M. Nasrollahzadeh et al. / Materials Research Bulletin 68 (2015) 150–154
151
highly active. We have recently developed synthesis of various
metallic and bimetallic nanoparticles (NPs) [6–15] which promote
various catalytic transformations under heterogeneous conditions.
Immobilization of metallic nanoparticles on solid supports is one
of the best methods to improve the efficiency and recovery of
catalysts. To prevent the agglomeration of Pd NPs, several materials
The current in the discharge process is maintained at 15 A. As the
rods are brought close together, discharge occurs resulting in the
formation of plasma. As more copper was ablated from the anode,
the plasma expands, pushing the liquid away, and a gaseous bubble
forms. Melted species can react with plasma and then condense
into the liquid. It is worth noting that during the process, as the
anode is consumed, the rods are kept at a constant distant from
each other of about 1–2 mm. The Cu nanoparticles solution was
3 4 2
such as polymer, Fe O , CuO, TiO , perlite have been used as a
support for metal NPs [6–8,10,11,16]. Among various metal oxide
nanoparticles, CuO NPs have received considerable attention
because of their unusual properties and used widely as efficient
support and catalysts in organic reactions due to their high
catalytic activity, ease of handling, reusability, and benign
character [17,18]. CuO NPs, in particular, being cheap, are
less harmful to the environment than any other metals in
cross-coupling reactions [8,18]. As a result, we decided to design
and prepare the Pd/CuO NPs and explore their catalytic ability for
the phosphine-free Heck coupling reaction under aerobic con-
ditions. This method provides an improvement on our previous
reported protocols for the phosphine-free Heck coupling reaction
added to a solution containing PdCl
dissolving 0.02 g (0.005 or 0.01 g) of PdCl
2
prepared by ultrasonic
2
powder (5 N), 99.9 mL DI
ꢀ
water and 0.1 mL HCl. Then, samples were heated at 90 C for 1 h. In
order to extract the dispersed nanoparticles, the solution was dried
À1
at a pressure of 10 Torr and centrifuged several times and then
dispersed on a glass substrate.
2.3. General procedure for the photochemical Heck cross-coupling
reaction
A
mixture of styrene or olefin (1.5 mmol), aryl halide
[
8–10].
(1.0 mmol), Et
3
N (2.0 mmol), DMF (4.0 mL) and the catalyst
ꢀ
An advantage of the Pd/CuO NPs catalyst was that its synthesis
(3.0 mol%) was stirred in an oil bath at 110 C for 20 h. The
progress of the reaction was monitored by thin layer chromatog-
raphy (TLC) and on completion, the reaction mixture was cooled to
room temperature and the catalyst was separated from the
reaction mixture by centrifugation. The remaining solution was
diluted with water and the products were extracted using ether,
was very simple. In the present study, high-quality Cu nano-
particles were synthesized without the use of vacuum equipment.
The nanoparticles were obtained in the form of dispersed particles
in the water by high current electrical arc discharge between two
copper electrodes which were submerged in deionized (DI) water.
To the best of our knowledge, there are no reports on the
fabrication of copper nanoparticles by the electrical arc discharge
method. In general, electrical arc discharge in water has the
advantage in this regard as it produces self-crystallized nano-
particles due to the high temperature caused by Joule heating.
Moreover, compared with other techniques, electrical arc dis-
charge in water is an attractive method because of the simplicity of
the experimental set-up, the lack of complicated equipment, low
impurity and less production steps leading to a high-throughput
and cost-effective procedure to generate high yields of nano-
particles.
2 4
dried over Na SO and purified by using column chromatography.
All products are known in the literature and the physical data of the
products were found to be identical with those reported in the
literature [8–10,19,20].
3. Results and discussion
3.1. Characterization of catalyst
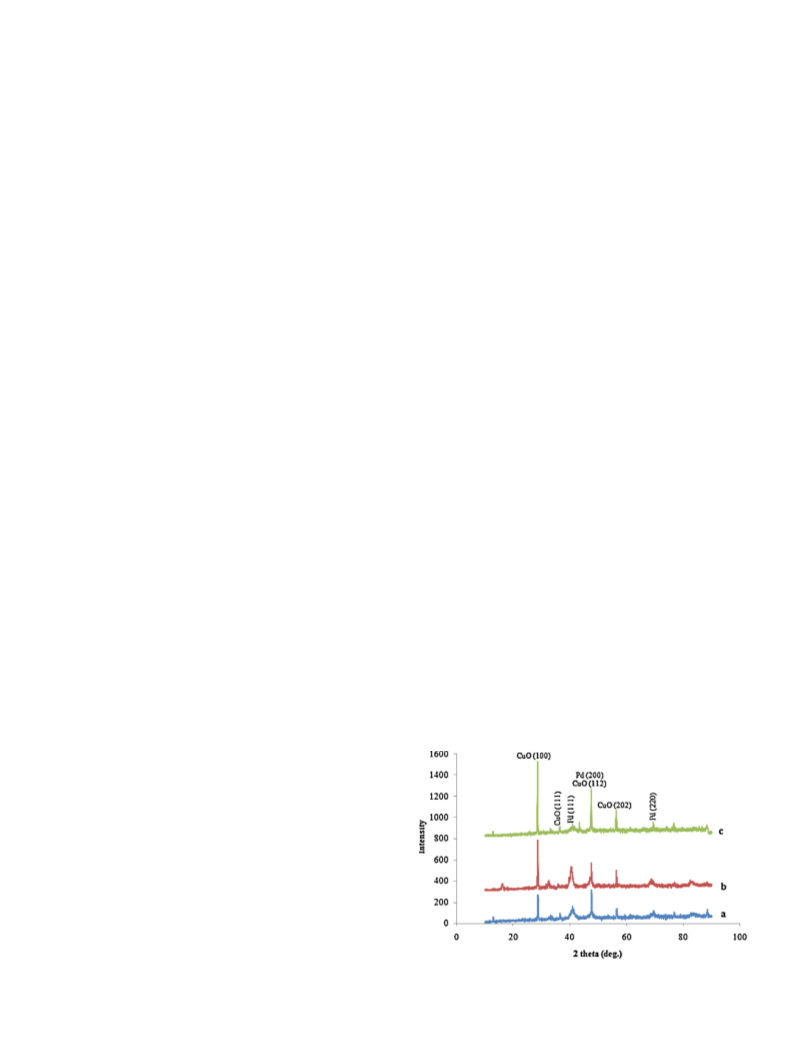
Pd/CuO NPs were characterized using the powder XRD,
FE-SEM and EDS. Fig. 1 shows XRD patterns of the three films
(
the PdCl
2
is equal to a = 0.005, b = 0.01, c = 0.02 g). Pd/CuO NPs
2. Experimental
exhibited Bragg diffractions corresponding to the cupric oxide
(
CuO) and palladium (Pd).
2.1. Instruments and reagents
FE-SEM, TEM and EDS were carried out for study of morphology,
size and composition of nanostructures. Typical FE-SEM and TEM
images of Pd/CuO NPs (PdCl = 0.02 g) are shown in Figs. 2 and 3,
High-purity chemical reagents were purchased from the Merck
2
and Aldrich chemical companies. All materials were of commercial
reagent grade. Melting points were determined in open capillaries
respectively. As it is clear, Pd/CuO NPs show morphology like
rectangular structures. According to FE-SEM analysis, the average
length, width and height of this rectangular are about 70, 70 and
250 nm, respectively. We used EDS to determine chemical
1
using a BUCHI 510 melting point apparatus and are uncorrected. H
13
NMR and C NMR spectra were recorded on a Bruker Avance
DRX-400 spectrometer at 400 and 100 MHz, respectively. FT-IR
spectra were recorded on a Nicolet 370 FT/IR spectrometer (Thermo
Nicolet, USA) using pressed KBr pellets. The element analyses (C, H,
N) were obtained from a Carlo ERBA Model EA 1108 analyzer carried
out on Perkin-Elmer 240C analyzer. X-ray diffraction (XRD)
measurements were carried out using a Philips powder diffractom-
eter type PW 1373 goniometer (Cu K
a
= 1.5406 Å). The scanning rate
ꢀ
ꢀ
was 2 /min in the 2
u
range from 10 to 80 . Scanning electron
microscopy (SEM) was performed on a Cam scan MV2300. EDS
using EDS (S3700N) was utilized for chemical analysis of prepared
nanostructures. Transmission electron microscopy (TEM) was
recorded on
a Philips EM208 microscope operating at an
accelerating voltage of 90 kV.
2.2. Preparation of Pd/CuO nanoparticles
The setup for synthesis of Cu comprises contains DC power
supply and a reactor including two electrodes and a glass chamber
filled by water. The cathode and anode are both pure copper rods.
Fig. 1. XRD patterns of the three films (the PdCl
c = 0.02 g).
2
is equal to a = 0.005, b = 0.01,

 Nasrollahzadeh, Mahmoud
Nasrollahzadeh, Mahmoud
 Sajadi, S. Mohammad
Sajadi, S. Mohammad
 Rostami-Vartooni, Akbar
Rostami-Vartooni, Akbar
 Azarian, Abbas
Azarian, Abbas