DOI: 10.1002/cctc.201600635
Communications
Boron Lewis Acid Promoted Ruthenium-Catalyzed
Hydrogenation of Amides: An Efficient Approach to
Secondary Amines
Ming-Lei Yuan,[a] Jian-Hua Xie,[a] and Qi-Lin Zhou*[a, b]
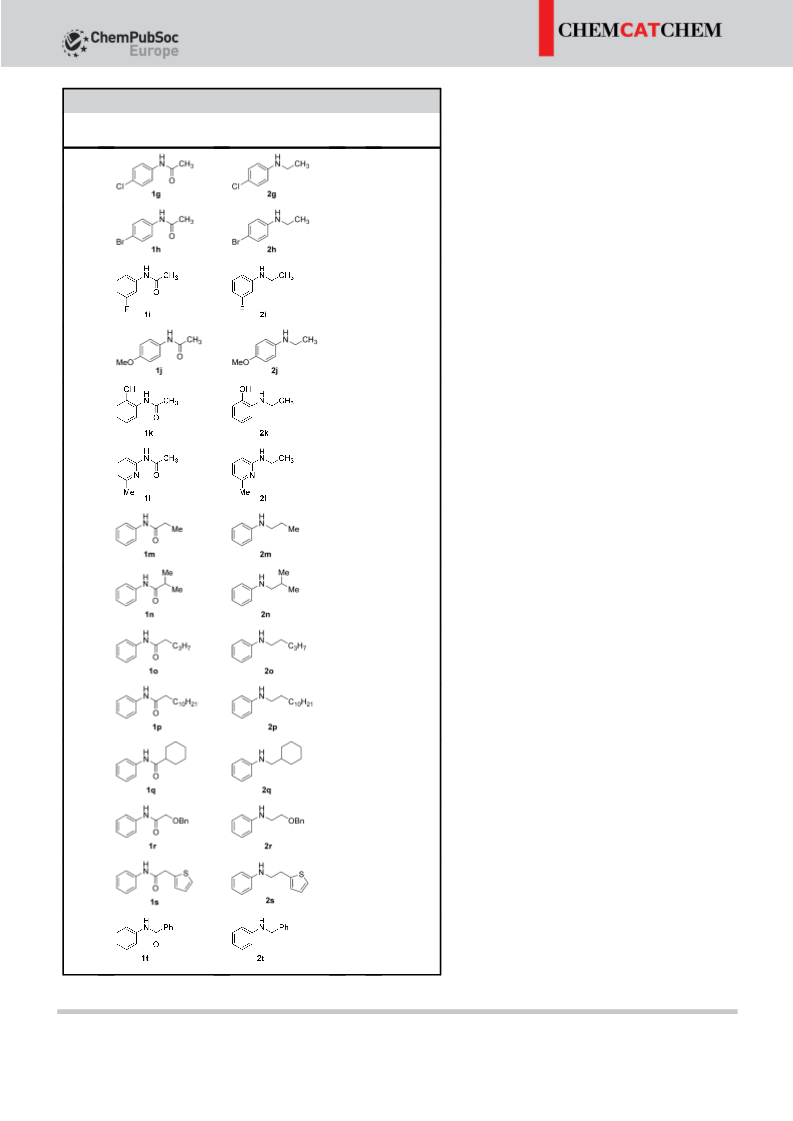
The hydrogenation of amides to amines has been developed
by using the catalyst [Ru(H)2(CO)(Triphos)] (Triphos=1,1,1-tri-
(diphenylphosphinomethyl)ethane) and catalytic boron Lewis
acids such as B(C6F5)3 or BF3·Et2O as additives. The reaction pro-
vides an efficient method for the preparation of secondary
amines from amides in good yields with high selectivity.
(1608C, 100 bar) are still needed.[6] In addition, with the Ru/Tri-
phos catalyst and expensive metal triflate Yb(OTf)3 as a co-cat-
alyst, Beller et al. realized the hydrogenation of a range of sec-
ondary and tertiary amides to the corresponding amines with
moderate to good selectivities under milder reaction condi-
tions (1508C, 5–15 bar).[7] Therefore, the development of highly
efficient and easily accessible catalysts for selective hydrogena-
tion of amides to the corresponding amines is highly desired.
As a part of our ongoing research on the development of ef-
ficient catalysts for the hydrogenation of carboxylic acid deriv-
atives under mild reaction conditions,[8] we have recently re-
ported an iridium catalyst with a P(O)C(O)P pincer ligand and
B(C6F5)3 as a Lewis acid for the hydrogenation of N-arylamides
and lactams to amines with excellent selectivity under relative-
ly mild conditions.[9] However, one equivalent of B(C6F5)3 was
Transition-metal-catalyzed hydrogenation of amides to amines,
which avoids the use of stoichiometric hydride reagents and
the generation of large amounts of waste, has received inten-
sive study in recent years.[1] The big challenge for this transfor-
mation is to find efficient catalysts that can selectively hydro-
genate amides to the corresponding amines without genera-
tion of alcohols and lower amines.[2] Several bifunctional/bi-
metallic heterogeneous catalysts have been devel-
oped for the hydrogenation of amides to amines
with high selectivity, albeit the substrate scope is
narrow and the reaction conditions are generally
harsh.[3] In contrast, homogeneous catalytic hydroge-
nation of amides can performed under milder condi-
tions, but most of the reported examples produced
a mixture of alcohols and lower amines.[4] A break-
through was made in 2007 by Cole-Hamilton et al.,
who introduced the ruthenium complex of the Tri-
phos ligand (1,1,1-tri(diphenylphosphinomethyl)-
ethane) into the hydrogenation of N-phenyl amide to
secondary amine with excellent selectivity, but rela-
tively harsh reaction conditions (1648C, 40 bar; or
2108C, 10 bar) are required.[5] Recently, Klankermayer
et al. modified the Triphos ligand and found that
ruthenium complexes of Xyl-Triphos ligands contain-
ing 3,5-dimethylphenyl groups have high activity and
selectivity for the hydrogenation of lactams to cyclic
amines in the presence of a catalytic amount of
Scheme 1. Catalysts for the homogeneous hydrogenation of amides to amines.
methanesulfonic acid as
a co-catalyst. However,
a high reaction temperature and high H2 pressure
required to activate the amide substrates in that reaction. In
the search for low-cost, efficient, and practical catalysts for the
hydrogenation of amides to amines, we found that catalytic
boron Lewis acids can promote the hydrogenation of amides
catalyzed by Ru/Triphos complexes. We herein report the
boron Lewis acid promoted Ru-catalyzed hydrogenation of
amides in excellent selectivity (Scheme 1).
[a] M.-L. Yuan, Prof. J.-H. Xie, Prof. Q.-L. Zhou
State Key Laboratory and Institute of Elemento-organic Chemistry
Nankai University, 94 Weijin Road, Tianjin (P.R. China)
[b] Prof. Q.-L. Zhou
Collaborative Innovation Center of Chemical Science and
Engineering
We initially evaluated a series of ruthenium catalysts in the
reduction of N-phenylacetamide (1a) to N-ethylaniline (2a) in
the presence of 10 mol% of B(C6F5)3. The [Ru(H)2(CO)(Triphos)]
Nankai University, 94 Weijin Road, Tianjin (P.R. China)
Supporting information for this article can be found under http://
ChemCatChem 2016, 8, 1 – 6
1
ꢀ 2016 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
&
These are not the final page numbers! ÞÞ

 Yuan, Ming-Lei
Yuan, Ming-Lei
 Xie, Jian-Hua
Xie, Jian-Hua
 Zhou, Qi-Lin
Zhou, Qi-Lin