
Helvetica Chimica Acta p. 969 - 977 (2013)
Update date:2022-08-11
Topics:
 Aga, Mushtaq A.
Aga, Mushtaq A.
 Kumar, Brijesh
Kumar, Brijesh
 Rouf, Abdul
Rouf, Abdul
 Shah, Bhahwal A.
Shah, Bhahwal A.
 Andotra, Samar S.
Andotra, Samar S.
 Taneja, Subhash C.
Taneja, Subhash C.
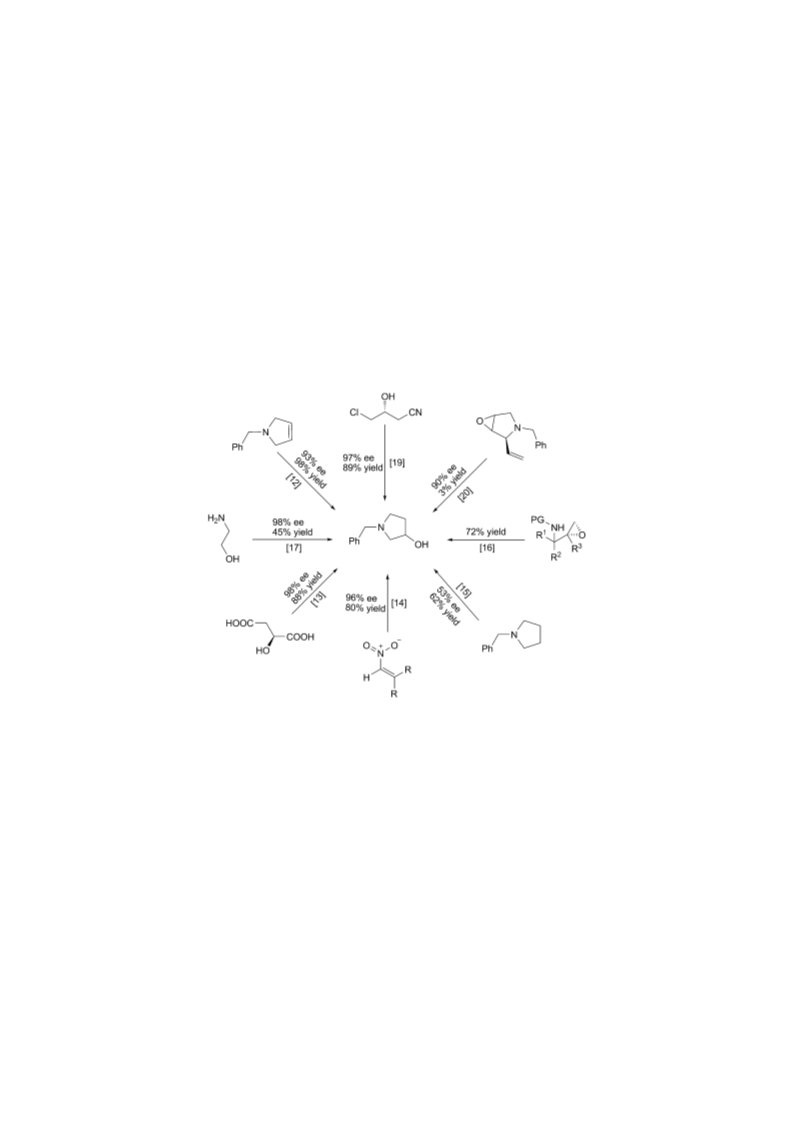
A facile and scalable methodology for the preparation of optically active (3S)-1-benzylpyrrolidin-3-ol (3), an important drug precursor, is reported. Starting from the naturally occurring alkaloid (-)-vasicine (1), a major alkaloid of the plant Adhatoda vasica, 3 was obtained in 84% overall yield (Scheme 3). Copyright
View More




Hunan Zhongqi Pharmaceutical Co., Ltd
website:http://www.hnzqzy.com
Contact:0730-8722288 13807308622
Address:Wanjiafan Road ,Yueyang Economic And Technological Development Zone ,Hunan,PRC
Hubei Sky Lake Chemical Co., Ltd.
Contact:+86-27-87385545
Address:Te-2, Xijiao Chemical Park, Yuekou Town, Tianmen, Hubei, China.
Contact:+852-8198 2399
Address:9E, Leapont Industrial Building, 18-28 Wo Liu Hang Road, Shatin, New Territories, Hong Kong
Qingdao Kylin Trading Co., Ltd.
Contact:0086-532-68979884/58972912/68972263/65/88171519
Address:Room 2308,A building International Trade Center No.230 Changjiang Middle Road of Qingdao Economic Development Zone,Shandong,China.
Fusilin chemical science & technology co., ltd.
Contact:532-80698166/86057573, +86-400-669-7885
Address:School of Material Science & Engineering, Shandong Uinversity of Science & Technology, Huangdao Zone, Qindao, Shandong
Doi:10.1016/j.electacta.2006.08.025
(2007)Doi:10.1016/S0040-4020(00)00850-4
(2000)Doi:10.1021/ja00130a008
(1995)Doi:10.1002/pola.28536
(2017)Doi:10.1016/0031-9422(95)00262-6
(1995)Doi:10.1016/0040-6031(90)80638-F
(1990)