3
484
V. K. Aggarwal et al. / Tetrahedron Letters 50 (2009) 3482–3484
9.
(a) Llera, J. M.; Fernández, I.; Alcudia, F. Tetrahedron Lett. 1991, 32, 7299–7302;
b) Fernández, I.; Khiar, N.; Llera, J. M.; Alcudia, F. J. Org. Chem. 1992, 57, 6789–
References and notes
(
6
5
796; (c) Khiar, N.; Fernández, I.; Alcudia, F. Tetrahedron Lett. 1994, 35, 5719–
722; (d) Khiar, N.; Alcudia, F.; Espartero, J.-L.; Rodríguez, L.; Fernández, I. J. Am.
1
.
(a) Liu, G.; Cogan, D. A.; Ellman, J. A. J. Am. Chem. Soc. 1997, 119, 9913–9914; (b)
Davis, F. A.; Reddy, R. T.; Reddy, R. E. J. Org. Chem. 1992, 57, 6387–6389; (c)
Weix, D. J.; Ellman, J. A. Org. Synth. 2005, 82, 157–162.
Chem. Soc. 2000, 122, 7598–7599; (e) Khiar, N.; Araujo, C.; Alcudia, F.;
Fernández, I. J. Org. Chem. 2002, 67, 345–356; (f) Ballcells, D.; Maseras, F.;
Khiar, N. Org. Lett. 2004, 6, 2197–2200; (g) Balcells, D.; Ujaque, G.; Fernández, I.;
Khiar, N. Adv. Synth. Catal. 2007, 349, 2103–2110.
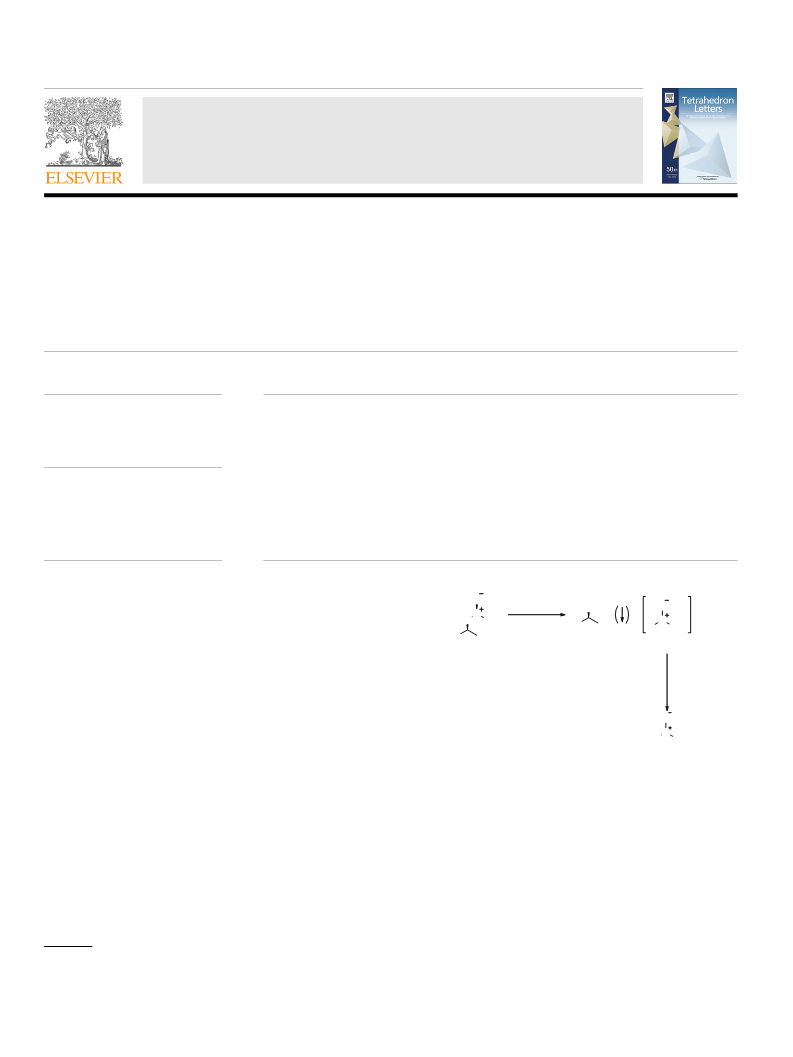
0. Drabowicz et al. have previously studied the reaction of t-BuSOCl with alcohols
and found that NEt and 8 in Et O gave a dr of 91:9 and 66% yield after 3 days.
2
.
For reviews see: (a) Ellman, J. A.; Owens, T. D.; Tang, T. P. Acc. Chem. Res. 2002,
35, 984–995; (b) Ellman, J. A. Pure Appl. Chem. 2003, 75, 39–46; (c) Zhou, P.;
Chen, B.-C.; Davis, F. A. Tetrahedron 2004, 60, 8003–8030; (d) Daniel, M.;
Stockman, R. A. Tetrahedron 2006, 62, 8868–8905; (e) Lin, G.-Q.; Xu, M.-H.;
Zhong, Y.-W.; Sun, X.-W. Acc. Chem. Res. 2008, 41, 831–840.
1
1
3
2
They attributed the low yield to trapping of 8 as its HCl salt but noted that
added base had a limited effect on yield and dr. They also noted that there was
little effect of solvent on diastereocontrol: Drabowicz, J.; Bujnicki, B.; Biscarini,
P.; Mikolajczyk, M. Tetrahedron: Asymmetry 1999, 10, 3177–3187.
3
.
.
Unthank, M. G.; Hussain, N.; Aggarwal, V. K. Angew. Chem., Int. Ed. 2006, 45,
7
066–7069. Angew. Chem. 2006, 118, 7224–7227.
(a) Wuts, P. G. M.; Greene, T. W. Greene’s Protective Groups in Organic Synthesis,
th ed.; John Wiley & Sons: Hoboken, 2007; (b) Sun, P.; Weinreb, S. M. J. Org.
4
4
1. For other dynamic kinetic resolution approaches see: (a) Evans, J. W.; Fierman,
M. B.; Miller, S. J.; Ellman, J. A. J. Am. Chem. Soc. 2004, 126, 8134–8135; (b)
Peltier, H. M.; Evans, J. W.; Ellman, J. A. Org. Lett. 2005, 7, 1733–1736; (c)
Shibata, N.; Matunaga, M.; Nakagawa, M.; Fukuzumi, T.; Nakamura, S.; Toru, T.
J. Am. Chem. Soc. 2005, 127, 1374–1375; (d) Shibata, N.; Matsunaga, M.;
Fukuzumi, T.; Nakamura, S.; Toru, T. Synlett 2005, 1699–1702.
2. Kice, J. L.; Lee, T. W. S.; Pan, S.-T. J. Am. Chem. Soc. 1980, 102, 4448–4455.
3. For capture of the p-toluenesulfinyl auxiliary with menthol followed by cycles
of fractional crystallization and epimerization see: Davis, F. A.; Reddy, R. E.;
Szewczyk, J. M. J. Org. Chem. 1995, 60, 7037–7039.
4. (a) Han, Z.; Krishnamurthy, D.; Grover, P.; Fang, Q. K.; Su, X.; Wilkinson, H. S.;
Lu, Z.-H.; Magiera, D.; Senanayake, C. H. Tetrahedron 2005, 61, 6386–6408; (b)
Han, Z.; Krishnamurthy, D.; Grover, P.; Fang, Q. K.; Senanayake, C. H. J. Am.
Chem. Soc. 2002, 124, 7880–7881; (c) Han, Z.; Krishnamurthy, D.; Pflum, D.;
Grover, P.; Wald, S. A.; Senanayake, C. H. Org. Lett. 2002, 4, 4025–4028.
5. The standard work-up uses MeOH/HCl which presumably traps the sulfinyl
chloride as methyl tert-butylsulfinate ester.
Chem. 1997, 62, 8604–8608.
5
.
.
(a) Greenwood, N. N.; Earnshaw, A. Chemistry of the Elements; Pergammon Press
plc: Oxford, 1984; pp 821–824; (b) Grainger, R. S.; Procopio, A.; Steed, J. W. Org.
Lett. 2001, 3, 3565–3568.
6
Ellman reported the production of tert-butanesulfinic acid on treatment of a
sulfinamide with TFA-d
Brinner, K. M.; Ellman, J. A. Org. Biomol. Chem. 2005, 3, 2109–2113; Ellman used
NH Cl as an additive for auxiliary cleavage by acidic resins and MeOH, and
1 2
/D O in an NMR study but it was not isolated: (a)
1
1
4
suggested it was effective due to attack of chloride at sulfur: (b) Mukade, T.;
Dragoli, D. R.; Ellman, J. A. J. Comb. Chem. 2003, 5, 590–596.
For leading references see: (a) Solladié, G.; Hutt, J.; Girardin, A. Synthesis 1987,
1
7
.
.
1
73; (b) Solladié, G. Synthesis 1981, 185–196.
Milder conditions (dilute HCl, or use of NH Cl) either caused racemization or no
reaction. Use of an acid with a non-nucleophilic counterion (e.g., HBF ) with
MeOH or menthol gave racemization or no deprotection. Activation of the
sulfinamide with a Boc group (successful) followed by treatment with NH or
LiNH returned starting material.
8
4
4
1
3
2

 Aggarwal, Varinder K.
Aggarwal, Varinder K.
 Barbero, Nekane
Barbero, Nekane
 McGarrigle, Eoghan M.
McGarrigle, Eoghan M.
 Mickle, Greg
Mickle, Greg
 Navas, Raquel
Navas, Raquel
 Suárez, José Ramón
Suárez, José Ramón
 Unthank, Matthew G.
Unthank, Matthew G.
 Yar, Muhammad
Yar, Muhammad