V.D. Makhae6 et al. / Journal of Organometallic Chemistry 590 (1999) 222–226
223
of ca. 80 cm3. Steel balls 4.8, 12.3 and 14.3 mm in
diameter were used as activating filling.
mm in diameter; total weight, 150 g). A dark orange
reaction mixture was obtained. The weighed amount of
the reaction mixture (0.5750 g) was transferred into a
sublimation apparatus. Sublimation was carried out at
a temperature of external heating of 80–95°C in vac-
uum (0.1 mmHg). 1,1%-Dimethylferrocene was collected
on the cooled part of the sublimation apparatus as
orange crystals. The yield was 0.1420 g (90.4%). Anal.
Found, %: C, 67.0; H, 6.6; Fe, 25.7. For C12H14Fe
Calc., %: C, 67.3; H, 6.59; Fe, 26.09.
To carry out the reactions, a reactor was charged
with weighed amounts of starting materials and activat-
ing filling, hermetically closed, installed on a vibration
ball mill, and subjected to vibrations for scheduled
time. After completion of the process, the reactor was
opened, the reaction mixture was separated from acti-
vating filling and used for physicochemical analyses and
isolation of target products.
Isolation of metallocenes from the reaction mixtures
was carried out by vacuum sublimation or extraction
with benzene. The products obtained were identified
according to the data of chemical analyses and physico-
chemical investigations.
2.7. Preparation of nickelocene by reaction of NiCl2
and CpTl
A mixture of CpTl (0.8940 g, 3.2 mmol) and NiCl2
(0.2332 g, 1.8 mmol) was subjected to mechanical load-
ing for 60 min at a frequency of 12 Hz and an ampli-
tude of 11 mm (activating filling, steel balls 4.8 mm in
diameter; total weight, 150 g). A gray–green reaction
mixture was obtained. The weighed amount of the
reaction mixture (0.8086 g) was transferred into a subli-
mation apparatus. Sublimation was carried out at a
temperature of external heating of 100–120°C in vac-
uum (0.1 mm Hg). Nickelocene was obtained as dark-
green crystals in a yield of 0.198 g (89.5%). The
compound was characterized by physicochemical
methods.
2.4. Preparation of ferrocene by interaction of FeCl2
and CpTl
A mixture of CpTl (0.7356 g, 2.7 mmol) and FeCl2
(0.1922 g, 1.5 mmol) was subjected to mechanical load-
ing for 60 min at a frequency of 12 Hz and an ampli-
tude of 11 mm (activating filling, steel balls 4.8 mm in
diameter; total weight, 150 g). The reaction mixture
became orange. A portion of the reaction mixture
(0.5164 g) was transferred into a sublimation apparatus.
Sublimation was carried out at a temperature of exter-
nal heating of 120–140°C in vacuum (0.1 mmHg).
Ferrocene was obtained as orange crystals in a yield of
0.1179 g (84%).
2.8. Reaction of ferrocene with thallium chloride
Anal. Found, %: C, 65.2; H, 4.94; Fe, 30.15. For
C10H10Fe Calc., %: C, 64.56; H, 5.42; Fe, 30.02.
A mixture of TlCl (0.6020 g, 2.5 mmol) and ferrocene
(0.2334 g, 1.25 mmol) was subjected to mechanical
loading for 75 min (filling, steel balls 12.3 mm in
diameter; total weight, 150 g). The mixture obtained
smelled like cyclopentadiene, which testified to the de-
composition of ferrocene. A portion (0.7391 g) of the
reaction mixture was transferred into a sublimation
apparatus, and ferrocene was sublimed at a tempera-
ture of external heating of 120°C in vacuum. Ferrocene
was recovered in a yield of 0.1590 g, (68%). The in-
crease of temperature up to 160°C caused sublimation
of 0.003 g of pale-yellow cyclopentadienylthallium
(yield 0.4%), which was identified by its IR spectrum
and diffraction pattern.
2.5. Preparation of ferrocene by reaction of FeCl2 and
CpK
A mixture of CpK (0.3616 g, 3.47 mmol) and FeCl2
(0.1380 g, 1.04 mmol) was subjected to mechanical
loading for 15 min at a frequency of 12 Hz and an
amplitude of 11 mm (activating filling, steel balls 12.3
mm in diameter; total weight, 150 g). A dark orange
reaction mixture was obtained. A portion of the reac-
tion mixture (0.367 g) was transferred into a sublima-
tion apparatus. The sublimation was carried out at a
temperature of external heating of 120–140°C in vac-
uum (0.1 mmHg). Ferrocene was obtained as orange
crystals in a yield of 0.1725 g (85.2%) and characterized
by physicochemical methods.
3. Results and discussion
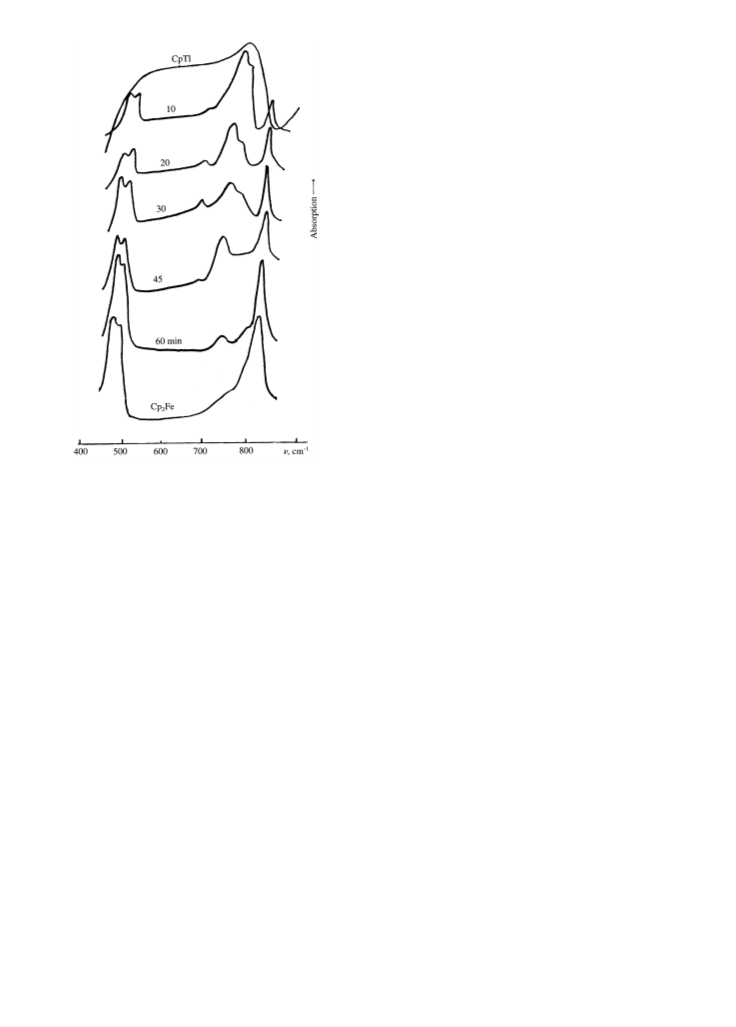
Mechanical loading (activation) of mixtures of
iron(II) chloride and cyclopentadienides of alkaline
metals using a vibration ball mill resulted in the forma-
tion of alkaline metal chloride and ferrocene [1]. The
study of the dependence of the yield of ferrocene on the
ratio of reagents in the reaction with CpK showed that
the yield of ferrocene increased with an increase in CpK
excess. Conversion of FeCl2 into ferrocene achieved
2.6. Preparation of 1,1%-dimethylferrocene by reaction
of FeCl2 and CH3C5H4Na
A mixture of CH3C5H4Na (0.4390 g, 4.25 mmol) and
FeCl2 (0.2538 g, 2.0 mmol) was subjected to mechanical
loading for 30 min at a frequency of 12 Hz and an
amplitude of 11 mm (activating filling, steel balls 4.8

 Makhaev
Makhaev
 Borisov
Borisov
 Petrova
Petrova