Journal of Natural Products
Article
Octadecyl ferulate (7): white, amorphous solid, yield 60%; VLC
mobile phase: toluene−ethyl acetate (9.5:0.5); H NMR (500 MHz,
standard ACSF at a constant flow rate of ∼2 mL/min. All recordings
were performed at room temperature.
1
GABAergic-evoked postsynaptic currents were recorded with an
Axopatch 200-B amplifier, filtered at 2 kHz, and digitized at 5 kHz.
Recording pipets were prepared from borosilicate capillaries with an
internal filament using a Fleming Brown micropipet puller (Molecular
Devices, Novato, CA, USA). Resistance of the pipets ranged from 4.5
to 6.0 MΩ when they were filled with, in mM, 140 CsCl, 2 MgCl, 2
CaCl, 10 EGTA, 10 HEPES, 2 ATP-Na, pH 7.3 with 5 N CsOH.
Only recordings with access resistance of <25 MΩ (the values usually
ranged from 9 to 20 MΩ) were analyzed. Series resistance was not
compensated, and cells were excluded from a further analysis if access
resistance changed by >20% during the course of the recording. A
bipolar concentric stimulating electrode was placed near the granule
neurons’ cell bodies in order to evoke the depolarization of
presynaptic terminals and thus the GABA release from nearby
GABAergic interneurons. The nonselective glutamatergic receptor
antagonist kynurenic acid (3 mM) was added to the solution in order
to exclude excitatory currents when granule cells were clamped at −65
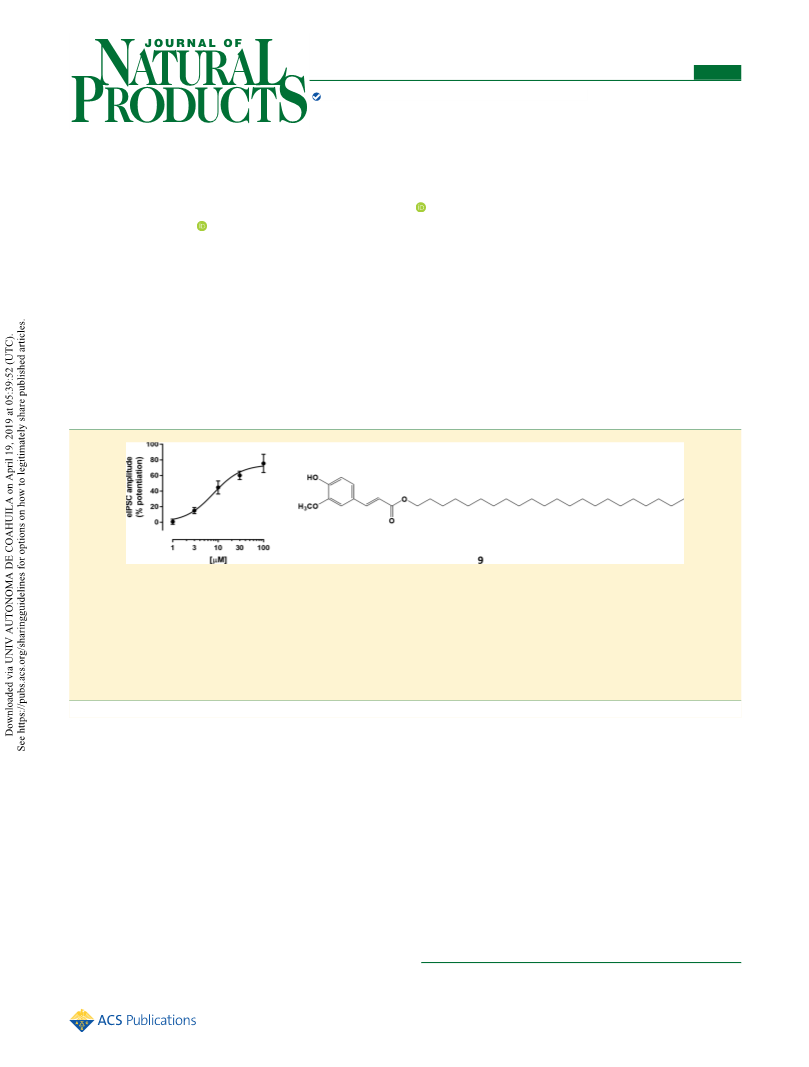
mV. The variation of the IPSC amplitude before and after drug
perfusion was considered as the parameter for evaluating the drug
effect on GABAAR activity.
CDCl3) δH 7.61 (1H, d, J = 16 Hz, H-3), 7.07 (1H, dd, J = 2, 8 Hz, H-
6′), 7.03 (1H, d, J = 2 Hz, H-2′), 6.91 (1H, d, J = 8 Hz, H-5′), 6.29
(1H, d, J = 16 Hz, H-2), 5.94 (1H, s, OH), 4.19 (2H, t, J = 6.8 Hz, H-
1″), 3.92 (3H, s, OCH3-3′), 1.69 (2H, m, H-2″), 1.40 (2H, m, H-3′′),
1.26 (28H, m, H-4′′−H-17′′), 0.88 (3H, t, J = 6.8, Hz, H-18″); 13C
NMR (100 MHz, CDCl3) δC 167.3 (C-1), 147.9 (C-3′), 146.8 (C-
4′), 144.6 (C-3), 127.1 (C-1′), 123.0 (C-6′), 115.7 (C-2), 114.7 (C-
5′), 109.3 (C-2′), 64.6 (C-1″), 55.9 (OCH3), 31.9 (C-16″), 29.7,
29.6, 29.5 (C-5′′−C-14′′), 29.4 (C-15′′), 29.3 (C-4′′), 28.8 (C-2′′),
26.0 (C-3′′), 22.7 (C-17′′), 14.1 (C-18′′); ESIMS m/z (negative
mode) 445 [M − H]−, 487 [M + CH3CN]−.
Eicosanyl ferulate (8): white, amorphous solid; yield 75%; VLC
1
mobile phase: toluene−ethyl acetate (9.5:0.5); H NMR (500 MHz,
CDCl3) δH 7.61 (1H, d, J = 16 Hz, H-3), 7.07 (1H, dd, J = 2, 8.5 Hz,
H-6′), 7.03 (1H, d, J = 2 Hz, H-2′), 6.91 (1H, d, J = 8.5 Hz, H-5′),
6.29 (1H, d, J = 16 Hz, H-2), 4.19 (2H, t, J = 6.8 Hz, H-1″), 3.92
(3H, s, OCH3-3′), 1.69 (2H, m, H-2″), 1.40 (2H, m, H-3′′), 1.26
(32H, m, H-4′′−H-19′′), 0.88 (3H, t, J = 6.5, Hz, H-20″); 13C NMR
(100 MHz, CDCl3) δC 167.3 (C-1), 147.9 (C-3′), 146.8 (C-4′), 144.6
(C-3), 127.1 (C-1′), 123.0 (C-6′), 115.7 (C-2), 114.7 (C-5′), 109.3
(C-2′), 64.6 (C-1″), 56.0 (OCH3), 31.9 (C-18″), 29.7, 29.6, 29.5 (C-
5′′− C-16′′), 29.4 (C-17′′), 29.3 (C-4′′), 28.8 (C-2′′), 26.0 (C-3′′),
22.7 (C-19′′), 14.1 (C-20′′); ESIMS m/z (negative mode) 473 [M −
H]−, 515 [M + CH3CN]−.
Statistical Analysis. Data are presented as means
standard
error of the mean (SEM) and were compared by one-way analysis of
variance (ANOVA) followed by Bonferroni’s post hoc test, or
Student’s t test with the use of Prism software (version 6, Graphpad).
A p value of <0.05 was considered statistically significant.
Ligand Preparation and Qikprop Analysis. Compound 9 was
prepared with Maestro GUI34 and subject to conformational search
by means of MacroModel program version 9.2.35 Merck molecular
force fields36 were applied, and the implicit solvation model
generalized Born/surface area was used.37 Therefore, the compound
geometry was energy minimized using the Polak-Ribier conjugate
gradient method, 10 000 iterations, and a convergence criterion of
0.01 kcal/(mol Å). Conformational searching was performed using
the MCMM method, allowing 5000 steps. The compound global
Docosanyl ferulate (9): white, amorphous solid; yield 69%; VLC
1
mobile phase: toluene−ethyl acetate (9.5:0.5); H NMR (500 MHz,
CDCl3) δH 7.61 (1H, d, J = 16 Hz, H-3), 7.07 (1H, dd, J = 2, 8.5 Hz,
H-6′), 7.03 (1H, d, J = 2 Hz, H-2′), 6.91 (1H, d, J = 8.5 Hz, H-5′),
6.29 (1H, d, J = 16 Hz, H-2), 4.19 (2H, t, J = 6.8 Hz, H-1″), 3.93
(3H, s, OCH3-3′), 1.70 (2H, m, H-2″), 1.40 (2H, m, H-3′′), 1.26
(32H, m, H-4′′−H-21′′), 0.88 (3H, t, J = 6.5, Hz, H-22″); 13C NMR
(100 MHz, CDCl3) δC 167.3 (C-1), 148 (C-3′), 146.8 (C-4′), 144.6
(C-3), 127.1 (C-1′), 123.0 (C-6′), 115.7 (C-2), 114.7 (C-5′), 109.3
(C-2′), 64.6 (C-1″), 55.9 (OCH3), 31.9 (C-20″), 29.7, 29.6, 29.5 (C-
5′′−C-18′′), 29.4 (C-19′′), 29.3 (C-4′′), 28.8 (C-2′′), 26.0 (C-3′′),
22.7 (C-21′′), 14.1 (C-22′′); ESIMS m/z (negative mode) 501 [M −
H]−, 543 [M + CH3CN]−.
minimum conformation was considered for QikProp analysis
38
̈
(QikProp, Schrodinger, LLC, New York, NY, USA).
Electrophysiology Experiments. Male Sprague−Dawley rats
ASSOCIATED CONTENT
■
(Charles River, Italy), weighing 125−155 g, were maintained in
S
* Supporting Information
controlled environmental conditions (temperature 22
2 °C and
The Supporting Information is available free of charge on the
humidity 60−65%), under a 12 h light/12 h dark cycle. All
experiments were conducted in conformity with the regulations of
the Committee for the Protection and Use of Animals of the
University of Cagliari, in accordance with current Italian legislation on
animal experimentation (D.L. 26/2014) and the European directives
(2010/63/EU) on care and use of laboratory animals. In particular,
this study was approved by the Organization for Animal Care of the
University of Cagliari (OPBA-UniCA) and performed in accordance
with the Ministry of Health authorization number 1177/2016-pr
(December 15, 2016). Furthermore, every effort was made to
minimize the number of animals used. Coronal brain slices containing
the hippocampus were prepared as previously described.33 Briefly,
animals were subjected to deep anesthesia with isoflurane 2−5% and
decapitated. Their brain was rapidly removed from the skull and
transferred to a modified artificial cerebrospinal fluid (ACSF)
containing (in mM) 220 sucrose, 2 KCl, 0.2 CaCl2, 6 MgSO4, 26
NaHCO3, 1.3 NaH2PO4, and 10 D-glucose (pH 7.4, set by aeration
with 95% O2 and 5% CO2). Coronal brain slices (thickness, 260 μm)
containing the dentate gyrus of the hippocampus were cut in ice-cold
modified ACSF with the use of a Leica VT1200S vibratome (Leica,
Heidelberg, Germany). Slices were then transferred immediately to a
nylon net submerged in standard ACSF containing (in mM) 126
NaCl, 3 KCl, 2 CaCl2, 1 MgCl2, 26 NaHCO3, 1.25 NaH2PO4, and 10
D-glucose (pH 7.4, set by aeration with 95% O2/5% CO2) for at least
40 min at a controlled temperature of 35 °C. After subsequent
incubation for at least 1 h at room temperature, hemislices were
transferred to the recording chamber and continuously perfused with
Additional information (PDF)
AUTHOR INFORMATION
■
Corresponding Author
*Tel: + 39-706758979. Fax: + 39-706758553. E-mail:
ORCID
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
This study was financially supported by Regione Autonoma
della Sardegna (RAS) (CRP26805 CUP B71J11001480002).
The authors wish to thank Dr. A. Agarwal (Natural Remedies
Pvt Ltd. Bangalore, India) for the generous gift of W. somnifera
root extract.
G
J. Nat. Prod. XXXX, XXX, XXX−XXX

 Sonar, Vijay P.
Sonar, Vijay P.
 Fois, Benedetta
Fois, Benedetta
 Distinto, Simona
Distinto, Simona
 Maccioni, Elias
Maccioni, Elias
 Meleddu, Rita
Meleddu, Rita
 Cottiglia, Filippo
Cottiglia, Filippo
 Acquas, Elio
Acquas, Elio
 Kasture, Sanjay
Kasture, Sanjay
 Floris, Costantino
Floris, Costantino
 Colombo, Daniele
Colombo, Daniele
 Sissi, Claudia
Sissi, Claudia
 Sanna, Enrico
Sanna, Enrico
 Talani, Giuseppe
Talani, Giuseppe