem., 2018, 20, 3271; (f) Z.-X. Zhang,
B.-H. Zhu, P.-X. Xie, J.-Q. Tang, X.-L. Li, C. Zhu, Y.-W. Yin
and L.-W. Ye, RSC Adv., 2018, 8, 18308; (g) X. Liu,
Z.-X. Zhang, B. Zhou, Z.-S. Wang, R.-H. Zheng and L.-W. Ye,
Org. Biomol. Chem., 2017, 15, 10156; (h) A. Ragupathi,
V. P. Charpe, A. Sagadevan and K. C. Hwang, Adv. Synth.
Catal., 2017, 359, 1138; (i) F. Pan, X.-L. Li, X.-M. Chen,
7 B. Peng, X. Huang, L.-G. Xie and N. Maulide, Angew. Chem.,
Int. Ed., 2014, 53, 8718.
8 A. Siva Reddy, A. L. Siva Kumari, S. Saha and K. C. Kumara
Swamy, Adv. Synth. Catal., 2016, 358, 1625.
9 A. Siva Reddy and K. C. Kumara Swamy, Angew. Chem., Int.
Ed., 2017, 56, 6984.
C. Shu, P.-P. Ruan, C.-H. Shen, X. Lu and L.-W. Ye, ACS 10 (a) L. V. Graux, H. Clavier and G. Buono, ChemCatChem,
Catal., 2016, 6, 6055; ( j) H. Huang, L. Tang, Q. Liu, Y. Xi,
G. He and H. Zhu, Chem. Commun., 2016, 52, 5605;
2014, 6, 2544; (b) R. Plamont, L. V. Graux and H. Clavier,
Eur. J. Org. Chem., 2018, 1372.
(k) L. Li, C. Shu, B. Zhou, Y.-F. Yu, X.-Y. Xiao and L.-W. Ye, 11 the metal-free intramolecular hydroalkoxylation followed
Chem. Sci., 2014, 5, 4057; (l) F. Pan, S. Liu, C. Shu,
R.-K. Lin, Y.-F. Yu, J.-M. Zhou and L.-W. Ye, Chem.
Commun., 2014, 50, 10726; (m) C.-H. Shen, L. Li, W. Zhang,
by a [1,3]-rearrangement, see: B. Zhou, Y.-Q. Zhang,
K. Zhang, M.-Y. Yang, Y.-B. Chen, Y. Li, Q. Peng, S.-F. Zhu,
Q.-L. Zhou and L.-W. Ye, Nat. Commun., 2019, 10, 3234.
S. Liu, C. Shu, Y.-E. Xie, Y.-F. Yu and L.-W. Ye, J. Org. Chem., 12 J. A. Mulder, K. C. M. Kurtz, R. P. Hsung, H. Coverdale,
2014, 79, 9313; (n) L.-Q. Yang, K.-B. Wang and C.-Y. Li,
Eur. J. Org. Chem., 2013, 2775; (o) Y. Minko, M. Pasco,
M. O. Frederick, L. Shen and C. A. Zificsak, Org. Lett., 2003,
5, 1547.
L. Lercher, M. Botoshansky and I. Marek, Nature, 2012, 490, 13 For the reaction of terminally unsubstituted ynamide 1n
522; (p) R. B. Dateer, K. Pati and R.-S. Liu, Chem. Commun.,
2012, 48, 7200; (q) P. W. Davies, A. Cremonesi and N. Martin,
Chem. Commun., 2011, 47, 379; (r) C.-F. Xu, M. Xu, Y.-X. Jia
and C.-Y. Li, Org. Lett., 2011, 13, 1556; (s) A. Mukherjee,
R. B. Dateer, R. Chaudhuri, S. Bhunia, S. N. Karad and
R.-S. Liu, J. Am. Chem. Soc., 2011, 133, 15372.
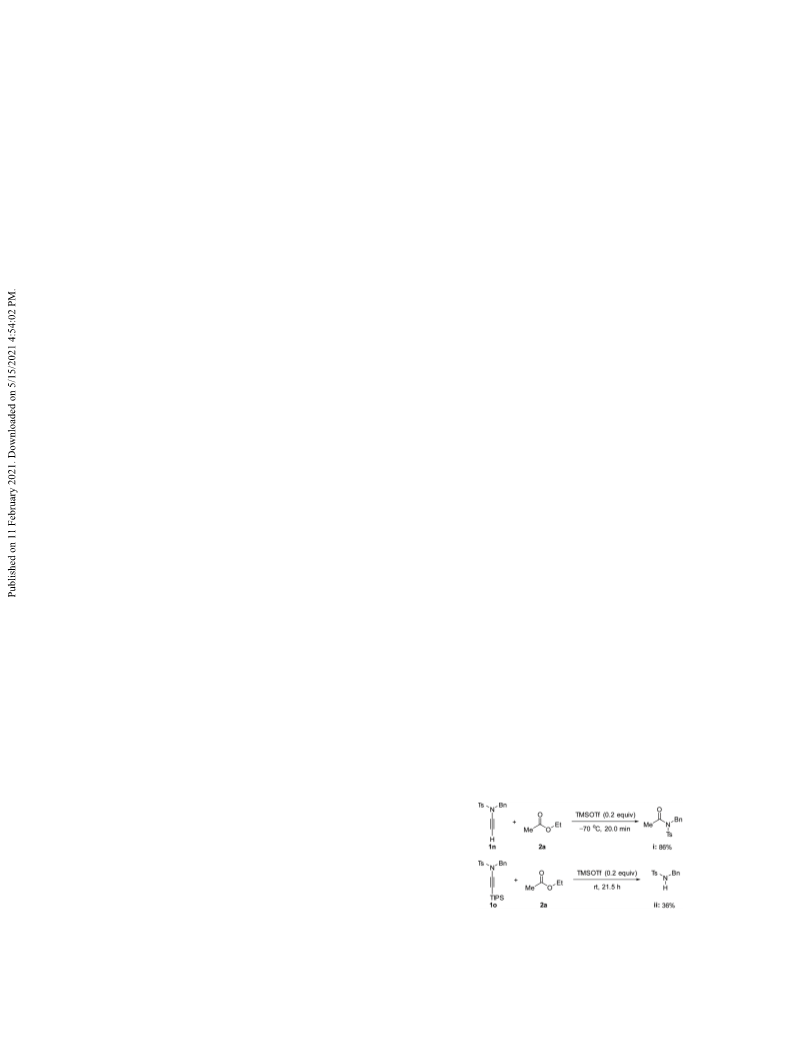
with ethyl acetate 2a, and TIPS-substituted ynamide 1o
with ethyl acetate 2a.
3 (a) L. You, Z. F. Al-Rashid, R. Figueroa, S. K. Ghosh, G. Li,
T. Lu and R. P. Hsung, Synlett, 2007, 1656;
(b) K. C. M. Kurtz, R. P. Hsung and Y. Zhang, Org. Lett.,
2006, 8, 231; (c) H. Liu, Y. Yang, J. Wu, X.-N. Wang and
J. Chang, Chem. Commun., 2016, 52, 6801; (d) L. Chen,
.
Y.-M. Cui, Z. Xu, J. Cao, Z.-J. Zheng and L.-W. Xu, Chem. 14 For the crystal data of 3a and 3y′, see the ESI.†
Commun., 2016, 52, 11131; (e) S. Bhunia, C.-J. Chang and 15 Y. Li, Z. Wang and X.-F. Wu, Green Chem., 2018, 20, 969.
This journal is © The Royal Society of Chemistry 2021
Org. Biomol. Chem., 2021, 19, 2182–2185 | 2185

 Gao, Erhui
Gao, Erhui
 Peng, Cheng
Peng, Cheng
 Zhang, Jingyi
Zhang, Jingyi
 Wang, Xiao-Na
Wang, Xiao-Na
 Chang, Junbiao
Chang, Junbiao