LETTER
Selective Nitration of Aromatic Olefins
1999
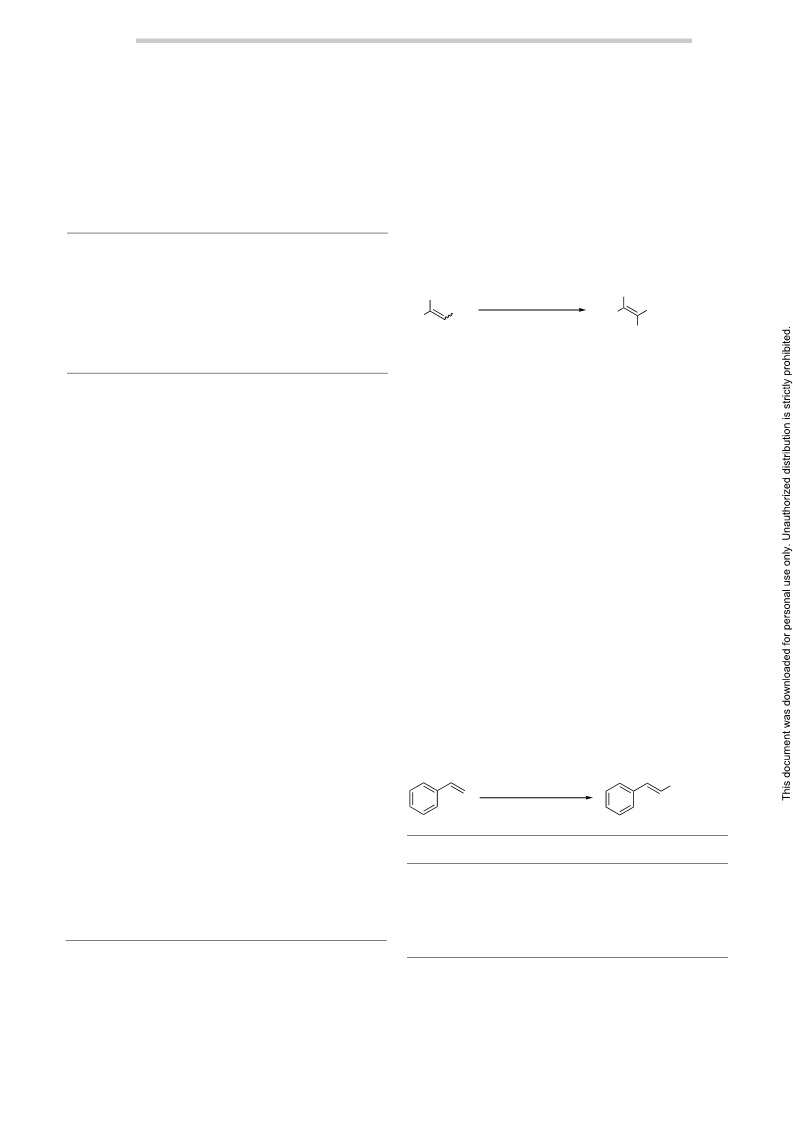
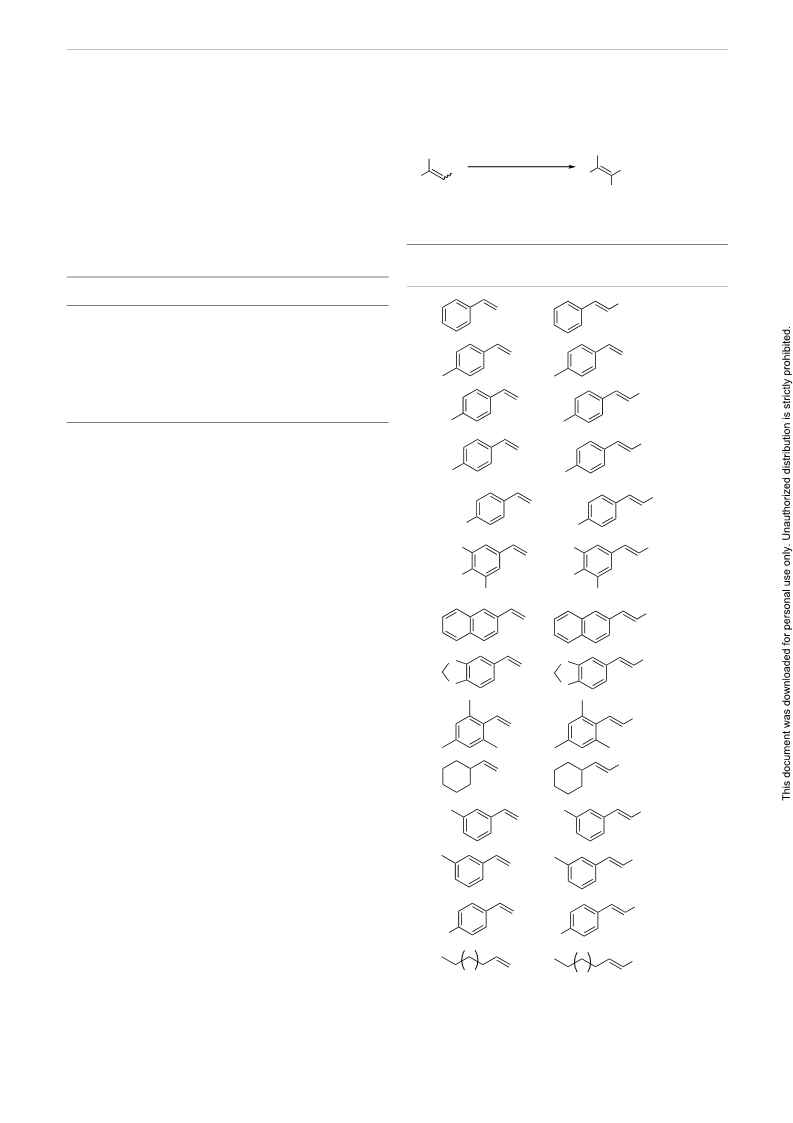
Table 3 Selective Nitration of Aromatic Olefinsa (continued)
A. T., Eds.; VCH: Weinheim, 1990, Chap. 1, 1–135.
(c) Perekalin, V. V.; Lipina, E. S.; Berestovitskaya, V. M.;
Efremov, D. A. Nitroalkenes: Conjugated Nitro Compounds;
Wiley: Chichester, 1994, Chap. 2, 53–168. (d) Barrett, A. G.
M. Chem. Soc. Rev. 1991, 20, 95. (e) Basavaiah, D.; Reddy,
B. S.; Badsara, S. S. Chem. Rev. 2010, 110, 5447.
(f) Tripathi, C. B.; Kayal, S.; Mukherjee, S. Org. Lett. 2012,
14, 3296. (g) Ishii, T.; Fujioka, S.; Sekiguchi, Y.; Kotsuki,
H. J. Am. Chem. Soc. 2004, 126, 9558. (h) Denmark, S. E.;
Thorarensen, A. Chem. Rev. 1996, 96, 137. (i) Evans, D. A.;
Mito, S.; Seidel, D. J. Am. Chem. Soc. 2007, 129, 11583.
(j) Liu, Y. K.; Nappi, M.; Arceo, E.; Vera, S.; Melchiorre, P.
J. Am. Chem. Soc. 2011, 133, 15212.
Claycop (1.5 equiv)
R2
R2
TEMPO (20 mol%)
R3
NO2
R1
E only
1,4-dioxane (5 mL)
air, 80 °C, 1 h
R1
R3
R1 = aromatic, aliphatic
R2, R3 = H
Entry Alkene
Productb
Yieldc
(%)
(3) (a) Naveen, T.; Maity, S.; Sharma, U.; Maiti, D. J. Org.
Chem. 2013, 78, 5949. (b) Maity, S.; Manna, S.; Rana, S.;
Naveen, T.; Mallick, A.; Maiti, D. J. Am. Chem. Soc. 2013,
135, 3355. (c) Maity, S.; Naveen, T.; Sharma, U.; Maiti, D.
Org. Lett. 2013, 15, 3384. (d) Manna, S.; Jana, S.; Saboo, T.;
Maji, A.; Maiti, D. Chem. Commun. 2013, 49, 5286.
(e) Eiichiro, H.; Tohru, Y.; Teruaki, M. Bull. Chem. Soc.
Jpn. 1995, 68, 3629. (f) Tinsley, S. W. J. Org. Chem. 1961,
26, 4723. (g) Grebenyuk, A. D.; Ismailova, R. A.;
Tokbolatov, R. B.; Ovadova, T. Zh. Org. Khim. 1990, 26,
680. (h) Corey, E. J.; Estreicher, H. J. Am. Chem. Soc. 1978,
100, 6294. (i) Kancharla, P. K.; Reddy, Y. S.; Dharuman, S.;
Vankar, Y. D. J. Org. Chem. 2011, 76, 5832. (j) Mukaiyama,
T.; Hata, E.; Yamada, T. Chem. Lett. 1995, 505. (k) Suzuki,
H.; Mori, T. J. Org. Chem. 1997, 62, 6498. (l) Campos, P. J.;
Garcıa, B.; Rodrıguez, M. A. Tetrahedron Lett. 2000, 41,
979. (m) Sy, W. W.; By, A. W. Tetrahedron Lett. 1985, 26,
1193. (n) Jovel, I.; Prateeptongkum, S.; Jackstell, R.; Vogl,
N.; Weckbecker, C.; Beller, M. Adv. Synth. Catal. 2008, 350,
2493. (o) Yan, G.; Borah, A. J.; Wang, L. Org. Biomol.
Chem. 2014, DOI: 10.1039/C4OB00573B.
(4) (a) Henry, L. C. R. Seances Acad. Sci., Ser. C 1895, 120,
1265. (b) Henry, L. Bull. Soc. Chim. Fr. 1895, 13, 999.
(c) Fioravanti, S.; Pellacani, L.; Tardella, P. A.; Vergari, M.
C. Org. Lett. 2008, 10, 1449. (d) Ballini, R.; Bosica, G.
J. Org. Chem. 1997, 62, 425. (e) Shulgin, A. T. J. Med.
Chem. 1966, 9, 445.
(5) (a) Horváth, I. T.; Anastas, P. T. Chem. Rev. 2007, 107,
2169. (b) Horváth, I. T. Chem. Rev. 1995, 95, 1. (c) Yücel,
A. S. Egit. Arast. 2008, 32, 145.
NO2
NO2
15
65
16
80
82
Cl
Cl
NO2
17
O2N
O2N
a The reactions were performed on a 1-mmol scale.
b All the products were characterized by 1H and 13C NMR spectrosco-
py and by GC/MS.
c Yield of pure product after column chromatography.
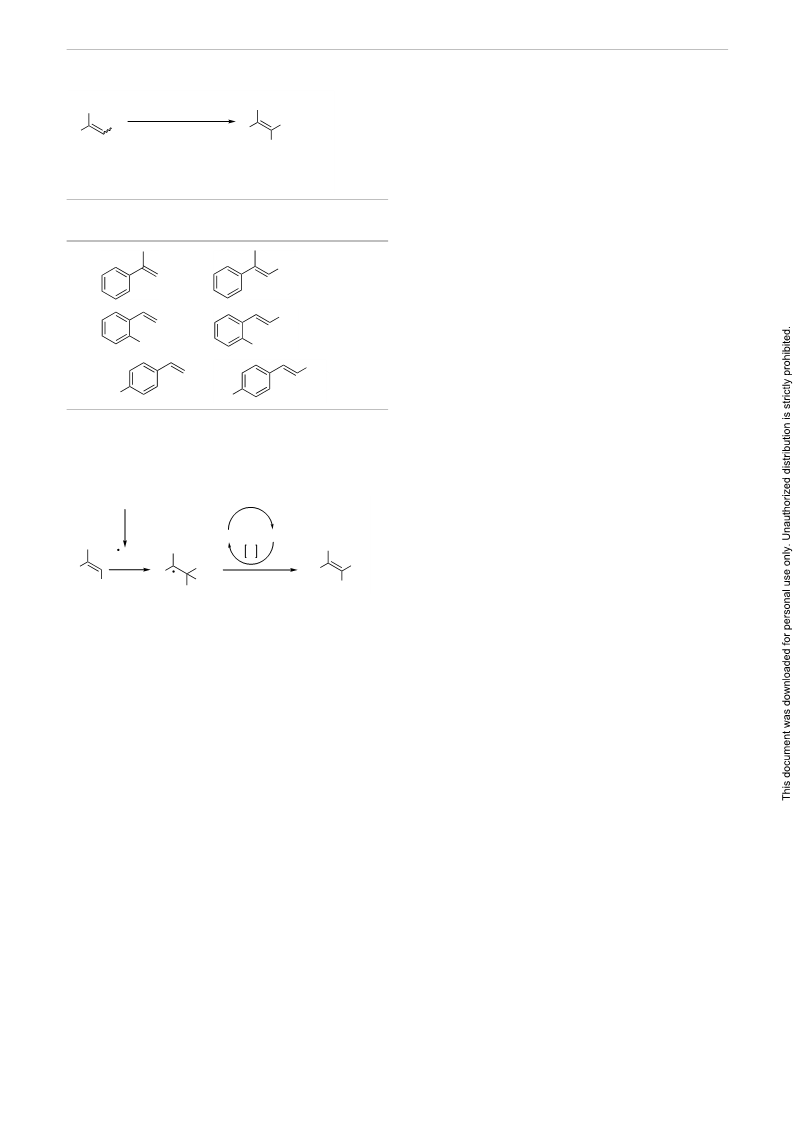
Claycop
heat
TEMPO TEMPOH
R2
R2
R2
O
NO2
NO2
NO2
R3
R1
R1
R1
R3
R3
H
Scheme 2 A plausible mechanism for TEMPO-catalyzed nitration of
aromatic olefins
(6) (a) Preparative Chemistry Using Supported Reagents;
Laszlo, P., Ed.; Academic Press: San Diego, 1987, 2nd ed.
(b) McKillop, A.; Young, D. W. Synthesis 1979, 401.
(c) McKillop, A.; Young, D. W. Synthesis 1979, 481.
(7) (a) Varma, R. S. Tetrahedron 2002, 58, 1235. (b) Posner, G.
H. Angew. Chem. Int. Ed. 1978, 17, 487. (c) Clark, H. P.;
Kybett, A. P.; Macquarie, D. J. Supported Reagents:
Preparation, Analysis and Applications; VCH: New York,
1992.
(8) Laszlo, P.; Pennetreau, P. J. Org. Chem. 1987, 52, 2407.
(9) (a) Varma, R. S.; Naicker, K. P.; Liesen, P. J. Tetrahedron
Lett. 1998, 39, 3977. (b) Pérez, C.; Pérez-Gutiérrez, S.;
Gómez, S. A.; Fuentes, G. A.; Zavala, M. A. Org. Prep.
Proced. Int. 2005, 37, 387. (c) Mohammed, S.; Padala, A.
K.; Dar, B. A.; Singh, B.; Sreedhar, B.; Vishwakarma, R. A.;
Bharate, S. B. Tetrahedron 2012, 68, 8156. (d) Mallouk, S.;
Bougrin, K.; Doua, H.; Benhida, R.; Soufiaoui, M.
Tetrahedron Lett. 2004, 45, 4143. (e) Meshram, H. M.;
Thakur, P. B.; Madhu Babu, B.; Bangade, V. M.
Acknowledgment
E.B. thanks DST New Delhi for an INSPIRE Fellowship (IFA12-
CH-29). C.R.S. and U.N.R thank OSDD and UGC, New Delhi, re-
spectively, for research fellowships. The authors thank CSIR, New
Delhi for financial support as part of the XII Five-Year Plan under
the title ORIGIN (CSC0108).
References
(1) (a) Lu, L.-Q.; Chen, J.-R.; Xiao, W.-J. Acc. Chem. Res.
2012, 45, 1278. (b) Reddy, M. A.; Jain, N.; Yada, D.;
Kishore, C.; Reddy, V. J.; Reddy, P. S.; Addlagatta, A.;
Kalivendi, S. V.; Sreedhar, B. J. Med. Chem. 2011, 54, 6751.
(c) Uehara, H.; Imashiro, R.; Hernández-Torres, G.; Barbas,
C. F. III. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 20672.
(d) Barrett, A. G. M.; Graboski, G. G. Chem. Rev. 1986, 86,
751. (e) Kaap, S.; Quentin, I.; Tamiru, D.; Shaheen, M.;
Eger, K.; Steinfelder, H. J. Biochem. Pharmacol. 2003, 65,
603. (f) Meah, Y.; Massey, V. Proc. Natl. Acad. Sci. U. S. A.
2000, 97, 10733.
Tetrahedron Lett. 2012, 53, 1780. (f) Varma, R. S.
Tetrahedron 2002, 58, 1235.
(10) (a) Cornelis, A.; Laszlo, P.; Pennetreau, P. J. Org. Chem.
1983, 48, 4772. (b) Gigante, B.; Prazeres, A. O. Marcelo-
Curto M. J. 1995, 60, 3445.
(2) (a) Kabalka, G. W.; Varma, R. S. Org. Prep. Proced. Int.
1987, 19, 283. (b) Ono, N. In Nitro Compounds: Recent
Advances in Synthesis and Chemistry; Feuer, H.; Nielsen,
© Georg Thieme Verlag Stuttgart · New York
Synlett 2014, 25, 1997–2000

 Begari, Eeshwaraiah
Begari, Eeshwaraiah
 Singh, Chandani
Singh, Chandani
 Nookaraju
Nookaraju
 Kumar, Pradeep
Kumar, Pradeep