G Model
CATTOD-8977; No. of Pages13
ARTICLE IN PRESS
10
S. Noël et al. / Catalysis Today xxx (2014) xxx–xxx
hydrogel spontaneously returned to the gel state and consequently
the products could be easily recovered. The recycling of the NPs
entrapped in the hydrogel has been successfully performed using
1-decene (Scheme 12).
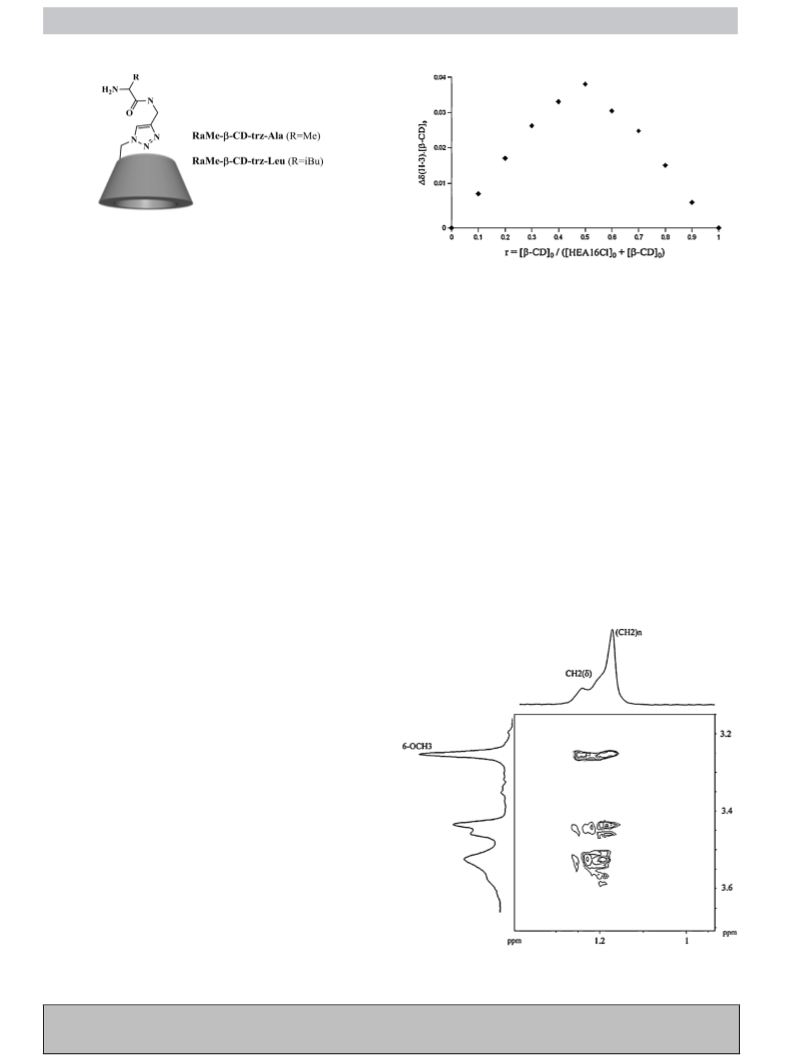
Scheme 11. Thermoresponsive N-alkylpyridinium amphiphile-(␣-CD)2 hydrogel
obtained by self-assembly [68].
6. Adsorption of CD-stabilized metal(0) NPs onto porous
materials
the polycarboxylate solution. The TOF was equal to 400 h−1, very
close to the value of the polycarboxylate alone but the colloidal
suspension after the catalytic test was unstable. All these experi-
ments undoubtedly supported the view that the -CD covalently
linked to the polymer chain induced a significant effect in terms of
between the cyclodextrin cavity and the hydrophobic substrate
could be confirmed by the 1-tetradecene hydrogenation in pres-
ence of 1-adamantane-carboxylate sodium salt, which is known
to strongly interact with -CD [31]. Thus, an activity decrease was
measured when the guest was added and this result confirmed that
the cyclodextrin influenced the catalytic behavior of the NPs by
facilitating the meeting of the substrate with the metal surface.
The recyclability was studied by reusing the aqueous catalytic
layer in five successive hydrogenation runs of 1-tetradecene. No
loss of stability and activity was noted. The rhodium leaching in
the organic phase of each catalytic test was very low (<0.2 ppm)
while the TEM experiments confirmed the robustness of the col-
loidal suspensions, with no change in terms of particle size and
morphology.
The development of efficient heterogeneous catalytic systems
is a well-developed research area both in academic and indus-
trial domains. Among the methods which are referenced to obtain
supported metallic nanoparticles, their deposition onto a porous
support from stabilized colloidal suspensions has received consid-
erable attention over the past decade. At the end of the synthesis,
the excess stabilizing molecules can be removed from the nanopar-
ticle surface by washing or thermal treatments to increase the
accessibility of active sites to reactants and intermediates. How-
ever, the design of well-structured and stable supported metal
NPs should take into account not only the stability of the solvent
dispersed particles, but also the metal–support interactions that
exert a dominant effect during the deposition step. Thus, among
the materials used as carriers for nanoparticles immobilization,
porous carbon materials are of increasing interest because of their
surface area, high thermal and chemical stabilities, high adsorp-
tion capacity). In this context and knowing that cyclodextrins can
act as efficient protective agents for the stabilization of ruthenium
NPs in aqueous phase and can spontaneously adsorb onto car-
bon supports [70], we have studied their use in the preparation of
carbon-supported ruthenium catalysts for gas-phase hydrogena-
tion reactions. The idea was to take advantage of an easy anchoring
of the metallic nanoparticles onto the carbon support via the
cyclodextrin combined with interesting activities and selectivities.
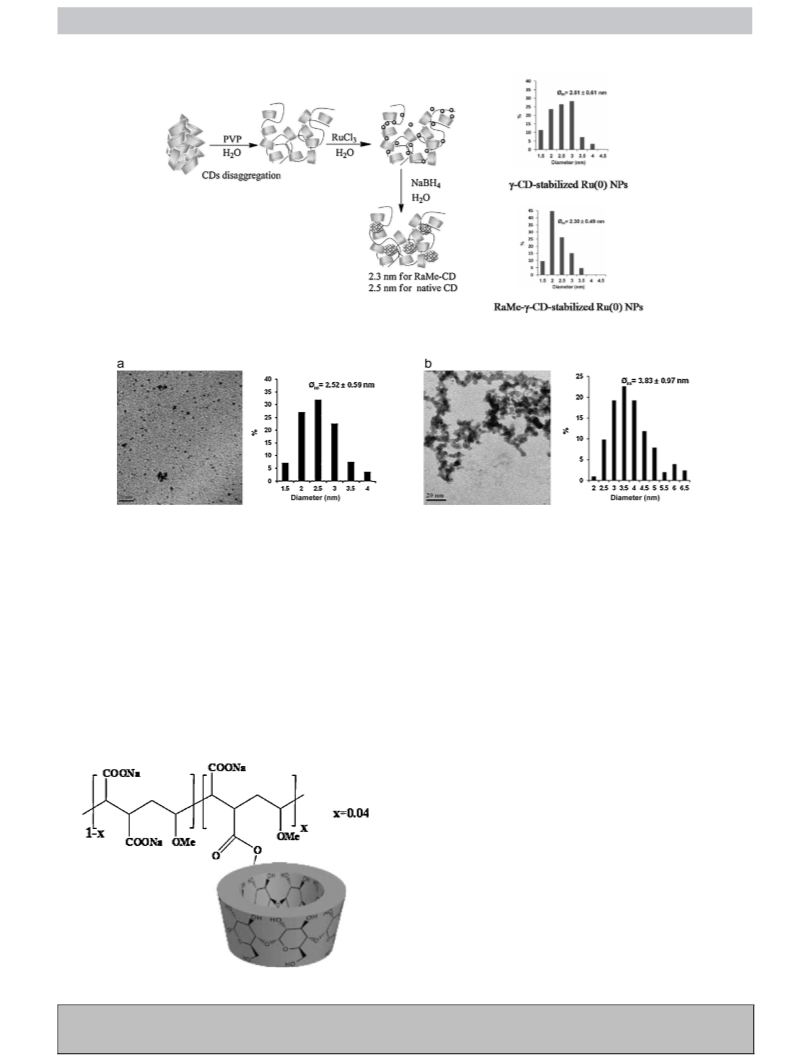
To validate the strategy, a series of carbon-supported ruthe-
nium nanocatalysts were prepared by the adsorption on a porous
activated carbon of Ru(0) NPs pre-formed in aqueous solution by
chemical reduction of RuCl3 in the presence of RaMe-CD (␣-, -
and ␥-) [71,72]. After the adsorption step, the solids were recov-
ered by filtration and thoroughly washed with water. According to
nitrogen adsorption measurements, it has clearly been shown that
the immobilization of the RaMe-CD-stabilized Ru(0) NPs by deeply
affected the textural properties of the porous carbon carrier. It was
interesting to note that, whatever the initial molar ratio of CD to
RuCl3, the Ru content was about 1.4 0.2 wt.%. In addition, thermo-
gravimetric measurements proved that the prepared nanocatalysts
were thermally stable up to 235 ◦C under both inert and reducing
instance, the TEM characterization of the Ru-3-CD/C sample for
with an average diameter of 2.4 nm (Fig. 8).
Hydrogels are hydrophilic polymer networks that can absorb
a large amount of water but can not be dissolved in water. For
this reason, hydrogels have become popular in several applications
including catalysis [67]. For instance, polymer hydrogels proved to
be efficient templates for the in situ synthesis of metal NPs. Even
if these metal particles are embedded in the polymer matrix, they
showed catalytic activity. In parallel, responsive hydrogels can also
undergo a swelling transition in response to environmental stimuli
such as temperature, light or pH. Indeed, the thermoreversibility
allows the NPs stabilization at room temperature and their activa-
tion at high temperature. More precisely, once that metal NPs have
been embedded into the supramolecular matrix, the system can be
In our case, the polypseudorotaxane [68] template could have
been prepared from a mixture of N-alkylpyridinium amphiphilic
[py-N-(CH2)12OC6H3-3,5-(OMe)2]+ (Br−) and ␣-CD in the ratio of
1:2 (Scheme 11). The self-assembly of these molecules yields a ther-
moresponsive hydrogel, with a sol–gel transition temperature of
42 ◦C.
The synthesis of Ru(0) NPs was realized by classical chemical
analysis clearly highlighted the stabilization of a homogeneous dis-
persion of spherical Ru(0) NPs with an average diameter of 1.6 nm
within the hydrogel network, which is smaller than that observed
using surfactants [26,46] or ionic liquids [69] as Ru(0) NPs stabi-
lizers. This result emphasized the effective control exerted by the
hydrogel internal network structure over the Ru NPs growth (Fig. 7).
Very interestingly, the concept has been validated for the
catalytic hydrogenation of various substrates, ranging from
hydrophobic long-chain to hydrophilic olefins, such as 2-methyl-
3-buten-2-ol. Under H2 pressure ranging from 10 to 40 bar at 50 ◦C,
The catalytic activity of the carbon supported RaMe-CD stabi-
lized Ru NPs was evaluated in the hydrogenation of xylene isomers
in gas phase at 85 ◦C (Table 8).
First, the catalytic results have clearly shown that the Ru(0)
nanocatalysts prepared with randomly methylated cyclodextrins
are more efficient than the control Ru/C (entries 1 and 8). How-
ever, the catalytic activity depends on the cyclodextrin size and
initial CD/Ru ratio. The best results have been obtained with RaMe-
-CD with a molar ratio of cyclodextrin to ruthenium of 3. When
this ratio is greater, a lower accessibility of the substrate to the
metal site is postulated, leading to an activity decrease. In terms
Please cite this article in press as: S. Noël, et al., Cyclodextrin-based systems for the stabilization of metallic(0) nanoparticles and their

 No?l, Sébastien
No?l, Sébastien
 Léger, Bastien
Léger, Bastien
 Ponchel, Anne
Ponchel, Anne
 Philippot, Karine
Philippot, Karine
 Denicourt-Nowicki, Audrey
Denicourt-Nowicki, Audrey
 Roucoux, Alain
Roucoux, Alain
 Monflier, Eric
Monflier, Eric