
Journal of Heterocyclic Chemistry p. 692 - 698 (2017)
Update date:2022-08-11
Topics:
 Karatas, Erhan
Karatas, Erhan
 Ucan, Halil Ismet
Ucan, Halil Ismet
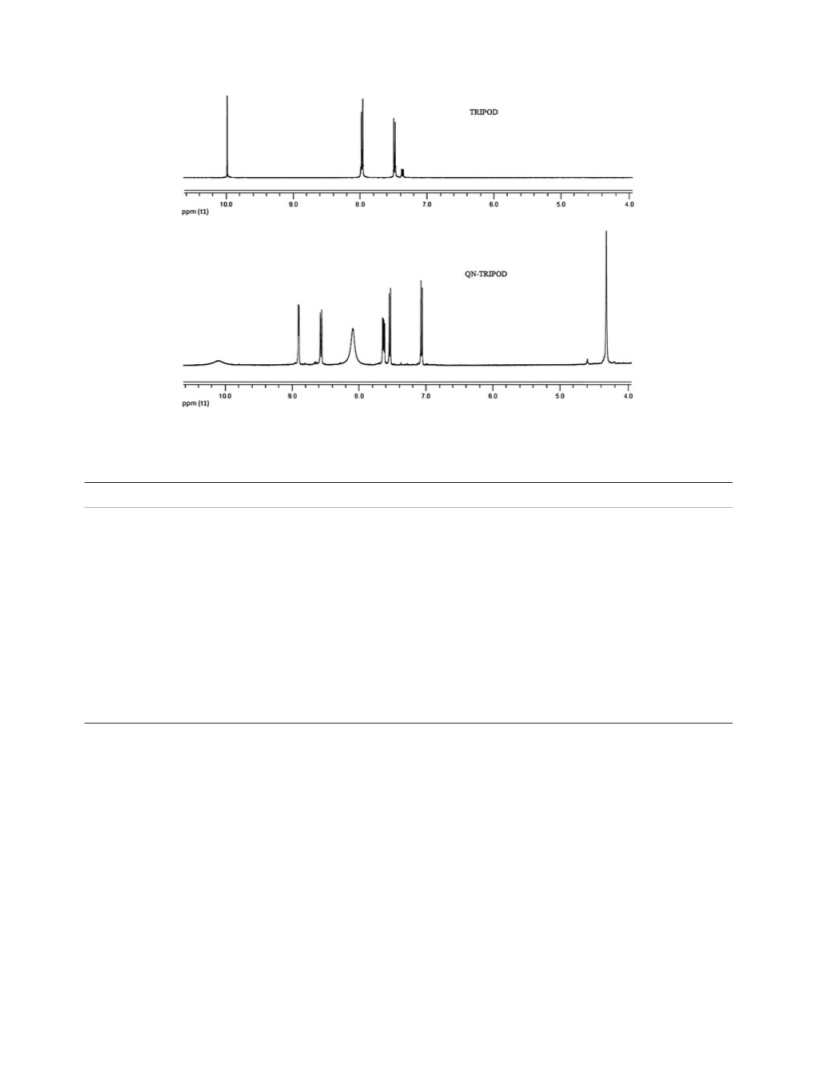
A novel Schiff base compound was synthesized, and its complexation properties with Fe(III) and Cr(III) were investigated. Tripodal ligand was synthesized by the reaction of s-triazine and 4-hydroxybenzaldehyde. Then a Schiff base involving 8-hydroxyquinoline was synthesized by the reaction of 5-aminomethyl-8-hydroxyquinoline (QN) and 2,4,6-tris(p-formylphenoxy)-1,3,5-triazine (TRIPOD) in methanol/chloroform media. The obtained Schiff base (QN-TRIPOD) was then reacted with four trinuclear Fe(III) and Cr(III) complexes including tetradentate Schiff bases N,N′-bis(salicylidene)ethylenediamine (salenH2)/bis(salicylidene)-o-phenylenediamine (SalophenH2). The synthesized ligand and complexes were characterized by means of elemental analysis carrying out1H NMR, FTIR spectroscopy, thermal analyses, and magnetic susceptibility measurements. Finally, metal ratios of the prepared complexes were determined by using atomic adsorption spectrometry.
View More




Hangzhou yi lu biont technology Co., LTD
Contact:+86-571-88152630
Address:Hangzhoushi HuafengRoad Yicheng3Building1001room.
Contact:0027-717-456976
Address:2ND FLOOR, 325 VAUSE ROAD, OVERPORT, 4001, SOUTH AFRICA
Shanghai Massive Chemical Technology Co., Ltd.
website:http://www.massivechem.com/
Contact:+86 21 34943721
Address:Room 435, 4th floor, Building 9, No. 2568 Gudai Road,Minhang District, Shanghai,
Hangzhou Hysen Pharma co.,Ltd.
website:http://www.hysenpharma.cn/
Contact:0086-571-88298791
Address:#701,Gudun Road Hangzhou
Guangzhou Reachin Chemical Co., Ltd
Contact:+86-20-37087379 ext.604
Address:A122C-1, Tianyuan Plaza, 401 Tianyuan Rd., Tianhe, Guangzhou, China
Doi:10.1002/ejic.202000910
(2020)Doi:10.1016/j.tet.2017.12.007
(2018)Doi:10.1021/jacs.7b05209
(2017)Doi:10.1039/c7nj01059a
(2017)Doi:10.1007/s00726-012-1424-6
(2013)Doi:10.1002/anie.202102353
()