
Journal of Organic Chemistry p. 8380 - 8391 (2019)
Update date:2022-08-10
Topics:
 Fujita, Hikaru
Fujita, Hikaru
 Nishikawa, Riho
Nishikawa, Riho
 Sasamoto, Ozora
Sasamoto, Ozora
 Kitamura, Masanori
Kitamura, Masanori
 Kunishima, Munetaka
Kunishima, Munetaka
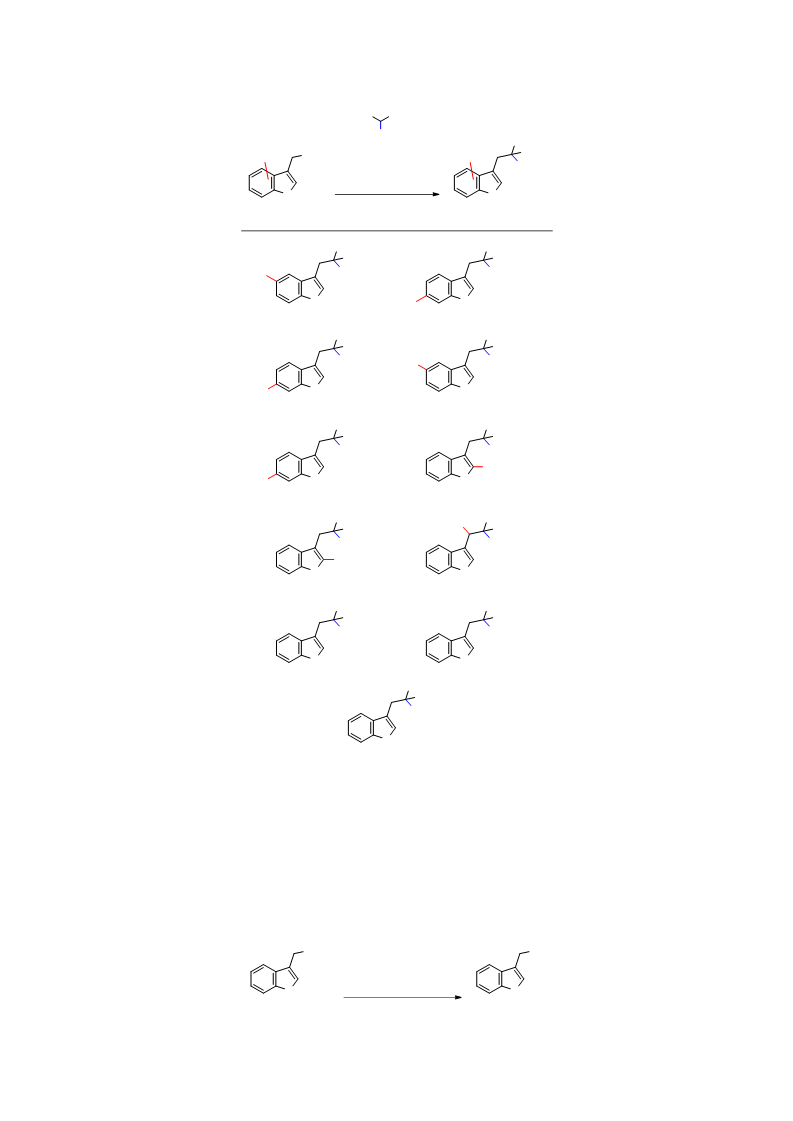
A new method for the substitution of 3-[(dimethylamino)methyl]indoles (gramines) with malonate-based nucleophiles was developed using 2-chloro-4,6-dimethoxy-1,3,5-triazine (CDMT) as the activating agent for the dimethylamino group. The reaction was completed in 1.5-6 h at room temperature in the presence of a tert-amine base and lithium salt. CDMT afforded superior results to methyl iodide, a common activating agent for the dimethylamino group in Mannich bases, particularly in the reactions of 1-substituted gramines. The reactivity of the possible intermediates, bis(indol-3-ylmethyl)dimethylammonium salts, was examined to obtain mechanistic insights on the reaction. This substitution method with CDMT enabled the sequential transformation of gramines: substitution with (N-alkylidene)aminomalonates followed by the Pictet-Spengler reaction under acidic conditions afforded 1,2,3,4-tetrahydro-β-carboline derivatives in one pot.
View More










zhuzhou zhongle chemical co. ltd.
Contact:+86-0731 28228409
Address:Zhuzhou, Hunan, China
Zhejiang Hoshine Silicon Industry Co., Ltd.
Contact:86-573-89179966
Address:Zhapu Town, Pinghu City, Zhejiang, China
Tianjin Pharmacn Medical Technology Co.,Ltd.
Contact:86-22-60122566ext.866(English),23359620
Address:Green Industrial Base, 6 Haitaifazhan Sixth Rd., Huayuan Industrial Area, Tianjin, 300384, China
Shanghai Pinewood Fine Chemical Co., Ltd.
website:http://www.pinewoodchem.com
Contact:0086-21-62417129,62414096
Address:Suite B, 27F, No.2, Lane 600, Tianshan Road, Shanghai
NINGXIA DARONG CHEMICALS & METALLURGY CO.,LTD.
Contact:86-952-2179751
Address:Darong Road, Dawukou, Shizuishan, Ningxia 753001, China
Doi:10.1016/S0040-4039(01)91834-9
(1974)Doi:10.1016/0022-1902(75)80533-1
(1975)Doi:10.1021/acsmedchemlett.7b00188
(2017)Doi:10.1039/c2dt31844j
(2013)Doi:10.1016/0040-4039(95)02048-9
(1995)Doi:10.1002/anie.201612225
(2017)