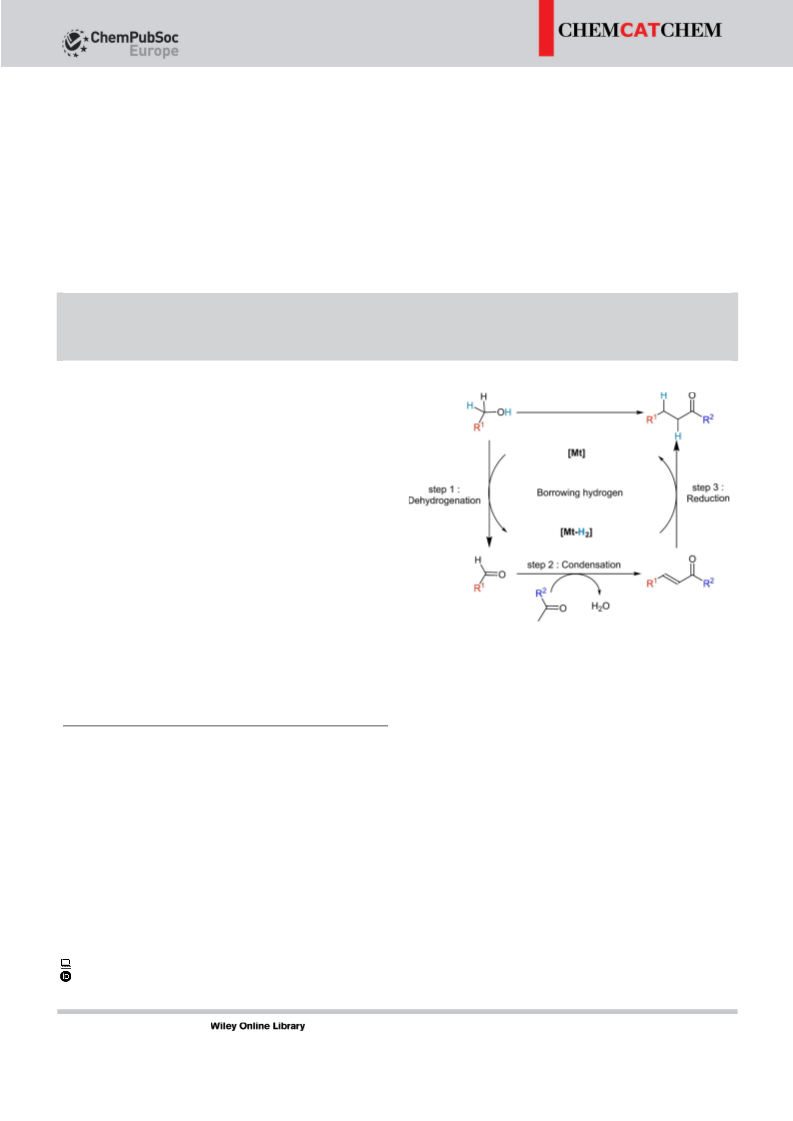
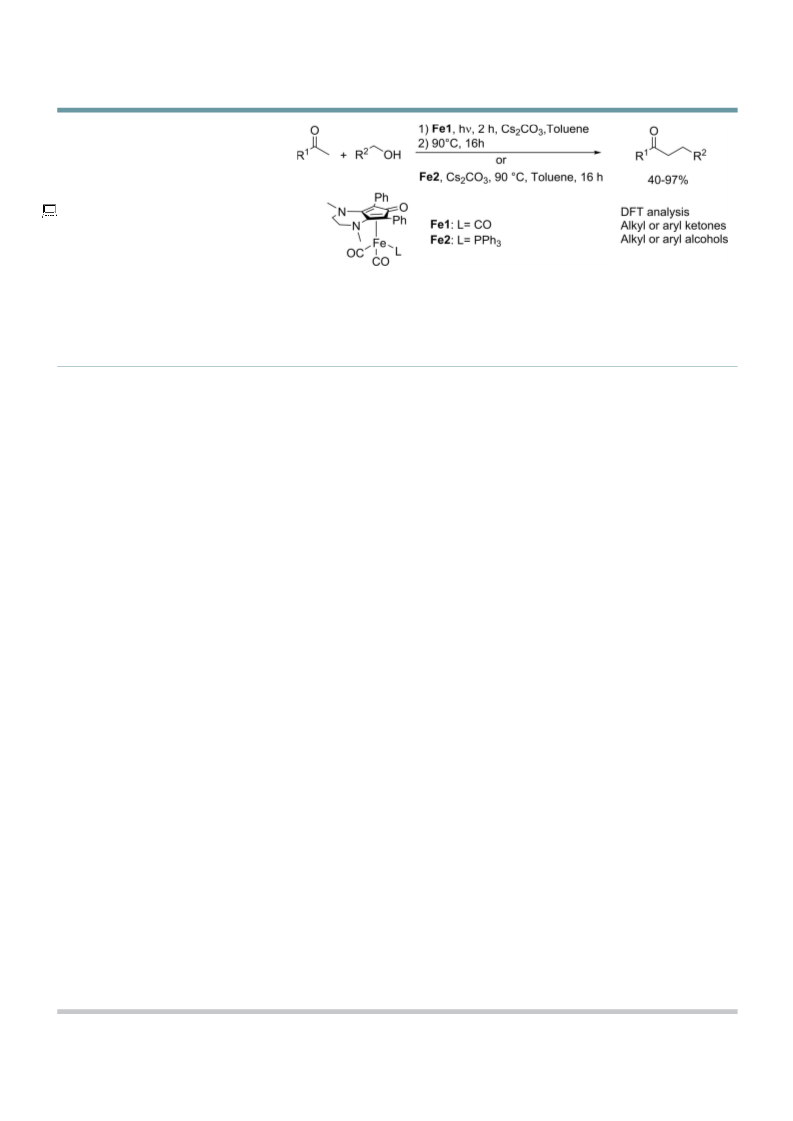
Full Papers
122 mg), complex Fe2 was obtained as an orange powder
(144 mg, 48%) after purification by flash column chromatography
on neutral aluminum oxide topped with a pad of Celite (eluent:
pentane/AcOEt [90:10] to [70:30]). X-ray-quality crystals were
grown by slow diffusion of pentane in dichloromethane (vide
Table 3. Alkylation of aromatic ketones with aliphatic alcohols or aliphat-
ic ketones with benzylic alcohols.
1
infra). H NMR (CDCl3, 400 MHz) d 7.82 (d, J=7.0 Hz, 4H), 7.39 (s,
2H), 7.34–7.20 (m, 9H), 7.16 (t, J=7.0 Hz, 4H), 7.07–7.03 (m, 6H),
3.49 (s, 4H), 2.59 (s, 6H) ppm. 13C NMR (CDCl3, 100 MHz) d=216.9,
160.9, 133.9, 133.8, 133.3, 133.2, 130.8, 128.9, 127.5, 127.4, 125.9,
108.6, 71.3, 47.1, 40.2 ppm. 31P NMR (CDCl3, 162 MHz) d=
50.68 ppm. IR (neat) n˜ =3058, 2925, 1969, 1902, 1603, 1573, 1542,
1498, 1484, 1470, 1434, 1417, 1380, 1354, 1332, 1251, 1175, 1089,
1029, 930, 846, 762, 750, 740, 693 cmꢀ1. HRMS [M+H]+ calculated
for C41H36N2O3PFe: 691.1813; found : 691.1812.
Entry[a]
Temp. [8C]
Yield [%][b]
1
110
70
2[c]
3
49
45
Synthesis of (1,4-dimethyl-5,7-diphenyl-1,2,3,4-tetrahydro-6H-cyclo-
penta[b]pyrazin-6-one) trimethyl phosphite dicarbonyl iron com-
plex [Fe3]: Following the general procedure, starting from complex
Fe1 (0.22 mmol, 100 mg) and trimethyl phosphite (0.23 mmol,
27.5 mL), complex Fe3 was obtained as a yellow powder (63 mg,
52%) after purification by flash column chromatography on neutral
aluminum oxide topped with a pad of Celite (eluent: CH2Cl2/MeOH
[100:0] to [98:2]). X-ray-quality crystals were grown by slow diffu-
sion of pentane in dichloromethane (see below). 1H NMR (CDCl3,
400 MHz) d=7.84 (d, J=7.3 Hz, 4H), 7.33 (t, J=7.3 Hz, 4H), 7.28–
7.26 (m, 2H), 3.44 (d, J=10.8 Hz, 9H), 3.38–3.28 (m, 2H), 3.11–3.03
(m, 2H), 3.29 ppm (s, 6H). 13C NMR (CDCl3, 100 MHz) d=214.9,
214.7, 163.1, 134.1, 132.1, 127.7, 126.9, 110.2, 69.8, 52.5, 49.1,
41.3 ppm. 31P NMR (CDCl3, 160 MHz) d=164.8 ppm. IR (neat) n˜ =
2951, 2851, 1989, 1938, 1909, 1603, 1593, 1540, 1497, 1444, 1419,
1380, 1355, 1177, 1059, 1020, 782, 739, 698 cmꢀ1. HRMS [M+H]+
calculated for C26H30FeN2O6P: 553.1191; found: 553.1199.
110
4
5
90
90
55
72
6
7
90
90
66
45
Synthesis of (1,4-dimethyl-5,7-diphenyl-1,2,3,4-tetrahydro-6H-cyclo-
penta[b]pyrazin-6-one) tributylphosphine dicarbonyl iron complex
[Fe4]: Following the general procedure, starting from complex Fe1
(0.44 mmol, 200 mg) and tributylphosphine (0.47 mmol, 116 mL),
complex Fe4 was obtained as an orange powder (219 mg, 79%)
after purification by flash column chromatography on neutral alu-
minum oxide topped with a pad of Celite (eluent: pentane/AcOEt
[100:0] to [9:1]). X-ray-quality crystals were grown by slow diffusion
of pentane in dichloromethane (see below). 1H NMR (CDCl3,
400 MHz) d=8.15 (d, J=7.4 Hz, 4H), 7.33–7.25 (m, 4H), 7.20 (t, J=
7.4 Hz, 2H), 3.42–3.35 (m, 2H), 3.31–3.24 (m, 2H), 2.49 (s, 6H),
1.43–1.37 (m, 6H), 1.06–0.86 (m, 12H), 0.68 ppm (t, J=7.1 Hz, 9H).
13C NMR (CDCl3, 100 MHz) d=217.3, 217.2, 157.8, 135.1, 130.5,
127.5, 126.3, 107.9, 70.5, 47.5, 40.8, 29.7, 25.2, 25.2, 24.3, 24.2, 23.6,
23.4, 13.6 ppm. 31P NMR (CDCl3, 162 MHz) d=32.24 ppm. IR (neat)
n˜ =2957, 2929, 2858, 2324, 2111, 1965, 1910, 1599, 1580, 1532,
1498, 1464, 1442, 1417, 1380, 1355, 1336, 1263, 1215, 1176, 1041,
1030, 913, 763, 742, 714, 698 cmꢀ1. HRMS [M+H]+ calculated for
C35H48FeN2O3P: 631.2752; found: 631.2761.
8
90
90
90
98
87
98
9
10
[a] General conditions: carbonyl derivative (1 mmol), alcohol (1.2 mmol),
complex Fe2 (2 mol%), CsOH (10 mol%) in toluene (2 mL) for 16 h.
[b] Yield was based on the purified product. [c] With Cs2CO3 (10 mol%) as
base.
General procedure for ligand exchange
Synthesis of (1,4-dimethyl-5,7-diphenyl-1,2,3,4-tetrahydro-6H-cyclo-
penta[b]pyrazin-6-one) tris(p-tolylphosphine) dicarbonyl iron com-
plex [Fe5]: Following the general procedure, starting from complex
Fe1 (0.44 mmol, 200 mg) and tris(p-tolyl)phosphine (0.47 mmol,
141 mg), complex Fe5 was obtained as an orange powder
(190 mg, 59%) after purification by flash column chromatography
on neutral aluminum oxide topped with a pad of Celite (eluent:
pentane/AcOEt [90:10] to [70:30]). X-ray-quality crystals were
grown by slow diffusion of pentane in dichloromethane (see
below).
In a flame-dried Schlenk tube under an argon atmosphere, iron
complex Fe1 (1 equiv.) and the corresponding phosphine or phos-
phite (1.06 equiv.) were introduced in freshly distilled and degassed
xylenes (mixture of isomers) (c=0.03m). The reaction mixture was
heated under reflux overnight and then the solvent was removed
under vacuum to furnish the crude product.
Synthesis of (1,4-dimethyl-5,7-diphenyl-1,2,3,4-tetrahydro-6H-cyclo-
penta[b]pyrazin-6-one) triphenylphosphine dicarbonyl iron com-
plex [Fe2]: Following the general procedure, starting from complex
Fe1 (0.44 mmol, 200 mg) and triphenylphosphine (0.47 mmol,
1H NMR (CDCl3, 400 MHz) d=7.66 (d, J=7.4 Hz, 4H), 7.15–7.02 (m,
8H), 6.99 (t, J=7.4 Hz, 4H), 6.71 (d, J=7.4 Hz, 6H), 3.36–3.31 (m,
&
ChemCatChem 2017, 9, 1 – 8
6
ꢁ 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ÝÝ These are not the final page numbers!

 Seck, Charlotte
Seck, Charlotte
 Mbaye, Mbaye Diagne
Mbaye, Mbaye Diagne
 Coufourier, Sébastien
Coufourier, Sébastien
 Lator, Alexis
Lator, Alexis
 Lohier, Jean-Fran?ois
Lohier, Jean-Fran?ois
 Poater, Albert
Poater, Albert
 Ward, Thomas R.
Ward, Thomas R.
 Gaillard, Sylvain
Gaillard, Sylvain
 Renaud, Jean-Luc
Renaud, Jean-Luc