The Journal of Organic Chemistry
ARTICLE
2-(Thiophene-2-yl)quinazolin-4(3H)-one (3n).22 Eluent: pet-
roleum ether/ethyl acetate (2:1). Yield: 169 mg (74%). White solid. Mp:
275À276 °C (lit.22 275À276 °C). 1H NMR (DMSO-d6, 400 MHz): δ
12.64 (s, br, 1H), 8.23 (d, J = 8.0 Hz, 1H), 8.12 (d, J = 8.0 Hz, 1H), 7.86
(d, J = 8.0 Hz, 1H), 7.79 (t, J = 8.0 Hz, 1H), 7.64 (d, J = 8.0 Hz, 1H), 7.48
(t, J = 8.0 Hz, 1H), 7.23 (t, J = 8.0 Hz, 1H). 13C NMR (DMSO-d6,
100 MHz): δ 161.7, 148.5, 147.8, 137.3, 134.6, 132.1, 129.3, 128.4,
126.8, 126.2, 125.9, 120.8. HRMS (ESI): m/z calcd for C12H9N2OS,
229.0436; found, 229.0411.
NMR (DMSO-d6, 100 MHz): δ 162.0, 152.3, 148.7, 145.0, 132.7, 131.3,
128.5, 128.0, 127.6, 127.0, 125.6, 118.5, 21.3. HRMS (ESI): m/z calcd
for C15H13N2O, 237.1028; found, 237.1019.
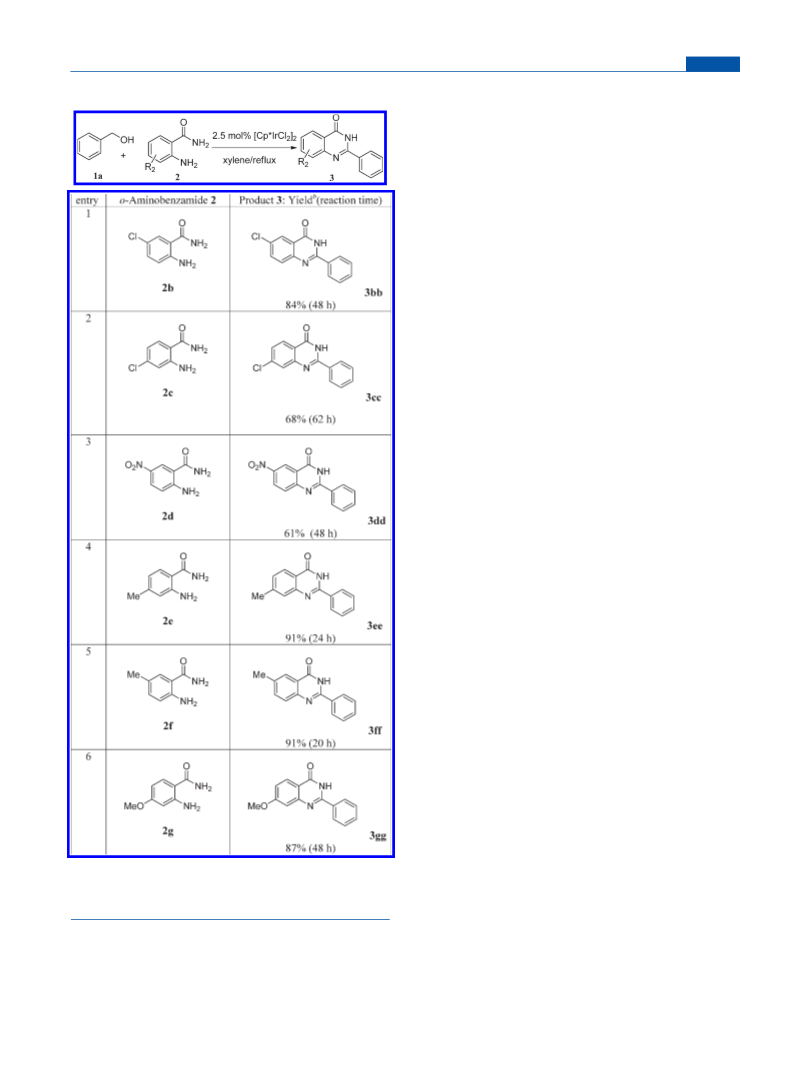
6-Methyl-2-phenylquinazolin-4(3H)-one (3ff).31 Eluent: petro-
leum ether/ethyl acetate (2:1). Yield: 215 mg (91%). White solid. Mp:
238À239 °C(lit.31 238À240 °C). 1H NMR (DMSO-d6, 400MHz):δ12.46
(s, br, 1H), 7.95 (s, 1H), 7.63À7.67 (m, 2H), 7.51À7.59 (m, 3H), 2.45
(s,3H).13C NMR (DMSO-d6,100MHz):δ162.1, 151.4, 146.6, 136.2, 135.8,
132.7, 131.1, 128.5, 127.5, 127.3, 125.0, 120.6, 20.8. HRMS (ESI): m/z calcd
for C15H13N2O, 237.1028; found, 237.1009.
2-Benzylquinazolin-4(3H)-one (3o).28 Eluent: petroleum
ether/ethyl acetate (2:1). Yield: 203 mg (86%). White solid. Mp:
7-Methoxy-2-phenylquinazolin-4(3H)-one (3gg).33 Eluent:
petroleum ether/ethyl acetate (2:1). Yield: 219 mg (87%). White solid.
Mp: 235À236 °C. 1H NMR (DMSO-d6, 400 MHz): δ 12.41 (s, br, 1H),
8.17 (d, J = 8.0 Hz, 2H), 8.04 (d, J = 8.0 Hz, 1H), 7.52À7.59 (m, 3H),
7.18 (d, J = 8.0 Hz, 1H), 7.09 (dd, J = 8.0 Hz, 1H), 3.91 (s, 3H). 13C
NMR (DMSO-d6, 100 MHz): δ 164.1, 161.7, 152.8, 150.9, 132.6, 131.3,
128.5, 127.6, 127.4, 116.1, 114.3, 108.4, 55.6. HRMS (ESI): m/z calcd
for C15H13N2O2, 253.0977; found, 253.0975.
1
244À246 °C (lit.28 245À247 °C). H NMR (DMSO-d6, 400 MHz):
δ 12.42 (s, br, 1H), 8.07 (d, J = 8.0 Hz, 1H), 7.76 (t, J = 8.0 Hz, 1H), 7.60
(d, J = 8.0 Hz, 1H), 7.45 (t, J = 8.0 Hz, 1H), 7.38 (d, J = 8.0 Hz, 2H), 7.31
(t, J = 8.0 Hz, 2H), 7.23 (t, J = 8.0 Hz, 1H), 3.93 (s, 1H). 13C NMR
(DMSO-d6, 100 MHz): δ 161.8, 155.9, 148.7, 136.5, 134.3, 128.8, 128.4,
126.8, 126.7, 126.1, 125.6, 120.6, 40.7. HRMS (ESI): m/z calcd for
C15H13N2O, 237.1028; found, 237.1015.
2-Phenyl-2,3-dihydroquinazolin-4(1H)-one (5a).24 To o-
aminobenzamide (2a; 1.36 g, 10 mmol) in toluene (20 mL) was added
benzaldehyde (4a; 1.06 g, 10 mmol) under N2 at room temperature. The
reaction mixture was refluxed for 8 h. The solution was then cooled to
room temperature, the solvent was removed, and the residue was
purified by silica gel chromatography (EtOAc:n-heptane = 1:5). Yield:
2.08 g (97%). White solid. Mp: 218À219 °C (lit.24 218À219 °C). 1H
NMR (CDCl3, 400 MHz): δ 7.95 (d, J = 8.0 Hz, 1H), 7.59À7.60 (m,
2H), 7.44À7.46 (m, 3H), 7.37 (t, J = 8.0 Hz, 1H), 6.91 (t, J = 8.0 Hz,
1H), 6.67 (d, J = 8.0 Hz, 1H), 5.91 (s, 1H), 5.76 (s, 1H), 4.39 (s, 1H).
13C NMR (CDCl3, 100 MHz): δ 164.6, 147.2, 138.5, 134.0, 130.1,
129.1, 128.7, 127.2, 119.7, 115.6, 114.5, 69.1. HRMS (ESI): m/z calcd
for C14H13N2O, 225.1028; found, 225.1029.
2-Phenethylquinazolin-4(3H)-one (3p).29 Eluent: petroleum
ether/ethyl acetate (2:1). Yield: 227 mg (91%). White solid. Mp:
207À208 °C (lit.29 208 °C). 1H NMR (DMSO-d6, 400 MHz):
δ 12.25 (s, br, 1H), 8.09 (d, J = 8.0 Hz, 1H), 7.78 (t, J = 8.0 Hz, 1H),
7.62 (d, J = 8.0 Hz, 1H), 7.47 (t, J = 8.0 Hz, 1H), 7.27À7.29 (m, 4H),
7.16À7.20 (m, 1H), 3.05 (t, J = 8.0 Hz, 2H), 2.89 (t, J = 8.0 Hz, 2H). 13C
NMR (DMSO-d6, 100 MHz): δ 161.6, 156.7, 148.4, 140.6, 134.3, 128.3,
128.3, 126.4, 126.0, 125.7, 120.7, 36.2, 32.4. HRMS (ESI): m/z calcd for
C16H15N2O, 251.1184; found, 251.1150.
2-Butylquinazolin-4(3H)-one (3q). Eluent: petroleum ether/
ethyl acetate (2:1). Yield: 170 mg (84%). White solid. Mp: 108À109 °C
(lit.30 109À110 °C). 1H NMR (DMSO-d6, 400 MHz): δ 12.16 (s, br, 1H),
8.06 (d, J = 8.0 Hz, 1H), 7.75 (t, J = 8.0 Hz, 1H), 7.58 (d, J = 8.0 Hz, 1H),
7.44 (t, J = 8.0 Hz, 1H), 2.58 (t, J = 8.0 Hz, 2H), 1.65À1.73 (m, 2H),
1.29À1.38 (m, 2H), 0.89 (t, J = 8.0 Hz, 3H), 13C NMR (DMSO-d6,
100 MHz): δ 161.7, 157.4, 148.8, 134.2, 126.7, 125.8, 125.6, 120.7, 34.1,
28.8, 21.6, 13.6. HRMS (ESI): m/z calcd for C12H15N2O, 203.1184;
found, 203.1160.
’ ASSOCIATED CONTENT
S
Supporting Information. Description of the general
b
experimental procedures and NMR spectra of the compounds.
This material is available free of charge via the Internet at http://
pubs.acs.org.
6-Chloro-2-phenylquinazolin-4(3H)-one (3bb).22 Eluent:
petroleum ether/ethyl acetate (2:1). Yield: 215 mg (84%). White solid.
Mp: 296À297 °C (lit.22 295À296 °C). 1H NMR (DMSO-d6, 400
MHz): δ 12.70 (s, br, 1H), 8.17 (d, J = 8.0 Hz, 2H), 8.09 (s, 1H), 8.17
(dd, J = 8.0 Hz, 1H), 7.76 (d, J = 8.0 Hz, 1H), 7.53À7.62 (m, 3H). 13C
NMR (DMSO-d6, 100 MHz): δ 161.1, 152.8, 147.4, 134.6, 132.4,
131.5, 130.7, 129.7, 128.5, 127.8, 124.8, 122.2. HRMS (ESI): m/z
calcd for C14H10ClN2O, 257.0482; found, 257.0466.
’ AUTHOR INFORMATION
Corresponding Author
*E-mail: jianguang.zhou@novartis.com.
7-Chloro-2-phenylquinazolin-4(3H)-one (3cc).31 Eluent:
petroleum ether/ethyl acetate (2:1). Yield: 174 mg (68%). White solid.
Mp: 287À288 °C (lit.31 286À288 °C). 1H NMR (DMSO-d6, 400
MHz): δ 12.66 (s, br, 1H), 8.13À8.18 (m, 3H), 7.79 (s, 1H), 7.53À7.61
(m, 4H). 13C NMR (DMSO-d6, 100 MHz): δ 161.6, 153.7, 149.8, 139.1,
132.3, 131.6, 128.6, 127.9, 127.8, 126.7, 126.5, 119.7. HRMS (ESI): m/z
calcd for C14H10ClN2O, 257.0482; found, 257.0451.
’ ACKNOWLEDGMENT
We thank Wei Li for assistance with HPLC and HRMS.
’ REFERENCES
(1) Mhaske, S. B.; Argade, N. P. Tetrahedron 2006, 62, 9787.
(2) Ma, Z.-Z.; Hano, Y.; Nomura, T.; Chen, Y.-J. Heterocycles 1997,
46, 541.
(3) Chen, A. L.; Chen, K. K. J. Am. Pharm. Assoc. 1933, 22, 716.
(4) (a) Horton, D. A.; Bourne, G. T.; Smythe, M. L. Chem. Rev. 2003,
103, 893. (b) Witt, A.; Bergman, J. Curr. Org. Chem. 2003, 7, 659.
(5) Mitobe, Y.; Ito, S.; Mizutani, T.; Nagase, T.; Sato, N.; Tokita, S.
Bioorg. Med. Chem. Lett. 2009, 19, 4075.
(6) Abdel-Jalil, R. J.; Aldoqum, H. M.; Ayoub, M. T.; Voelter, W.
Heterocycles 2005, 65, 2061.
(7) Balakumar, C.; Lamba, P.; Kishore, D. P.; Narayana, B. L.; Rao,
K. V.; Rajwinder, K.; Rao, A. R.; Shireesha, B.; Narsaiah, B. Eur. J. Med.
Chem. 2010, 45, 4904.
6-Nitro-2-phenylquinazolin-4(3H)-one (3dd).32 The product
was washed with petroleum ether/ethyl acetate (5:1). Yield: 163 mg
(61%). Brown solid. Mp: 297À298 °C (lit.32 297À299 °C). 1H NMR
(DMSO-d6, 400 MHz): δ 13.01 (s, br, 1H), 8.81 (s, 1H), 8.53 (d,
J = 8.0 Hz, 1H), 8.21 (d, J = 8.0 Hz, 2H), 7.89 (d, J = 8.0 Hz, 1H),
7.55À7.65 (m, 3H). 13C NMR (DMSO-d6, 100 MHz): δ 161.8, 155.8,
152.9, 144.5, 132.1, 128.9, 128.6, 128.5, 128.3, 128.2, 127.0, 122.0, 120.9.
HRMS (ESI): m/z calcd for C14H10N3O3, 268.0722; found, 268.0705.
7-Methyl-2-phenylquinazolin-4(3H)-one (3ee).22 Eluent:
petroleum ether/ethyl acetate (2:1). Yield: 215 mg (91%). Slight yellow
soild. Mp: 240À241 °C (lit.22 240À241 °C). 1H NMR (DMSO-d6, 400
MHz): δ 12.45 (s, br, 1H), 8.16 (d, J = 8.0 Hz, 2H), 8.03 (d, J = 8.0 Hz,
1H), 7.54À7.58 (m, 4H), 7.34 (d, J = 8.0 Hz, 1H), 2.47 (s, 3H). 13C
(8) Hisano, T.; Ichikawa, M.; Nakagawa, A.; Tsuji, M. Chem. Pharm.
Bull. 1975, 23, 1910.
7735
dx.doi.org/10.1021/jo201054k |J. Org. Chem. 2011, 76, 7730–7736

 Zhou, Jianguang
Zhou, Jianguang
 Fang, Jie
Fang, Jie