Month 2016
Cyclization–oxidation of Benzylidenehydrazinecarbothioamides and Synthesis of
New Substituted Arylthiazolo [1,2,4]Triazols
(KBr, cmꢀ1): 1727 (C¼O), 1640 (C¼N). 1H-NMR
(DMSO-d6, 400MHz) δ= 1.33 (t, 3H, CH3, JHH = 7.2 Hz),
4.35 (q, 2H, CH2, JHH = 6.8 Hz), 7.01(s, 1H,C-H, alkene),
7.94 (d, 2H-aromatic, JHH =8.4 Hz), 8.06 (d, 2H-aromatic,
JHH = 8.4Hz) ppm. 13C-NMR (DMSO-d6, 100MHz)
δ= 13.94 (CH3), 39.9 (OMe), 62.10 (CH2), 110.85,
127.63, 128.69, 129.74, 129.83, 137.91 (C-aromatic),
149.91, 156.19 (C¼N), 158.74, 163.11 (C¼O) ppm. Anal.
Calcd for C14H10ClN3O3S: C, 50.08; H, 3.00; N, 12.51; S,
9.55%, Found: C, 50.26; H, 3.190; N, 12.62; S, 9.53%.
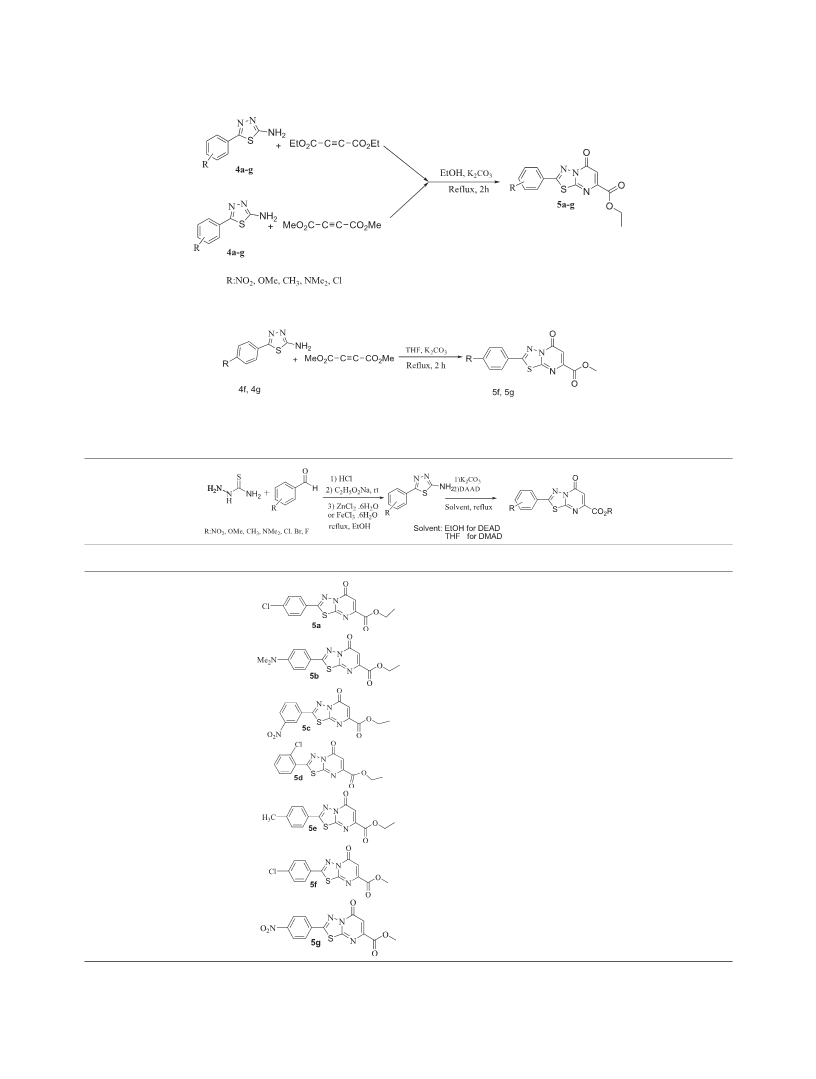
Ethyl
5-oxo-2-p-tolyl-5H-[1,3,4]thiadiazolo[3,2-α]pyrimi-
dine-7-carboxylate (5e).
Prepared according to General
Procedure, mp=198–199°C, yield= 85%, IR (KBr, cmꢀ1):
1735 (C¼O), 1608 (C¼N), 1H-NMR (DMSO-d6,
400 MHz) δ= 1.37 (t, 3H, JHH = 6.8 Hz), 2.41 (s, 3H,
CH3), 4.45 (q, 2H, JHH = 6.8 Hz), 6.65 (s,1H, C–H, alken),
7.44 (d, 2H- aromatic, JHH =8.0 Hz), 7.80 (d, 2H-
aromatic, JHH = 8.0 Hz) ppm, 13C-NMR (DMSO-d6,
100 MHz) δ= 13.82 (CH3), 21.10 (CH3), 63.01 (CH2),
110.86, 111.89 (CH-alkene), 126.80, 130.15, 138.93,
143.26 (C-aromatic), 156.79, 158.85 (C¼N), 164.33,
165.72 (C¼O) ppm. Anal. Calcd for: C15H13N3O3S: C,
57.13; H, 4.16; N, 13.33; S, 10.17%, Found: C, 57.01; H,
4.11; N, 13.37; S, 10.15%.
Ethyl
2-(4-(dimethylamino)phenyl)-5-oxo-5H-[1,3,4]
thiadiazolo[3,2-a] pyrimidine-7-carboxylate (5b).
Prepared
according to general procedure, mp = 197–199°C,
yield = 90%, IR (KBr, cmꢀ1): 1741 (C¼O), 1632 (C¼N),
1H-NMR (DMSO-d6, 400 MHz) δ =1.37 (t,3H,
JHH = 6.8Hz), 3.02 (s, 6H), 3.35 (s, 3H), 4.44 (q, 2H,
JHH = 6.8Hz), 6.56 (s, 1H, C-H, alkene), 6.80 (d,
Methyl 2-(4-chlorophenyl)-5-oxo-5H-[1,3,4]thiadiazolo[3,2-
α]pyrimidine-7-carboxylate (5f).
Prepared according to
general procedure, yield= 90%, IR (KBr, cmꢀ1): 1727
(C¼O), 1640 (C¼N), 1H-NMR (DMSO-d6, 400MHz)
δ= 3.80 (s,3H), 7.01 (d, 2H- aromatic, JHH =8.0Hz),
7.31 (s, 1H, C–H, alken), 7.68 (d, 2H-aromatic,
JHH = 8.0 Hz) ppm, 13C-NMR (DMSO-d6, 100MHz)
δ= 55.29 (CH3), 111.91, 114.46 (CH-alkene), 115.04,
123.54, 127.87, 127.89, 128.77, 128.90 (C-aromatic),
156.24, 160.25 (C¼N), 167.83, 167.90 (C¼O) ppm.
Anal. Calcd for: C13H8ClN3O3S: C, 48.53; H, 2.51; N,
13.06; S, 9.97%, Found: C, 48.51; H, 2.53; N, 13.14;
S, 9.95%.
4H-aromatic,
JHH = 8.8 Hz),
7.63(d,
4H-aromatic,
JHH = 8.8Hz) ppm. 13C-NMR (DMSO-d6, 100MHz)
δ= 13.85(CH3), 39.57 (NMe2), 53.69 (CH3), 62.92
(CH2), 111.23, 111.47 (C–H, alkene) 111.64, 113.89,
128.30, 138.94 (C-aromatic), 156.98, 159.07 (C¼N),
163.88, 165.79 (C¼O) ppm. Anal. Calcd for
C15H14N4O3S: C, 54.53; H, 4.27; N, 16.96; S, 9.71%,
Found: C, 54.82; H, 4.38; N, 17.01; S, 9.69%.
Ethyl 2-(3-nitrophenyl)-5-oxo-5H-[1,3,4]thiadiazolo[3,2-α]
pyrimidine-7-carboxylate (5c).
Prepared according to
Methyl 2-(4-nitrophenyl)-5-oxo-5H-[1,3,4]thiadiazolo[3,2-α]
general procedure, mp = 219–222°C, yield= 85%, IR
(KBr, cmꢀ1): 1745 (C¼O),1634 (C¼N), 1H-NMR
(DMSO-d6, 400MHz) δ= 1.40 (t, 3H, CH3, JHH = 7.2 Hz),
4.47 (q, 2H, CH2, JHH = 7.2 Hz), 6.73 (s,1H, C–H, alkene),
7.93 (t,1H-aromatic, JHH = 8Hz), 8.34 (d,1H-aromatic,
pyrimidine-7-carboxylate (5g).
Prepared according to
General Procedure, yield = 80%, IR (KBr, cmꢀ1): 1742
(C¼O), 1606 (C¼N), 1H-NMR (DMSO-d6, 400MHz)
δ= 3.98 (s,3H), 6.68 (s, 1H, C–H, alken), 7.70 (d, 2H-
aromatic,
JHH = 8.0 Hz),
7.93
(d,
2H-aromatic,
J
HH = 8.4Hz), 8.49 (m,1H-aromatic), 8.63 (t,1H-aromatic,
JHH = 8.0 Hz) ppm, 13C-NMR (DMSO-d6, 100MHz)
δ= 53.78 (CH3), 111.18, 112.31 (CH-alkene), 128.67,
128.78, 129.76, 137.47, 138.06, 138.72 (C-aromatic),
155.74, 159.27 (C¼N), 164.59, 165.67 (C¼O) ppm. Anal.
Calcd for C14H11N3O4S: C, 52.99; H, 3.49; N, 13.24; S,
10.10%, Found: C, 53.01; H, 3.42; N, 13.23; S, 10.12%.
JHH = 2Hz) ppm, 13C-NMR (DMSO-d6, 100MHz) δ =13.77
(CH3), 63.12 (CH2), 111.20, 111.35 (CH-alkene), 126.73,
126.88, 129.31, 131.45, 133.32, 138.09 (C-aromatic),
155.12, 158.71 (C¼N), 164.66, 166.21 (C¼O) ppm. Anal.
Calcd for C14H10N4O5S: C, 48.55; H, 2.91; N, 16.18; S,
9.26%, Found: C, 48.78; H, 2.95; N, 16.11; S, 9.28%.
Ethyl 2-(2-chlorophenyl)-5-oxo-5H-[1,3,4]thiadiazolo[3,2-α]
pyrimidine-7-carboxylate (5d).
Prepared according to
Acknowledgments. We gratefully acknowledge, the Vail-e-Asr
University of Rafsanjan Faculty Research Grant for financial
support.
General Procedure, mp = 155–157°C, yield = 95%, IR
(KBr, cmꢀ1): 1745 (C¼O), 1639 (C¼N).1H-NMR
(DMSO-d6, 400MHz) δ= 1.35 (t, 3H, CH3, JHH = 6.8 Hz),
4.43 (q, 2H, CH2, JHH = 6.8Hz), 6.70 (s, 1H, C-H,alken),
7.60 (dt,1H- aromatic, JHH = 8.0, 1.6 Hz), 7.67
(dt,1H-aromatic, JHH = 7.6, 1.6 Hz), 7.73 (dd,1H-aromatic,
REFERENCES AND NOTES
[1] Jain, A. K.; Sharma, S.; Vaidya, A.; Ravichandran, V.;
Agrawal, R. K. Chem Biol Drug Des 2013, 81(5), 557.
[2] Kumar, D.; Maruthi Kumar, N.; Chang, K.-H.; Shah, K. Eur J
Med Chem 2010, 45, 4664.
[3] Demirbas, A.; Sahin, D.; Demirbas, N.; Karaoglu, S. A. Eur J
Med Chem 2009, 44, 2896.
[4] Foroumadi, A.; Kargar, Z.; Sakhteman, A.; Sharifzadeh, Z.;
Feyzmohammadi, R.; Kazemi, M.; Shafiee, A. Bioorg Med Chem Lett
2006, 16, 1164.
J
HH = 8.0, 1.6Hz), 7.99 (d, d,1H-aromatic, JHH = 7.6,
1.6Hz) ppm. 13C-NMR (DMSO-d6, 100MHz) δC =13.79
(CH3), 63.05 (CH2), 112.48, 126.36 (CH-alkene), 128.27,
131.11, 133.69, 138.68 (C-aromatic), 153.38, 158.78
(C¼N), 164.60, 165.71(C¼O) ppm. Anal. Calcd for
C14H10ClN3O3S: C, 50.08; H, 3.00; N, 12.51; S, 9.55%,
Found: C, 50.13; H, 3.10; N, 12.42; S, 9.53%.
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Darehkordi, Ali
Darehkordi, Ali
 Zarezadeh Abarqouei, Behnam
Zarezadeh Abarqouei, Behnam
 Rahmani, Fariba
Rahmani, Fariba