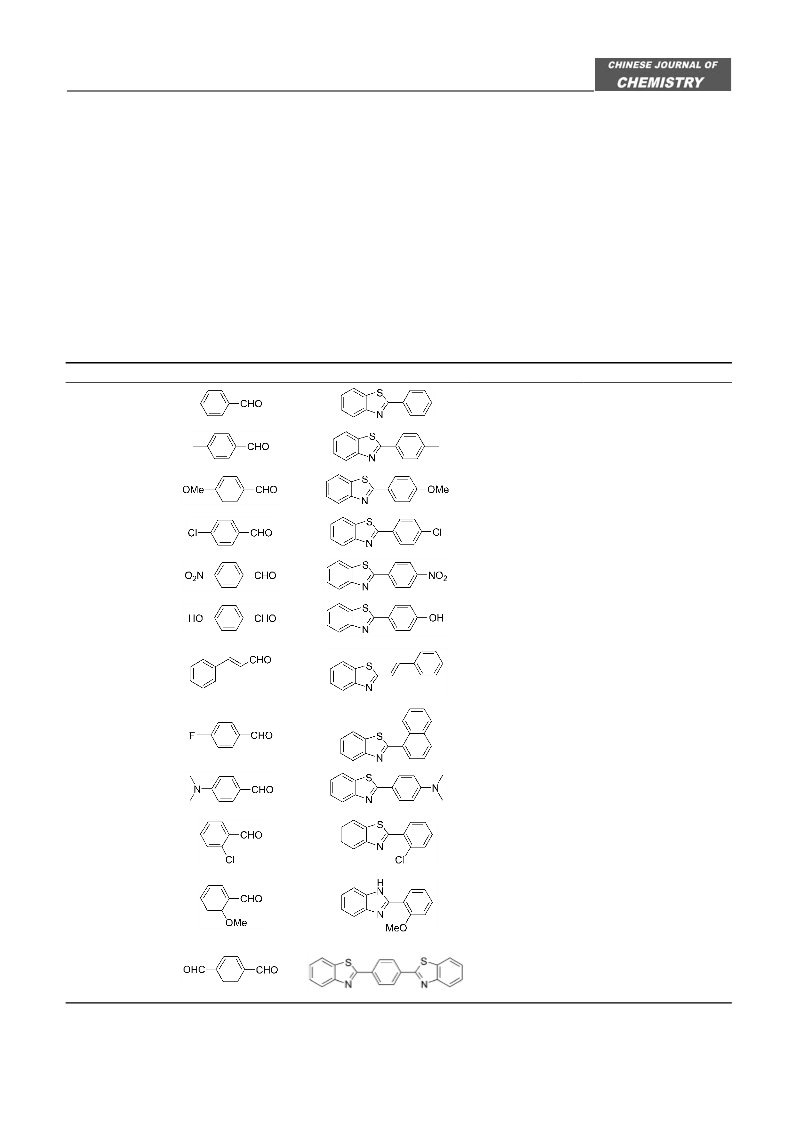
Application of N,N'-Diiodo-N,N'-1,2-ethandiylbis(p-toluene sulfonamide) as a New Reagent
A comparison of the efficiency of this method with
selected previous methods is collected in Table 3. The
results show that this method is superior to some previ-
ously reported methods in terms of yields and reaction
times.
After the reaction of NIBTS with [I+II], the sul-
phonamide is recovered and can be reused many times
without decreasing yield.
In conclusion, NIBTS was found to be mild and
effective new reagent for the convenient synthesis of
2-arylbenzimidazoles and 2-arylbenzothiazoles in ex-
cellent yields from 1,2-phenylenediamine and 2-amino-
thiophenol and a wide variety of aryl aldehydes under
solvent-free conditions. Moreover, the method has ad-
vantages in terms of product yields, absence of solvent,
short reaction times, non-corrosive, operational simplic-
ity, operational simplicity (easy work up of reactions).
Since NIBTS contain iodine atoms which are at-
tached to nitrogen atoms, it is very possible that they
+
release I in situ which can act as catalyst and oxidant
in the reaction medium. Therefore, the mechanism
shown in Scheme 2 can be suggested for the formation
1
2,14
of benzimidazoles.
Scheme 2
Acknowledgments
We are thankful to Bu-Ali Sina University, Payame
Noor University for financial support, and the Univer-
sity of Sheffield for NMR, Mass spectra and CHN.
References
1
(a) Schutz, H. Benzodiazepines, Spinger, Heidelberg, 1982.
b) Smalley, R. K. In Comprehensive Organic Chemistry,
Vol. 4, Eds. Barton, D.; Ollis, W. D., Pergamon, Oxford,
979, p. 600.
c) Landquist, J. K. In Comprehensive Heterocyclic Chem-
(
1
(
istry, Vol. 1., Eds. Katritzky, A. R.; Rees, C. W., Pergamon,
Oxford, 1984, pp. 166—170.
2
3
4
5
Barker, H. A.; Smyth, R. D.; Weissbach, H.; Toohey, J. I.;
Ladd, J. N.; Volcani, B. E. J. Biol. Chem. 1960, 235, 480.
Jackstell, A.; Frisch, A.; Beller, M.; Rottger, D.; Malaun, M.;
Bildstein, B. J. Mol. Catal. A: Chem. 2002, 185, 105.
Huynh, H. V.; Ho, J. H. H.; Neo, T. C.; Koh, L. L. J.
Organomet. Chem. 2005, 690, 3854.
(a) Erhardt, P. W. J. Med. Chem. 1987, 30, 231.
(
(
b) Preston, P. N. Chem. Heterocycl. Compd. 1980, 40, 531.
c) Gravatt, G. L.; Baguley, B. C.; Wilson, W. R.; Denny,
W. A. J. Med. Chem. 1994, 37, 4338.
6
7
(a) Kim, J. S.; Gatto, B.; Yu, C.; Liu, A.; Liu, L. F.; Lavoie,
E. J. J. Med. Chem. 1996, 39, 992.
(
b) Chen, A. Y.; Yu, C.; Gatto, B.; Liu, L. F. Proc. Natl.
Acad. Sci. U. S. A. 1993, 90, 8131.
c) Heravi, M. M.; Baghernejad, B.; Oskooie, H. A. Chin. J.
(
Chem. 2009, 27, 379.
Rott, T.; Morningstar, M. L.; Boyer, P. L.; Hughes, S. H.;
Buckheit, R. W.; Michejda, C. J. Med. Chem. 1997, 40,
4
199.
(
b) Cui, Y.; Tang, X. B.; Shao, C. X.; Li, J. T.; Sun, W. H.
Chin. J. Chem. 2005, 23, 589.
8
9
0
Lee, K. J.; Janda, K. D. Can. J. Chem. 2001, 79, 1556.
Bhatnagar, I.; George, M. V. Tetrahedron 1998, 24, 1293.
Stephenes, F. F.; Bower, J. D. J. Chem. Soc. 1949, 2971.
Beaulieu, P. L.; Hache, B.; Von Moos, E. Synthesis 2003,
1
1
1
1
683.
1
1
2
3
Du, L. H.; Wang, Y. G. Synthesis 2007, 675.
Curini, M.; Epifano, F.; Montanari, F.; Rosati, O.; Taccone,
S. Synlett 2004, 1832.
1
4
Bahrami, K.; Khodaei, M. M.; Kavianinia, I. Synthesis 2007,
5
47.
Chin. J. Chem. 2010, 28, 2249— 2254
© 2010 SIOC, CAS, Shanghai, & WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
www.cjc.wiley-vch.de
2253

 Veisi, Hojat
Veisi, Hojat
 Ghorbani-Vaghei, Ramin
Ghorbani-Vaghei, Ramin
 Faraji, Alireza
Faraji, Alireza
 Ozturk, Turan
Ozturk, Turan