Journal of the American Chemical Society
Page 4 of 5
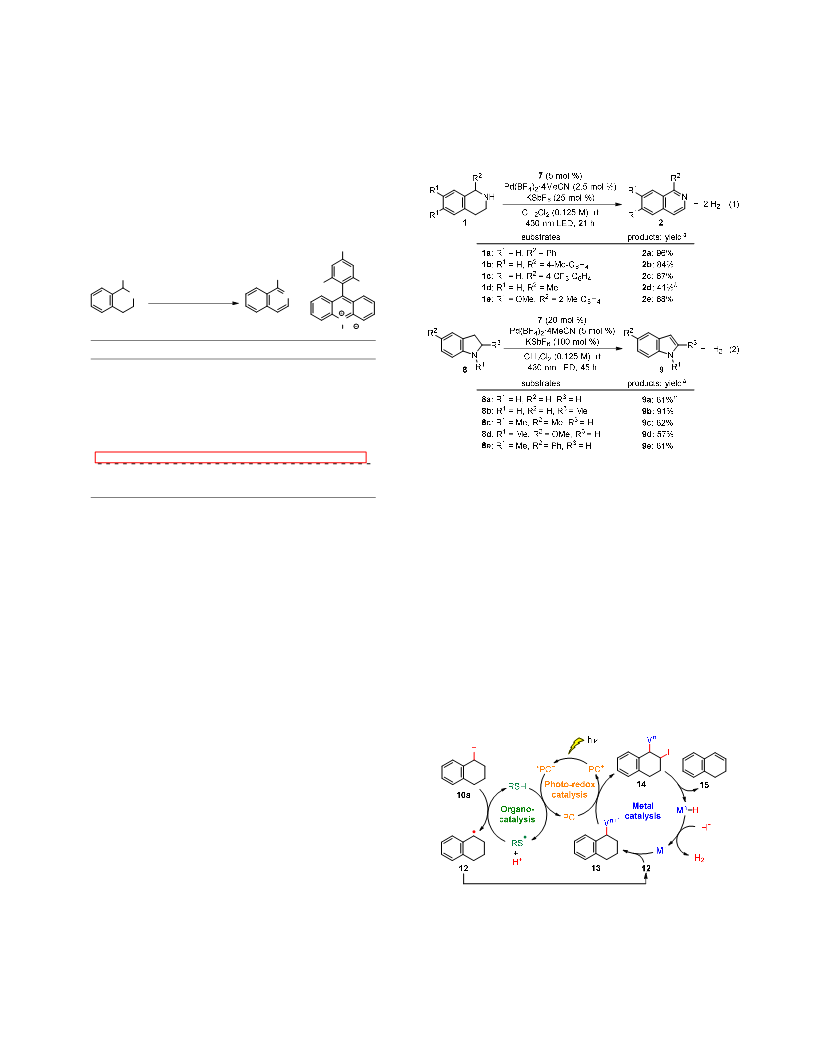
imide organocatalyst. These are the first molecular catalysts for
acceptorless dehydrogenation of N-heterocycles and hydrocar-
bons at room temperature and under visible light irradiation
conditions. The complete release of two-molar equivalents of
hydrogen gas from tetrahydroisoquinolines and tetrahydronaph-
thalenes is also unique in these processes. This study demon-
strates that proper hybridization of different catalyst types ena-
bles CAD from N-heterocycles and hydrocarbons under mild con-
ditions. Detailed studies to elucidate the reaction mechanism are
ongoing in our laboratories.
(7) (a) Nomura, K.; Saito, Y. J. Chem. Soc., Chem. Commun. 1988, 161.
(b) Maguire, J. A.; Boese, W. T.; Goldman, A.S. J. Am. Chem. Soc. 1989,
11, 7088. (c) Chowdhury, A. D.; Weding, N.; Julis, J.; Franke, R.; Jackstell,
R.; Beller, M. Angew Chem. Int. Ed. 2014, 53, 6477. (d) Chowdhury, A. D.;
Julis, J.; Grabow, K.; Hannebauer, B.; Bentrup, U.; Adam, M.; Franke, R.;
Jackstell, R.; Beller, M. ChemSusChem 2015, 8, 323.
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
1
(8) West, J. G.; Huang, D.; Sorensen, E. J. Nat. Commun. 2015, 6, 10093.
(9) For recent reviews of photo-redox catalysis in organic synthesis,
see; (a) Eckenhoff, W. T.; Eisenberg, R. Dalton Trans. 2012, 41, 13004. (b)
Prier, C. K.; Rankic, D. A.; MacMillan D. W. C. Chem. Rev. 2013, 113, 5322.
(
c) Xi, Y.; Yi, H.; Lei, A. Org. Biomol. Chem. 2013, 11, 2387. (d) Schultz, D.
M.; Yoon, T. P. Science 2014, 343, 985.
10) For recent representative reports of binary hybrid catalysis com-
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
(
ASSOCIATED CONTENT
Supporting Information
prising photo-redox catalysis and transition metal catalysis, see; (a) Kal-
yani, D.; McMurtrey, K. B.; Neufeldt, S. R.; Sanford, M. S. J. Am. Chem. Soc.
2
9
1
011, 133, 18566. (b) Ye, Y.; Sanford, M. S. J. Am. Chem. Soc. 2012, 134,
034. (c) Sahoo, B.; Hopkinson, M. N.; Glorius, F. J. Am. Chem. Soc. 2013,
35, 5505. (d) Shu, X.; Zhang, M; He, Y.; Frei, H.; Toste, F. D. J. Am. Chem.
The Supporting Information is available free of charge via the
Internet at http://pubs.acs.org.
Soc. 2014, 136, 5844. (e) Tellis, J. C.; Primer, D. N.; Molander, G. A. Sci-
ence 2014, 345, 433. (f) Zuo, Z.; Ahneman, D. T.; Chu, L.; Terrett, J. A.;
Doyle, A. G.; MacMillan, D. W. C. Science 2014, 345, 9195. (g) Cheng, W.-
M.; Shang, R.; Yu, H.-Z.; Fu, Y. Chem. Eur. J. 2015, 21, 13191. (h) Joe, C. L.;
Doyle, A. G. Angew. Chem. Int. Ed. 2016, 55, 4040. (i) Heitz, D. R.; Tellis, J.
C.; Molander, G. A. J. Am. Chem. Soc. 2016, 138, 12715. (j) Shields, B.
J.; Doyle, A. G. J. Am. Chem. Soc. 2016, 138, 12719.
(11) Reviews of hybrid catalysis merging photo-redox catalysts and
other catalysts: (a) Hopkinson, M. N.; Sahoo, B.; Li, J.-L.; Glorius, F. Chem.
Eur. J. 2014, 20, 3874. (b) Levin, M. D.; Kim, S.; Toste, F. D. ACS Cent. Sci.
2016, 2, 293. (c) Skubi, K. L.; Blum, T. R.; Yoon, T. P. Chem. Rev. 2016, 116,
Experimental details and characterization data (PDF)
AUTHOR INFORMATION
Corresponding Author
Author Contributions
ǁ
These authors contributed equally.
1
0035.
(12) (a) A similar approach for the generation of amidyl metal species
Notes
The authors declare no competing financial interests.
from amides under ambient conditions in the absence of strong base was
reported by Knowles. For a review, see: Nguyen, L. Q.; Knowles, R. R. ACS
Catal. 2016, 6, 2894. (b) An alternative possibility involving a benzyl radi-
cal generated from 3, followed by interception with the metal catalyst,
cannot be excluded.
ACKNOWLEDGMENT
This work was supported in part by ERATO from JST (MK) and a
Grant-in-Aid for Scientific Research (B) (No. 16H04125) from JSPS
(13) See Supporting Information (SI) for more details.
(14) (a) Brooks, E. H.; Glockling, F. J. Chem. Soc. A, 1967, 1030. (b) Ger-
ber, R.; Fox, T.; Frech, C. M. Chem. Eur. J. 2010, 16, 6771.
(
S. Masaoka). SK and M. Kojima thank JSPS for the fellowship. M.
Kojima thanks the Graduate Program for Leaders in Life Innova-
tion (GPLLI) for fellowships. We also thank Jun Ozawa for initially
providing 18.
(
15) (a) Kotani, H.; Ohkubo, K.; Fukuzumi, S. J. Am. Chem. Soc. 2004,
26, 15999. (b) Fukuzumi, S.; Ohkubo, K.; Suenobu, T. Acc. Chem. Res.
2014, 47, 1455. (c) Nicewicz, D. A.; Nguyen, T. M. ACS Catal. 2014, 4, 355.
16) Generation of two-molar equivalents of hydrogen gas was con-
1
(
firmed by a two-pot transfer hydrogenation experiment, connecting the
reaction atmosphere of the dehydrogenation reaction to a rhodium-
catalyzed hydrogenation reaction of cyclododecene performed in anoth-
er vessel. For detailed experiments, see SI.
(17) During preparation of this manuscript, MacMillan and coworkers
reported a cross-coupling reaction between aliphatic substrates and aryl
bromides by developing a triple catalysis involving an iridium photo-
redox catalyst, a quinuclidine hydrogen atom transfer organocatalyst,
and a nickel catalyst. See, Shaw, M. H.; Shurtleff, V. W.; Tarrett, J. A.;
Cuthbertson, J. D.; MacMillan, D. W. C. Science 2016, 352, 1304.
REFERENCES
(
1) (a) Dobereiner, G. E.; Crabtree, R. H. Chem. Rev. 2010, 110, 681. (b)
Gunanathan, G.; Milstein, D. Science 2013, 341, 249.
2) (a) Armaroli, N.; Balzani, V. ChemSusChem 2011, 4, 21. (b) Preuster,
(
P.; Papp, C.; Wasserscheid, P. Acc. Chem. Res. 2017, 50, 74.
(3) (a) Yamaguchi, R.; Ikeda, C.; Takahashi, Y.; Fujita, K. J. Am. Chem.
Soc. 2009, 131, 8410. (b) Wu, J.; Talwar, D.; Johnston, S.; Yan, M.; Xiao, J.
Angew. Chem. Int. Ed. 2013, 52, 6983. (c) Fujita, K.; Tanaka, Y.; Kobayashi,
M.; Yamaguchi, R. J. Am. Chem. Soc. 2014, 136, 4829. (d) Manas, M. G.;
Sharninghausen, L. S.; Lin, E.; Crabtree, R. H. J. Organomet. Chem. 2015,
(18) (a) Qvortrup, K.; Rankic, D. A.; MacMillan, D. W. C. J. Am. Chem.
Soc. 2014, 136, 626. (b) Cuthbertson, J. D.; MacMillan, D. W. C. Nature
2015, 519, 74. (c) Jin, J. MacMillan, D. W. C. Nature 2015, 525, 87. (d)
Jeffrey, J. L.; Terrett, J. A.; MacMillan, D. W. C. Science 2015, 349, 1532.
(19) A combination of 7 and Pd(BF ) •4MeCN was also optimum in the
7
92, 184. (e) Talwar, D.; Gonzalez-de-Castro, A.; Li, H. Y.; Xiao, J. Angew.
Chem. Int. Ed. 2015, 54. 5223.
4) (a) Luca, O. R.; Huang, D. L.; Takase, M. K.; Crabtree, R. H. New J.
(
Chem. 2013, 37, 3402. (b) Chakraborty, S.; Brennessel, W. W.; Jones, W.
D. J. Am. Chem. Soc. 2014, 136, 8564. (c) Xu, R.; Chakraborty, S.; Yuan, H.;
Jones, W. D. ACS Catal. 2015, 5, 6350.
4
2
case of CAD from 10a. See SI.
(20) Electrochemical and photochemical studies indicated that sulfur-
●
(5) (a) Maier, A. F. G.; Tussing, S.; Schneider, T.; Florke, U.; Qu, Z-W.;
centered radical species RS would be generated from thiophosphate 18
Grimme, S.; Paradies, J. Angew. Chem. Int. Ed. 2016, 55, 12219. (b) Koji-
ma, M.; Kanai, M. Angew. Chem. Int. Ed. 2016, 55, 12224.
via a single electron oxidation of 18 by the excited state of photo-redox
catalyst 7. SET from 18 to photo-excited 7 is also thermodynamically
feasible, based on their oxidation potential values. See SI for details.
(21) The ternary hybrid catalyst system produced less satisfactory re-
sults than the binary system in CAD from N-heterocycles (see Scheme 1):
CAD from 1a using 7 (5 mol %), 18 (2.5 mol %), and Pd(BF ) •4MeCN (2.5
(6) (a) Choi, J.; Roy, M. H. A.; Brookhart, M.; Goldman, A. S. Chem. Rev.
2
011, 111, 1761. (b) Crabtree, R. H.; Mihelcic, J. M.; Quirk, J. M. J. Am.
Chem. Soc., 1979, 101, 7738. (c) Crabtree, R. H.; Mellea, M. F.; Mihelcic, J.
M.; J. Am. Chem. Soc. 1982, 104, 107. (d) Burk, M. J.; Crabtree, R. H.;
McGrath, D. V. J. Chem. Soc., Chem. Commun. 1985, 1829. (e) Xu, W.-W.;
Rosini, G.P.; Gupta, M.; Jensen, C. M.; Kaska, W. C.; Krogh-Jespersena, K.;
Goldman, A. S. Chem. Commun. 1997, 2273. (f) Kusumoto, S.; Akiyama,
M.; Nozaki, K. J. Am. Chem. Soc. 2013, 135, 18726.
4
2
mol %) for 20 h afforded products 2a and 6 in 15% and 12% yields, re-
spectively.
ACS Paragon Plus Environment

 Kato, Shota
Kato, Shota
 Saga, Yutaka
Saga, Yutaka
 Kojima, Masahiro
Kojima, Masahiro
 Fuse, Hiromu
Fuse, Hiromu
 Matsunaga, Shigeki
Matsunaga, Shigeki
 Fukatsu, Arisa
Fukatsu, Arisa
 Kondo, Mio
Kondo, Mio
 Masaoka, Shigeyuki
Masaoka, Shigeyuki
 Kanai, Motomu
Kanai, Motomu