Organic Letters
Letter
4i, and 5c. This material is available free of charge via the
Scheme 2. Proposed Mechanism
AUTHOR INFORMATION
Corresponding Author
■
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
This work was supported by the National Natural Science
Foundation of China (21272232).
■
REFERENCES
(1) Shimizu, M.; Hiyama, T. Eur. J. Org. Chem. 2013, 8069.
(2) Fonseca-Berzal, C.; Rojas Ruiz, F. A.; Escario, J. A.; Kouznetsov,
■
́
V. V.; Gomez-Barrio, A. Biorg. Med. Chem. Lett. 2014, 24, 1209.
(3) Koike, T.; Hoashi, Y.; Takai, T.; Nakayama, M.; Yukuhiro, N.;
Ishikawa, T.; Hirai, K.; Uchikawa, O. J. Med. Chem. 2011, 54, 3436.
(4) (a) Ryckebusch, A.; Garcin, D.; Lansiaux, A.; Goossens, J.-F.;
́
Baldeyrou, B.; Houssin, R.; Bailly, C.; Henichar, J.-P. J. Med. Chem.
2008, 51, 3617. (b) Tseng, C.-H.; Tzeng, C.-C.; Yang, C.-L.; Lu, P.-J.;
Liu, Y.-P.; Chen, H.-L.; Chen, C.-Y.; Yang, C.-N.; Chen, Y.-L. Mol.
Divers. 2013, 17, 781.
(5) Kouznetsov, V. V.; Romero, B. A. R.; Saavedra, L. A. Synthesis
2009, 4219.
(6) Upadhayaya, R. S.; Lahore, S. V.; Sayyed, A. Y.; Dixit, S. S.;
Shinde, P. D.; Chattopadhyaya, J. Org. Biomol. Chem. 2010, 8, 2180.
(7) Upadhayaya, R. S.; Dixit, S. S.; Foldesi, A.; Chattopadhyaya, J.
̈
Bioorg. Med. Chem. 2013, 23, 2750.
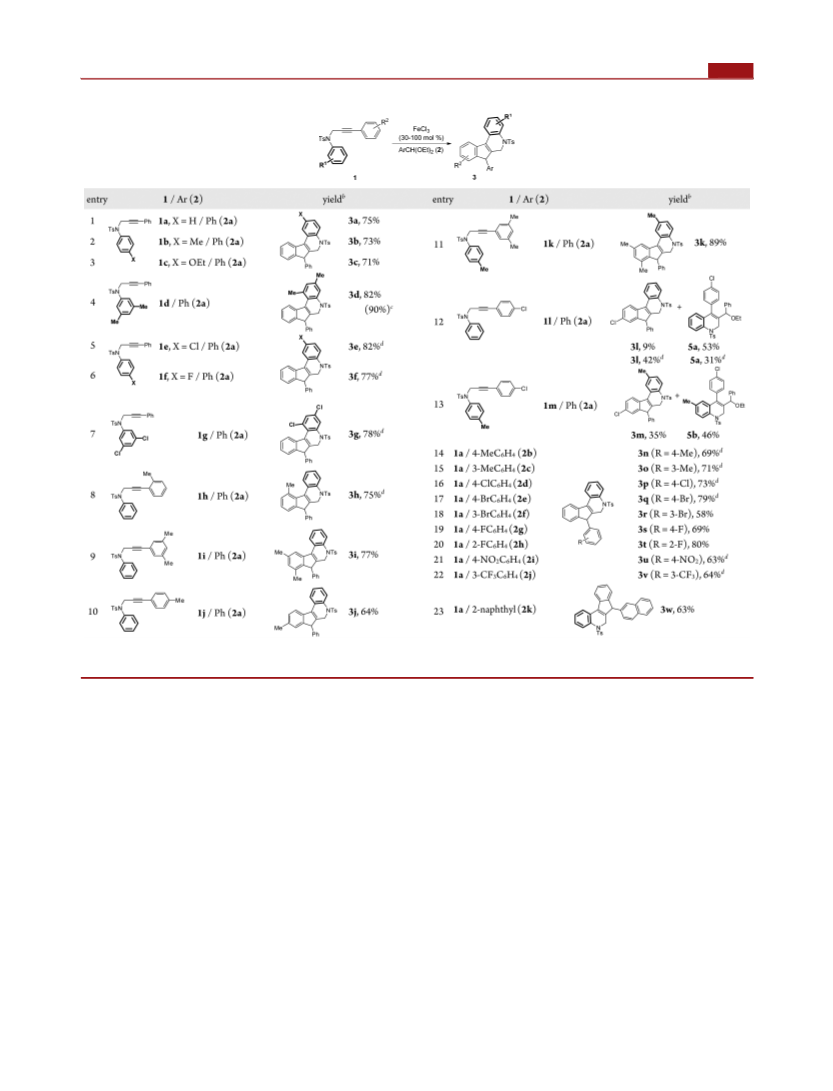
to 4a with FeCl3 or FeBr3 as the promoter. These results have
revealed that both 5 and 3 can act as the intermediates to form
4 in the catalytic cycle. 4-Phenylquinoline (6a)19 could also be
utilized to access 4a, further suggesting that species of types 5
and 6 may be generated as the reaction intermediates. It is
noteworthy that 3a, 4i, and 5c were structurally confirmed by
X-ray crystallographic analysis (see the Supporting Informa-
tion).
A plausible mechanism is proposed (Scheme 2). Acetal 2a
initially reacts with FeX3 (X = Cl or Br) to form FeX3(OEt)−
anion (A) and oxocarbonium cation PhCHOEt+ (B).17,18
Cation B interacts with propargylaniline 1a to generate vinyl
carbocation C stabilized by an aryl group, which undergoes
intramolecular Friedel−Crafts reaction to yield D. Deprotona-
tion of D by species A forms intermediate 5c and ethanol,
regenerating FeX3. Following path a, species 5c is converted to
product 3a20 via the possible cationic species E21 and F18
assisted by FeX3. Compound 3a further reacts with FeX3 to
undergo detosylation/aromatization,12 forming 4a. Compound
5c may also react with FeX3 to form 6a via species H by
detosylation/aromatization (path b), which further undergoes
carboarylation with FeX3 to furnish 4a and ethanol and
regenerate the catalyst.
(8) Jiang, B.; Feng, B.-M.; Wang, S.-L.; Tu, S.-J.; Li, G. G. Chem.
Eur. J. 2012, 18, 9823.
(9) Liu, X.; Zhang, Q.; Zhang, D. Y.; Xin, X. Q.; Zhang, R.; Zhou, F.
G.; Dong, D. W. Org. Lett. 2013, 15, 776.
(10) (a) Luo, Y.; Pan, X. L.; Yu, X. X.; Wu, J. Chem. Soc. Rev. 2014,
43, 834. (b) Gulevich, A. V.; Dudnik, A. S.; Chernyak, N.; Gevorgyan,
V. Chem. Rev. 2013, 113, 3084.
(11) Selected recent examples, see: (a) Seoane, A.; Casanova, N.;
Quinones, N.; Mascarenas, J. L.; Gulías, M. J. Am. Chem. Soc. 2014,
̃
̃
136, 834. (b) Zi, W. W.; Toste, F. D. J. Am. Chem. Soc. 2013, 135,
12600. (c) Walkinshaw, A. J.; Xu, W. S.; Suero, M. G.; Gaunt, M. J. J.
Am. Chem. Soc. 2013, 135, 12532.
(12) (a) Yamamoto, Y. Chem. Soc. Rev. 2014, 43, 1575. (b) Zeng, X.
M. Chem. Rev. 2013, 113, 6864. (c) Komeyama, K.; Igawa, R.; Takaki,
K. Chem. Commun. 2010, 46, 1748.
(13) Chen, M.; Sun, N.; Liu, Y. H. Org. Lett. 2013, 15, 5574.
(14) Pan, X. L.; Luo, Y.; Wu, J. Org. Biomol. Chem. 2012, 10, 1969.
(15) (a) Gopalaiah, K. Chem. Rev. 2013, 113, 3248. (b) Sun, C.-L.; Li,
B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293. (c) Sarhan, A. A. O.; Bolm,
C. Chem. Soc. Rev. 2009, 38, 2730.
́
(16) (a) Richard, V.; Ipouck, M.; Merel, D. S.; Gaillard, S.; Whitby, R.
J.; Witulski, B.; Renaud, J.-L. Chem. Commun. 2014, 50, 593. (b) Paul,
K.; Bera, K.; Jalal, S.; Sarkar, S.; Jana, U. Org. Lett. 2014, 16, 2166.
(c) Rana, S.; Brown, M.; Mukhopadhyay, C. RSC Adv. 2013, 3, 3291.
(17) (a) Xu, T. Y.; Yang, Q.; Li, D. P.; Dong, J. H.; Yu, Z. K.; Li, Y. X.
Chem.Eur. J. 2010, 16, 9264. (b) Xu, T. Y.; Yu, Z. K.; Wang, L. D.
Org. Lett. 2009, 11, 2113.
In summary, FeX3-mediated tandem reactions of propargy-
lanilines with aromatic aldehyde acetals form indeno[2,1-
c]quinolines in good to excellent yields through carboarylation/
cyclization under mild conditions. The present synthetic
method provides a concise and nontoxic metal-mediated
route to highly functionalized heteropolycyclic architectures.
(18) Xu, T. Y.; Yang, Q.; Ye, W. J.; Jiang, Q. B.; Xu, Z. Q.; Chen, J.
P.; Yu, Z. K. Chem.Eur. J. 2011, 17, 10547.
(19) Gurunathan, S.; Perumal, P. T. Tetrahedron Lett. 2011, 52, 1783.
(20) (a) Sawama, Y.; Shishido, Y.; Kawajiri, T.; Goto, R.; Monguchi,
Y.; Sajiki, H. Chem.Eur. J. 2014, 20, 510. (b) Stadler, D.; Bach, T.
Angew. Chem., Int. Ed. 2008, 47, 7557. (c) Iovel, I.; Mertins, K.;
Kischel, J.; Zapf, A.; Beller, M. Angew. Chem., Int. Ed. 2005, 44, 3913.
(21) Sawama, Y.; Goto, R.; Nagata, S.; Shishido, Y.; Monguchi, Y.;
Sajiki, H. Chem.Eur. J. 2014, 20, 2631.
ASSOCIATED CONTENT
* Supporting Information
■
S
Complete experimental procedures and characterization data
for the prepared compounds; X-ray crystallographic data for 3a,
D
dx.doi.org/10.1021/ol503039j | Org. Lett. XXXX, XXX, XXX−XXX

 Yang, Qin
Yang, Qin
 Xu, Tongyu
Xu, Tongyu
 Yu, Zhengkun
Yu, Zhengkun