The Journal of Organic Chemistry
Article
Ethyl 4-(Isopropylamino)-6-(1H-pyrazolo[3,4-c]pyridin-1-yl)-
nicotinate (10b). The reaction was carried out on a 500 mg scale
using Procedure A. The product was purified by column
chromatography using EtOAc/n-heptane (2:1) as the eluent to afford
= 8.0, 0.8 Hz, 1H), 8.46 (d, J = 0.8 Hz, 1H), 8.46−8.37 (m, 2H),
7.95−7.92 (dd, J = 8.0, 2 Hz, 1H), 7.86−7.82 (m, 1H), 7.16−7.12 (m,
13
2H), 6.64−6.63 (d, J = 4.0 Hz, 1H). C NMR (100 MHz, DMSO-d ):
6
δ 150.8, 148.2, 147.5, 143.1, 138.2, 129.1, 126.4, 123.3, 120.3, 117.2,
115.7, 102.6. HRMS (ESI-Orbitrap), m/z: [M + H]+ calcd for
C H N , 196.0875; found, 196.0861.
3
02 mg (45%) of compound 10b as an off-white solid (mp 149−151
1
°
C). H NMR (400 MHz, DMSO-d ): δ 10.09 (s, 1H), 8.84 (s, 1H),
6
12
10
3
8
1
2
1
1
2
.60 (d, J = 0.8 Hz, 1H), 8.43 (d, J = 5.2 Hz, 1H), 8.09 (d, J = 7.6 Hz,
H), 7.89 (dd, J = 5.3, 1.3 Hz, 1H), 7.22 (s, 1H), 4.33 (q, J = 7.2 Hz,
H), 3.89 (dd, J = 13.3, 6.8 Hz, 1H), 1.35 (t, J = 7.0 Hz, 3H), 1.32−
1
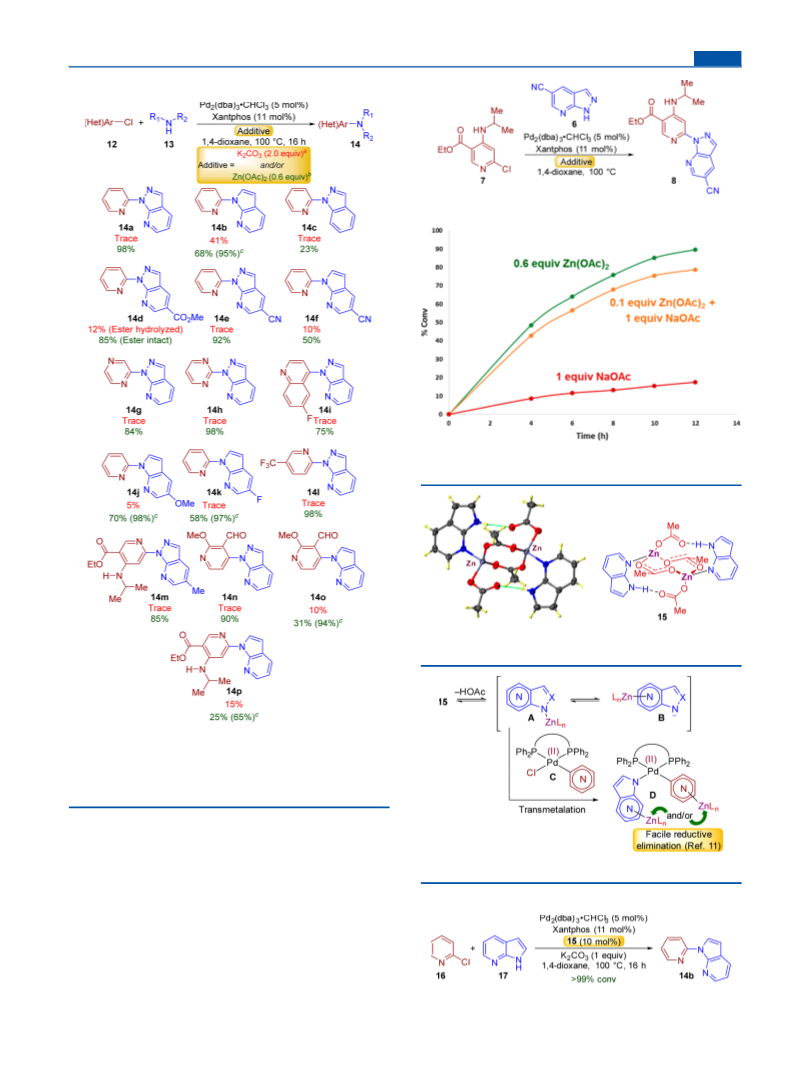
-(Pyridin-2-yl)-1H-indazole (14c). The reaction was carried out on
a 1.00 g scale using Procedure A. The product was purified by column
chromatography using EtOAc/n-heptane (1:5) as the eluent to afford
.23 (m, 6H). 13C NMR (100 MHz, DMSO-d ): δ 167.4, 156.1, 155.1,
1
6
395 mg (23%) of compound 14c as a white solid (mp 81−83 °C). H
52.9, 141.3, 139.3, 137.9, 135.7, 130.3, 115.7, 105.6, 92.7, 61.1, 43.7,
NMR (400 MHz, DMSO-d ): δ 8.78−8.76 (d, J = 8.8 Hz, 1H), 8.59−
6
2.5, 14.6. HRMS (ESI-Orbitrap), m/z: [M + H]+ calcd for
8
.58 (d, J = 4.8 Hz, 1H), 8.46 (s, 1H), 8.04−8.01 (m, 2H), 7.92−7.90
C H N O , 326.1617; found, 326.1596.
13
1
7
20
5
2
(d, J = 8.0 Hz, 1H), 7.59−7.55 (m, 1H), 7.35−7.31 (m, 2H).
C
Ethyl 4-(Isopropylamino)-6-(1H-pyrazolo[4,3-c]pyridin-1-yl)-
NMR (100 MHz, DMSO-d ): δ 153.6, 147.9, 139.1, 138.1, 137.3,
6
nicotinate (10c). The reaction was carried out on a 250 mg scale
using Procedure A. The product was purified by column
chromatography using EtOAc/n-heptane (2:1) as the eluent to afford
1
28.1, 125.7, 122.6, 121.2, 120.4, 114.8, 112.9. HRMS (ESI-Orbitrap),
m/z: [M + H] calcd for C H N , 196.0875; found, 196.0859.
+
12
10
3
Methyl 1-(Pyridin-2-yl)-1H-pyrazolo[3,4-b]pyridine-5-carboxylate
2
88 mg (86%) of compound 10c as an off-white solid (mp 128−130
(
14d). The reaction was carried out on a 500 mg scale using Procedure
1
°
C). H NMR (400 MHz, DMSO-d ): δ 9.22 (d, J = 0.8 Hz, 1H), 8.80
17
6
A. The product was purified by column chromatography using
(
s, 1H), 8.68 (s, 1H), 8.59−8.54 (m, 2H), 8.09 (d, J = 7.6 Hz, 1H),
EtOAc/n-heptane (1:2) as the eluent to afford 952 mg (85%) of
7
1
.23 (s, 1H), 4.35−4.30 (q, J = 7.2 Hz, 2H), 3.91−3.86 (m, 1H),
.37−1.33 (t, J = 7.2 Hz, 3H), 1.29−1.28 (d, J = 6.4 Hz, 6H).
1
compound 14d as a pale brownish solid (mp 134−136 °C). H NMR
13
C
(
400 MHz, CDCl ): δ 9.34 (d, J = 2.0 Hz, 1H), 8.83−8.82 (d, J = 2.4
3
NMR (100 MHz, DMSO-d ): δ 166.8, 155.9, 154.6, 152.2, 145.9,
6
Hz, 1H), 8.72 (m, 1H), 8.40 (m, 2H), 7.96−7.92 (m, 1H), 7.34−7.30
1
45.5, 141.4, 137.8, 122.8, 109.9, 105.0, 92.7, 60.6, 43.1, 21.9, 14.0.
13
(
m, 1H), 4.01 (s, 3H). C NMR (100 MHz, CDCl ): δ 165.7, 151.5,
+
3
HRMS (ESI-Orbitrap), m/z: [M + H] calcd for C H N O ,
17
20
5
2
1
5
2
51.1, 149.0, 139.2, 138.5, 136.3, 132.7, 122.0, 120.9, 117.1, 116.2,
2.5. HRMS (ESI-Orbitrap), m/z: [M + H] calcd for C H N O ,
3
26.1617; found, 326.1590.
Ethyl 4-(Isopropylamino)-6-(1H-pyrazolo[4,3-b]pyridin-1-yl)-
+
1
3
11
4
2
55.0882; found, 255.0863.
-(Pyridin-2-yl)-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile (14e).
nicotinate (10d). The reaction was carried out on a 250 mg scale
using Procedure A. The product was purified by column
chromatography using EtOAc/n-heptane (2:1) as the eluent to afford
1
The reaction was carried out on a 1.00 g scale using Procedure A.
The product was purified by column chromatography using EtOAc/n-
heptane (2:1) as the eluent to afford 1.79 g (92%) of compound 14e
2
04 mg (61%) of compound 10d as a pale pinkish solid (mp 140−142
1
°C). H NMR (400 MHz, DMSO-d ): δ 9.05−9.03 (d, J = 8.4 Hz,
6
1
as an off-white solid (mp 193−195 °C). H NMR (400 MHz, DMSO-
1
7
6
1
1
9
H), 8.77 (s, 1H), 8.68−8.67 (m, 2H), 8.08−8.06 (d, J = 7.2 Hz, 1H),
.58−7.55 (dd, J = 8.4, 4.4 Hz, 1H), 7.22 (s, 1H), 4.35−4.29 (q, J =
.8 Hz, 2H), 3.91−3.86 (m, 1H), 1.37−1.33 (t, J = 7.2 Hz, 3H), 1.29−
d ): δ 9.06−9.03 (m, 2H), 8.67−8.65 (m, 2H), 8.12−8.10 (m, 2H),
6
7
13
.53−7.50 (m, 1H). C NMR (100 MHz, DMSO-d ): δ 151.7, 150.1,
6
.28 (d, J = 6.4 Hz, 6H). 13C NMR (100 MHz, DMSO-d ): δ 166.8,
149.7, 148.1, 139.0, 136.9, 136.6, 123.1, 117.6, 117.3, 116.1, 103.0.
6
+
HRMS (ESI-Orbitrap), m/z: [M + H] calcd for C H N , 222.0780;
12
8
5
56.0, 154.5, 152.2, 147.1, 143.2, 138.0, 131.9, 123.4, 122.5, 104.9+,
found, 222.0771.
-(Pyridin-2-yl)-1H-pyrrolo[2,3-b]pyridine-5-carbonitrile (14f).
2.0, 60.5, 43.1, 21.9, 14.0. HRMS (ESI-Orbitrap), m/z: [M + H]
1
calcd for C H N O , 326.1617; found, 326.1603.
17
20
5
2
The reaction was carried out on a 500 mg scale using Procedure A.
The product was purified by column chromatography using EtOAc/n-
heptane (1:4) as the eluent to afford 485 mg (50%) of compound 14f
Ethyl 6-(1H-Indazol-1-yl)-4-(isopropylamino)nicotinate (10e).
The reaction was carried out on a 500 mg scale using Procedure A.
The product was purified by column chromatography using EtOAc/n-
heptane (1:3) as the eluent to afford 268 mg (40%) of compound 10e
1
as an off-white solid (mp 191−193 °C). H NMR (400 MHz, CDCl ):
3
1
δ 8.81−8.79 (d, J = 8.4 Hz, 1H), 8.65 (s, 1H), 8.64−8.51 (m, 2H),
as an off-white solid (mp 91−93 °C). H NMR (400 MHz, DMSO-
8
1
1
.25−8.24 (d, J = 2.0 Hz, 1H), 7.93−7.89 (m, 1H), 7.26−7.23 (m,
d ): δ 8.79−8.76 (m, 2H), 8.47 (s, 1H), 8.03 (d, J = 7.2 Hz, 1H),
6
13
H), 6.74−6.73 (d, J = 4.0 Hz, 1H). C NMR (100 MHz, CDCl ): δ
7
1
1
.90−7.88 (d, J = 8.0 Hz, 1H), 7.57−7.53 (m, 1H), 7.34−7.30 (m,
H), 7.22 (s, 1H), 4.36−4.23 (q, J = 6.8 Hz, 2H), 3.89−3.84 (m, 1H),
3
48.5, 145.8, 141.2, 138.5, 132.7, 129.3, 122.5, 121.5, 118.4, 112.4,
+
13
116.2, 102.9, 102.5. HRMS (ESI-Orbitrap), m/z: [M + H] calcd for
C H N , 221.0827; found, 221.0816.
13
1
was carried out on a 1.00 g scale using Procedure A. The product was
purified by column chromatography using EtOAc/n-heptane (2:1) as
the eluent to afford 1.45 g (84%) of compound 14g as a pale brownish
.37−1.30 (t, J = 7.2 Hz, 3H), 1.30−1.22 (d, J = 6.4 Hz, 6H).
C
NMR (100 MHz, DMSO-d ): δ 166.9, 156.5, 154.4, 152.2, 138.5,
9
4
6
-(Pyrazin-2-yl)-1H-pyrazolo[3,4-b]pyridine (14g). The reaction
1
1
3
37.9, 128.1, 125.8, 122.7, 121.2, 115.5, 104.6, 92.2, 60.4, 43.0, 21.9,
+
4.0. HRMS (ESI-Orbitrap), m/z: [M + H] calcd for C H N O ,
18
21
4
2
25.1665; found, 325.1638.
-(Pyridin-2-yl)-1H-pyrazolo[3,4-b]pyridine (14a). The reaction
1
1
solid (mp 94−96 °C). H NMR (400 MHz, DMSO-d ): δ 9.58 (m,
was carried out on a 1.00 g scale using Procedure A. The product was
purified by column chromatography using EtOAc/n-heptane (3:1) as
the eluent to afford 1.69 g (98.0%) of compound 14a as a brownish
6
1
(
H), 8.75−8.73 (m, 2H), 8.70−8.69 (m, 1H), 8.61 (s, 1H), 8.46−8.43
13
dd, J = 8.0, 1.6 Hz, 1H), 7.48−7.45 (dd, J = 8.0, 4.8 Hz, 1H). C
1
NMR (100 MHz, DMSO-d ): δ 150.2, 149.9, 147.3, 143.1, 142.2,
oil. H NMR (400 MHz, DMSO-d ): δ 8.69−8.64 (dd, J = 4.4, 1.6 Hz,
6
6
1
8
H), 8.64 (s, 1H), 8.49 (s, 1H), 8.39 (dd, J = 8.0, 1.6 Hz, 1H), 8.23−
138.0, 137.2 131.4, 118.9, 117.2. HRMS (ESI-Orbitrap), m/z: [M +
13
+
.21 (m, 1H), 8.09−8.05 (m, 1H), 7.46−7.39 (m, 2H). C NMR
H] calcd for C10
H
8
N
5
, 198.0780; found, 198.0771.
(
100 MHz, DMSO-d ): δ 150.7, 149.9, 149.6, 148.7, 138.7, 135.6,
1-(Pyrimidin-2-yl)-1H-pyrazolo[3,4-b]pyridine (14h). The reaction
was carried out on a 1.00 g scale using Procedure A. The product was
purified by column chromatography using EtOAc/n-heptane (2:1) as
6
1
31.1, 122.2, 118.5, 117.0, 116.7. HRMS (ESI-Orbitrap), m/z: [M +
+
H] calcd for C H N , 197.0827; found, 197.0816.
11
9
4
1
-(Pyridin-2-yl)-1H-pyrrolo[2,3-b]pyridine (14b). The reaction was
the eluent to afford 1.69 g (98%) of compound 14h as a brownish
1
carried out on a 1.00 g scale using Procedure A. The product was
purified by column chromatography using EtOAc/n-heptane (1:5) as
the eluent to afford 1.17 g (68%) of compound 14b as a white solid.
The reaction was carried out on a 1.00 g scale using Procedure B.
The product was purified by column chromatography using EtOAc/n-
solid (mp 90−92 °C). H NMR (400 MHz, DMSO-d
6
): δ 9.02 (d, J =
4.8 Hz, 2H), 8.71−8.70 (dd, J = 4.8, 1.6 Hz, 1H), 8.53 (s, 1H), 8.41−
8.39 (dd, J = 8.0, 1.6 Hz, 1H), 7.60−7.58 (t, J = 4.8 Hz, 1H), 7.44−
7.41 (dd, J = 8.0, 4.4 Hz, 1H). 13C NMR (100 MHz, DMSO-d
): δ
6
159.3, 156.3, 150.5, 149.9, 136.6, 130.9, 119.8, 118.9, 117.1. HRMS
(ESI-Orbitrap), m/z: [M + H] calcd for C H N , 198.0780; found,
198.0766.
+
heptane (1:5) as the eluent to afford 1.63 g (95%) of compound 14b
10
8
5
1
(
mp 64−66 °C). H NMR (400 MHz, DMSO-d ): δ 8.48−8.47 (dd, J
6
F
J. Org. Chem. XXXX, XXX, XXX−XXX

 Ayothiraman, Rajaram
Ayothiraman, Rajaram
 Rangaswamy, Sundaramurthy
Rangaswamy, Sundaramurthy
 Maity, Prantik
Maity, Prantik
 Simmons, Eric M.
Simmons, Eric M.
 Beutner, Gregory L.
Beutner, Gregory L.
 Janey, Jacob
Janey, Jacob
 Treitler, Daniel S.
Treitler, Daniel S.
 Eastgate, Martin D.
Eastgate, Martin D.
 Vaidyanathan, Rajappa
Vaidyanathan, Rajappa