
Synlett p. 1806 - 1809 (2016)
Update date:2022-08-11
Topics:
 Zhou, Weiyou
Zhou, Weiyou
 Taboonpong, Piyada
Taboonpong, Piyada
 Aboo, Ahmed Hamdoon
Aboo, Ahmed Hamdoon
 Zhang, Lingjuan
Zhang, Lingjuan
 Jiang, Jun
Jiang, Jun
 Xiao, Jianliang
Xiao, Jianliang
A convenient catalytic procedure has been developed for the oxidative dehydrogenations of N-heterocycles. Combining catalytic FeCl2 with DMSO yields a catalyst that promotes the dehydrogenation of tetrahydroquinolines and related heterocycles under 1 bar of O2, affording the corresponding N-heteroaromatic products in moderate yields.
View More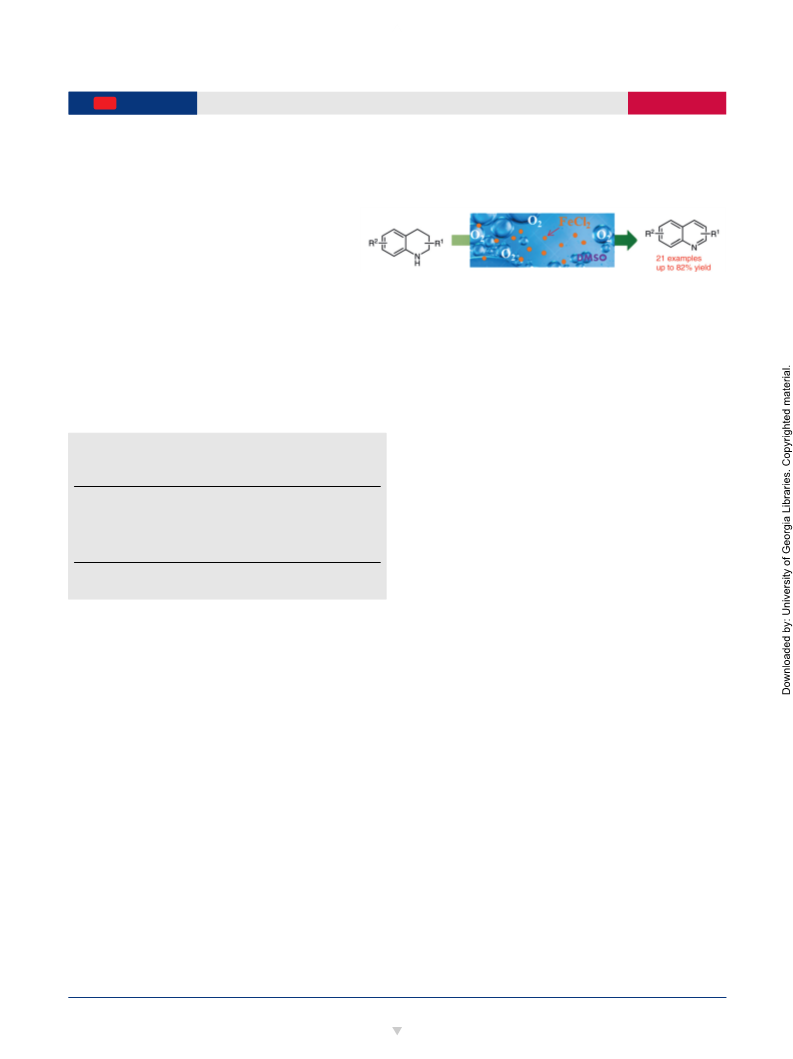


Contact:+86-731-84427351
Address:154 JIANXIANG SOUTH ROAD
ZHEJIANG JIANYE CHEMICAL CO.,LTD.
Contact:86-571-64149273,64149234
Address:No. 48, Fuxi Road, Meicheng Town
Jiangxi Hessence Chemicals Co., Ltd.
Contact:+86 796 3511924
Address:Chengxi Industrial Park, Jishui County, Jiangxi Province 331600 China.
Changzhou Hopschain Chemical Co.,Ltd
Contact:86-519-85528066
Address:Room 710, Unit A, Xingbei Development Mansion, Tongjiang Road, Changzhou City,213000, China
Anyang Double Circle Auxiliary CO.,LTD
Contact:0086-134 6082 4403
Address:dongfeng road, anyang city, henan province,china
Doi:10.1021/jo048413w
(2005)Doi:10.1246/cl.2003.718
(2003)Doi:10.1016/j.electacta.2005.12.019
(2006)Doi:10.1016/j.ica.2016.01.001
(2016)Doi:10.1016/S0925-8388(02)00369-9
(2002)Doi:10.1021/jo00228a001
(1987)