1
48
M. Ángeles Fuentes et al. / Inorganica Chimica Acta 369 (2011) 146–149
Table 1
exclusively to tertiary sites functionalized derivatives. In the case
of the Au–Ag compound 2, a remarkably high selectivity toward
the tertiary sites is observed. Thus, with 2,3-dimethylbutane as
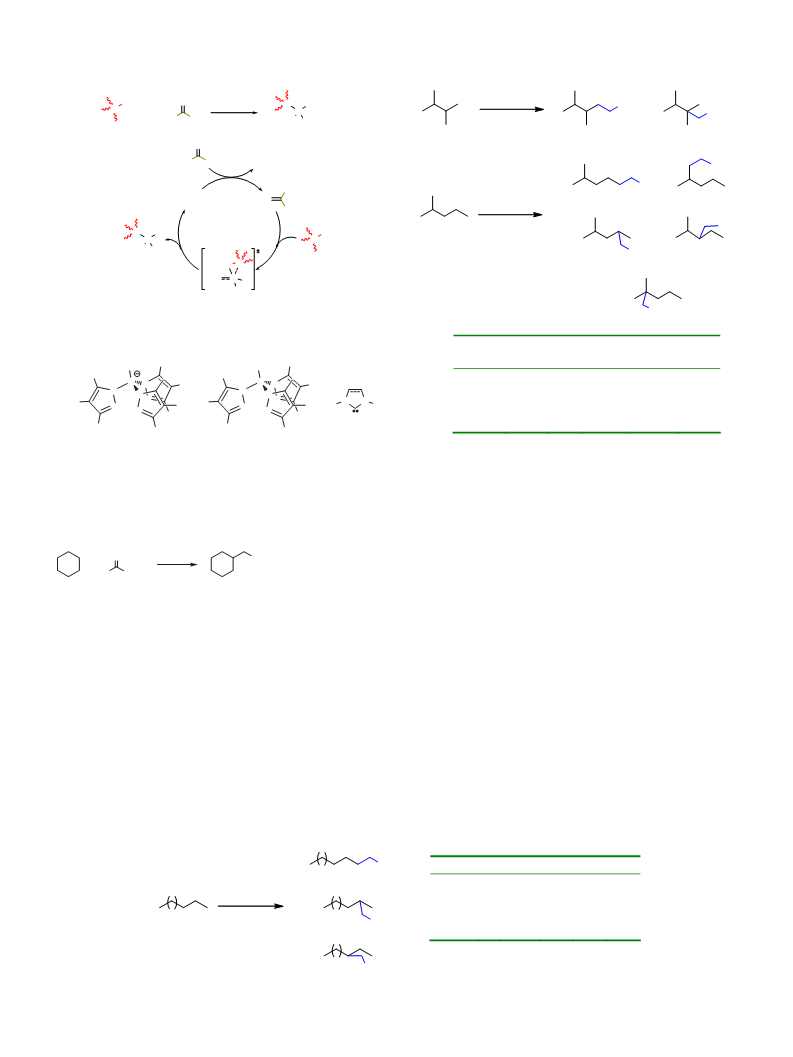
the substrate a 1:31 regioselectivity for primary:tertiary sites has
been found. Such ratio is similar to that with 2-methylpentane
Regioselectivitya observed for the functionalization of pentane with EDA in the
presence of several coinage metal catalysts.
Catalyst
Regios. 1ry
C1
Regios. 2ry
C2
Regios. 2ry
C3
Reference
1
2
–
1.00
–
1.00
1.00
1.50
8.70
1.77
2.94
3.17
1.00
6.8
1.00
1.44
2.19
This work
This work
[10b]
[10d]
[12a]
(1:33), but quite different from that previously reported with
Br3
[
2
Tp Ag] (1:3.97 and 1:3.70, respectively, for 2,3DMB and 2MP)
TpBr3Cu(NCMe)
TpBr3Ag]
[10d]. A comparison with the gold-based catalytic system [IPr-
[
2
0
0
AuCl] + NaBAr
would not explain those values: the latter catalyst
IPrAuCl + NaBAr
4
4
clearly favored the insertion into primary sites, the regioselectivity
for the tertiary sites being the lowest known to date (1:1.23,
primary:tertiary, for 2,3-dimethylbutane and 1:2.06, also primary:
tertiary, for 2-methylpentane) [12]. Therefore, data available indi-
cate that in compounds 1 and 2, the active catalytic center are
copper and silver, respectively, the gold center not participating
in the catalytic transformation. A second feature of this system is
that the silver catalyst induces an exceptionally high regioselection
toward tertiary sites, in contrast with previous work from our as
well as Rasika Dias’s groups [10d,13b].
In the search for additional evidences to support the role of gold
as an spectator in this catalytic system, we have carried out the
reaction of benzene and ethyl diazoacetate in the presence of 1
and 2 as the catalyst. The reaction of benzene and ethyl diazoace-
tate, the so-called Büchner reaction, usually provides a mixture of
cycloheptatrienes. But in the presence of an appropriate transition
metal catalyst (rhodium-, copper-, or silver-based), only one cyclo-
a
Values normalized for the relative number of C–H bonds of each type.
Table 2
Regioselectivitya observed for the functionalization of 2,3-dimethylbutane with EDA
in the presence of several coinage metal catalysts.
Catalyst
Regios. 1ry C–H
Regios. 3ry C–H
Reference
1
2
1.00
1.00
–
1.00
1.00
94.0
31.0
1.00
3.97
1.23
This work
This work
[10b]
[10d]
[12a]
TpBr3Cu(NCMe)
[
TpBr3Ag]
2
0
IPrAuCl + NaBAr
4
a
Values normalized for the relative number of C–H bonds of each type.
Table 3
Regioselectivitya observed for the functionalization of 2-methylpentane with EDA in
the presence of several coinage metal catalysts.
heptatriene is formed in a selective manner [17]. Recently, we dis-
0
covered IPrAuCl + NaBAr
4
for the catalytic transfer of carbenes
Catalyst
Regios. 1ry
C–H
Regios. 2ry
C–H
Regios. 3ry
C–H
Reference
from ethyl diazoacetate, using benzene as the substrate [18]. In
this reaction, and in addition to the expected cycloheptatriene,
the major product was ethyl 2-phenylacetate, the product derived
from the formal insertion of the carbene group into the benzene
C–H bond. When complexes 1 and 2 were employed in this reaction,
very low yields of the insertion product were observed, the major
product being, by far the cycloheptatriene (Eq. (2)) [19]. We believe
that, again, the catalytic results are in agreement with the role of
gold as a ‘‘ligand’’ for copper or silver, with no participation at all
in the catalytic reaction.
1
2
1.00
1.00
–
1.00
1.00
2.57
1.10
1.00
1.76
1.92
55.0
33.0
6.52
3.70
2.06
This work
This work
[10b]
[10d]
[12a]
TpBr3Cu(NCMe)
[
TpBr3Ag]
2
0
IPrAuCl + NaBAr
4
a
Values normalized for the relative number of C–H bonds of each type.
by the carbene insertion methodology, using EDA as the carbene
source. Those studies showed that the order of reactivity follows
the order tertiary > secondary > primary C–H bonds [15], that is,
the same trend that the C–H bond dissociation energy [16]. In or-
der to compare the regioselectivity induced by compounds 1 and
N
2
1
or 2
2
CO Et
+
+
2
CO Et
H
2
CO Et
-
N
2
ð2Þ
9
8%
2%
7%
2
, the distribution of products shown in Schemes 2 and 3 must
catalyst: 1
9
3%
2
be normalized with the number of hydrogens of each type (pri-
mary, secondary, tertiary). Tables 1–3 contains such values for
three representative examples, pentane, 2,3-dimethylbutane and
4
. Conclusion
2
-methylpentane. In addition of the values obtained with 1 and
2
, and for the sake of comparison, the regioselectivities previously
We have found that the complexes [Au
M = Cu, 1; M = Ag, 2) catalyze the functionalization of several al-
kanes, linear or branched, by means of the insertion of :CHCO Et,
2 2 6 5 4 2 n
M (C F ) (NCMe) ]
described with three other coinage metal catalysts are also
included.
(
2
In all cases, the aforementioned trend of reactivity matching the
bond dissociation energy has also been found with these interme-
tallic Au–M (M = Cu, Ag) compounds: tertiary sites are more prone
to functionalization as inferred for the highest value of the regiose-
lectivity with no a single exception. When pentane as the sub-
strate, the lack of tertiary sites made secondary positions as the
most favorable and, among them, those of C2. Since the BDEs of
both C2 and C3 C–H bonds do not differ much, this could be ex-
plained as the result of the steric effect, that plays a secondary
but somewhat relevant role in this reaction. The same behavior
and explanations apply to hexane as the substrate.
generated from ethyl diazoacetate, into the carbon–hydrogen
bonds. Data available seems to favor the proposal that the active
catalytic centers are copper or silver, with the gold Au(C F ) acting
6 5 2
as a ligand that affects the course of the catalytic transformation.
5. Experimental
5.1. General methods
All preparations and manipulations were carried out under an
oxygen-free nitrogen atmosphere using conventional Schlenk tech-
niques. The solvents employed for all preparations were purified
using a BRAUN-SPS system and were degassed before use. The
hydrocarbons and ethyl diazoacetate were purchased from Aldrich
and employed without further purification. The complexes 1 and 2
The copper-containing compound 1 seems to favor the insertion
of the carbene group into secondary and/or tertiary sites, whereas
the primary C–H bonds are less prone to undergo this transforma-
tion. This is in good agreement with the previous work carried out
Br3
with copper-based catalysts such as Tp Cu(NCMe) [10b], that led

 Fuentes, M. ángeles
Fuentes, M. ángeles
 Rodríguez-Castillo, María
Rodríguez-Castillo, María
 Monge, Miguel
Monge, Miguel
 Olmos, M. Elena
Olmos, M. Elena
 López-De-Luzuriaga, Jose M.
López-De-Luzuriaga, Jose M.
 Caballero, Ana
Caballero, Ana
 Pérez, Pedro J.
Pérez, Pedro J.