.
Angewandte
Communications
Figure 3. H O physisorption isotherms (298 K) for the isoreticular
2
[
Ni (L5-R) ] series and the activated carbon Blꢅcher-101408. Solid
Figure 2. N physisorption isotherms (77 K) for the isoreticular
8 6
2
symbols denote adsorption, open symbols desorption.
[Ni
8
(L)
6
] series. Solid symbols denote adsorption, open symbols
À1
desorption (PP0 =partial pressure).
and P/P > 0.6 for [Ni (L5) ]) and augment even further upon
o
8
6
its functionalized analogs, show type I isotherms, character-
istic of microporous crystalline solids. The L5-based species
feature pores in the mesopore range (2.4 nm wide); therefore,
inclusion of hydrophobic substituents on the benzene ring of
H L5 (namely P/P > 0.65 for [Ni (L5-CH ) ], and P/P > 0.80
2
o
8
3
6
o
for [Ni (L5-CF ) ]), which is indicative of the increasing
8
3 6
N uptake in the low-pressure region is not as steep as for the
hydrophobicity of the frameworks (Figure 3). The relevance
of these results is further evidenced if compared to those
obtained with prototypical mesoporous MOFs such as MIL-
100(Fe) and MIL-101(Cr). Indeed, [Ni (L5) ] is more hydro-
2
other compounds. The [Ni (L5-R) ] (R = CH , CF ) systems,
8
6
3
3
sharing very similar unit cell parameters, exhibit almost the
same uptake of N (Figure S3) with just minor differences,
2
8
6
[
21]
suggesting that ligand functionalization does not affect the
accessibility of this probe molecule to the porous network,
even if it significantly modifies the surface polarity.
phobic than both MIL-100(Fe) and MIL-101(Cr), even if it
is slightly less hydrophobic than Blꢁcher-101408, since water
vapor enters the structure of the latter only at relative
pressures higher than 0.6. Remarkably, while the introduction
of methyl groups, as in [Ni (L5-CH ) ], slightly enhances the
As mentioned above, stability towards hydrolysis is
a major requirement for practical applications. On the other
hand, surface hydrophobicity is also advantageous for appli-
cations that involve capture of organic molecules in moist
environments. The enhanced stability provided by metal–
pyrazolate coordinative bonds can be exemplified by the
sensitivity to water of the materials containing mixed
pyrazolate/carboxylate-based ligands ([Ni (L1) ] and
8
3 6
hydrophobicity, the functionalization with fluorinated resi-
dues, as in [Ni (L5-CF ) ], gives rise to a highly hydrophobic
8
3 6
material, which clearly outperforms Blꢁcher-101408. The low
affinity of [Ni (L5-CF ) ] to moisture is exemplified also by
8
3 6
the narrowing of the hysteresis loop, which is in agreement
with the narrow pore distribution and is also indicative of easy
desorption of the adsorbed moisture. The latter feature
further highlights the potential relevance of this MOF under
operative humid conditions. In this regard, although Omary
8
6
[
Ni (L2) ]): after soaking them in water at room temperature
8 6
for 24 h, a few minor changes are observed in their XRPD
patterns (Figure S4); at variance, all the species containing
bipyrazolate linkers remain completely unaltered. Measure-
ment of cycles of water adsorption–desorption isotherms at
[5]
and co-workers have shown that the introduction of
fluoroalkane residues in a MOF gives rise to a highly
hydrophobic material, no report on the systematic variation
of the pore size and functionalization to modulate the surface
hydrophobicity of the pore can be found in the literature.
The hydrophobic nature of the bipyrazolate [Ni (L) ]
2
98 K is a highly valuable tool to evaluate both stability
toward hydrolysis and pore surface hydrophobicity of
[21]
MOFs.
Consequently, all the [Ni (L) ] materials have
8 6
been studied in this regard (Figure 3 and Figure S5). The
behavior of the [Ni (L) ] species has been compared to that of
8
6
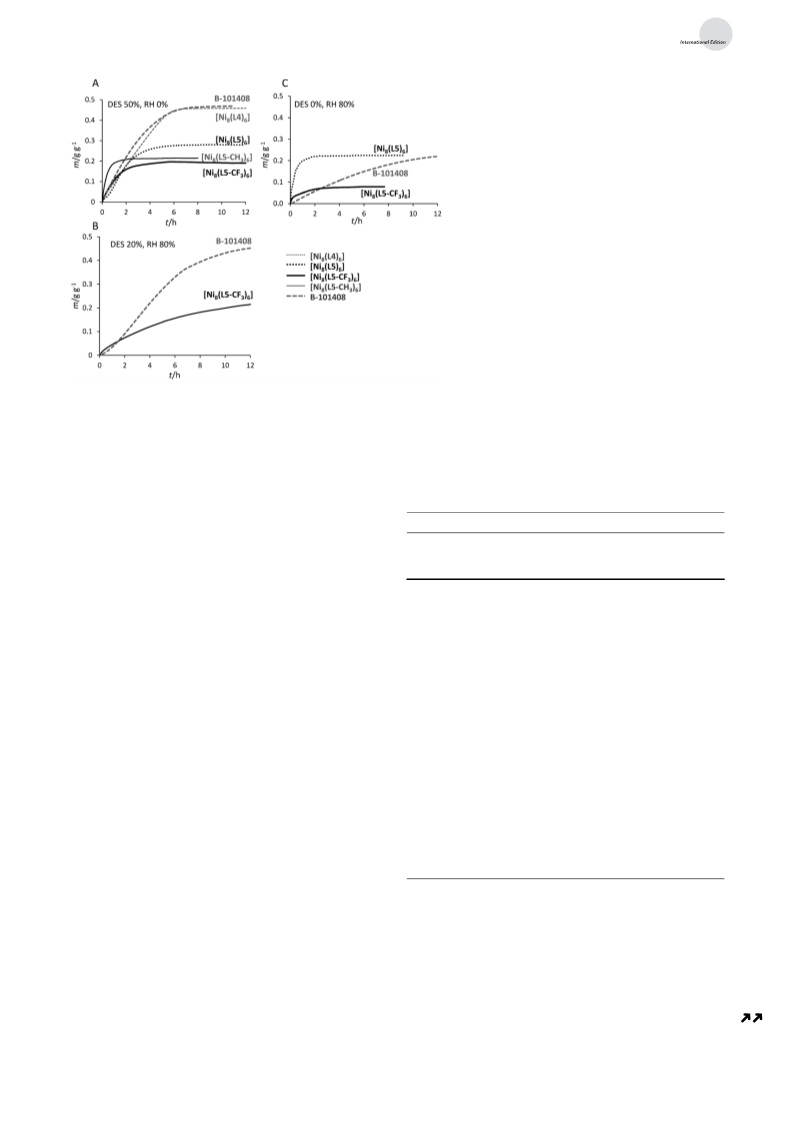
systems prompted us to study their possible use for air
purification processes, in which moisture is unavoidably
present and acts as a strong competitor with the adsorbates
of interest. Consequently, we have investigated the ability of
the activated [Ni (L) ] systems to capture hydrophobic,
8
6
the highly hydrophobic, commercially available, activated
carbon Blꢁcher-101408 (provided by Blꢁcher GmbH), the
active species employed in the state-of-the-art Saratoga
[
22]
filtering systems. The comparison shows that the maximum
amount of adsorbed water is in line with the pore size and cell
volume (see Figure 3 and Figure S5). Noteworthy, while the
mixed carboxylate/pyrazolate [Ni (L1) ] and [Ni (L2) ] sys-
8
6
harmful VOCs under dynamic conditions, in the presence
and absence of moisture. As a case of study, we have essayed
the performance of these materials for the capture of
diethylsulfide (DES, a model of mustard gas, already used
8
6
8
6
tems are sensitive to moisture, those containing bipyrazolate
linkers exhibit a low water affinity as well as a high stability in
environmental moisture. Indeed, partial pressures corre-
sponding to the beginning of water vapor condensation into
the porous frameworks increase upon extension of the spacer
[
23]
by us in previous studies; Scheme 2) from dry and humid
Ar/N streams. The experiments have been carried out by
2
checking the dynamic weight increase of the activated
bipyrazolate [Ni (L) ] systems as well as of the Blꢁcher-
8
6
(
namely P/P > 0.3 for [Ni (L3) ], P/P > 0.5 for [Ni (L4) ],
101408 reference material, at room temperature, upon
o
8
6
o
8
6
4
ꢁ 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2013, 52, 1 – 6
These are not the final page numbers!

 Padial, Natalia M.
Padial, Natalia M.
 Quartapelle Procopio, Elsa
Quartapelle Procopio, Elsa
 Montoro, Carmen
Montoro, Carmen
 Lopez, Elena
Lopez, Elena
 Oltra, J. Enrique
Oltra, J. Enrique
 Colombo, Valentina
Colombo, Valentina
 Maspero, Angelo
Maspero, Angelo
 Masciocchi, Norberto
Masciocchi, Norberto
 Galli, Simona
Galli, Simona
 Senkovska, Irena
Senkovska, Irena
 Kaskel, Stefan
Kaskel, Stefan
 Barea, Elisa
Barea, Elisa
 Navarro, Jorge A. R.
Navarro, Jorge A. R.