Paper
RSC Advances
in ppm and coupling constants given in Hz. The sonication was
performed in a UP 400S ultrasonic processor equipped with
a 3 mm wide and 140 mm long probe.
2 T. P. Umile, Catalysis for Sustainability, CRC Press, 2015.
3 R. D. Howells and J. D. Mc Cown, Chem. Rev., 1977, 77, 69–92.
4 N. C. Marziano, L. Ronchin, C. Tortato, A. Zingales and
A. A. Sheikh-Osman, J. Mol. Catal. A: Chem., 2001, 174,
265–277.
Catalyst synthesis
5 D.-S. Wang and Y.-G. Zhou, Tetrahedron Lett., 2010, 51, 3014–
3017.
6 N. Gigant and I. Gillaizeau, Org. Lett., 2012, 14, 4622–4625.
7 H. Ren, G. Zhao, S. Zhang, P. Cui and J. Huang, Catal.
Commun., 2012, 18, 85.
8 S. R. Mothe and P. W. H. Chan, J. Org. Chem., 2009, 74, 5887–
5893.
9 T. Jin, M. Himuro and Y. Yamamoto, Angew. Chem., Int. Ed.,
2009, 48, 5893–5896.
10 M. Blocker, S. Immaneni and A. Shaikh, Tetrahedron Lett.,
2014, 55, 5572–5575.
11 Z. P. Tachrim, L. Wang, Y. Murai, T. Yoshida, N. Kurokawa,
F. Ohashi, Y. Hashidoko and M. Hashimoto, Catalysts, 2017,
7, 40–68.
12 W.-Sh. Wang, P. Chen and Y. Tang, Tetrahedron, 2017, 73,
2731–2739.
13 A. S. Jadhav and R. V. Anand, Eur. J. Org. Chem., 2017, 3716–
3721.
14 M. Chidambaram, D. Curulla-Ferre, A. P. Singh and
B. G. Anderson, J. Catal., 2003, 220, 442–456.
15 D. O. Bennardi, G. P. Romanelli, J. C. Autino and L. R. Pizzio,
Appl. Catal., A, 2007, 324, 62–68.
16 D. O. Bennardi, G. P. Romanelli, J. C. Autino and L. R. Pizzio,
Catal. Commun., 2009, 10, 576–581.
17 P. N. Liu, F. Xia, Q. W. Wang, Y. J. Ren and J. Q. Chen, Green
Chem., 2010, 12, 1049–1055.
18 S. Khaksar, M. Tajbakhsh and M. Gholami, C. R. Chim.,
2014, 17, 30–34.
19 Y.-H. Liu, J. Deng, J.-W. Gao and Zh.-H. Zhang, Adv. Synth.
Catal., 2012, 354, 441–447.
20 G. M. Whitesides, Nature, 2006, 442, 368–373.
21 C. P. Park and D.-P. Kim, Angew. Chem., Int. Ed., 2010, 49,
6825–6829.
To a mixture of SPIONs (1.0 g) in diethyl ether (50 mL), TfOH
(1.5 mmol) was added dropwise, and the resulting suspension
was sonicated in the ultrasonic bath for 60 min at room
temperature. The solvent was removed and the residue was
heated under vacuum at 70 ꢀC for 24 h to obtain TfOH@SPION.
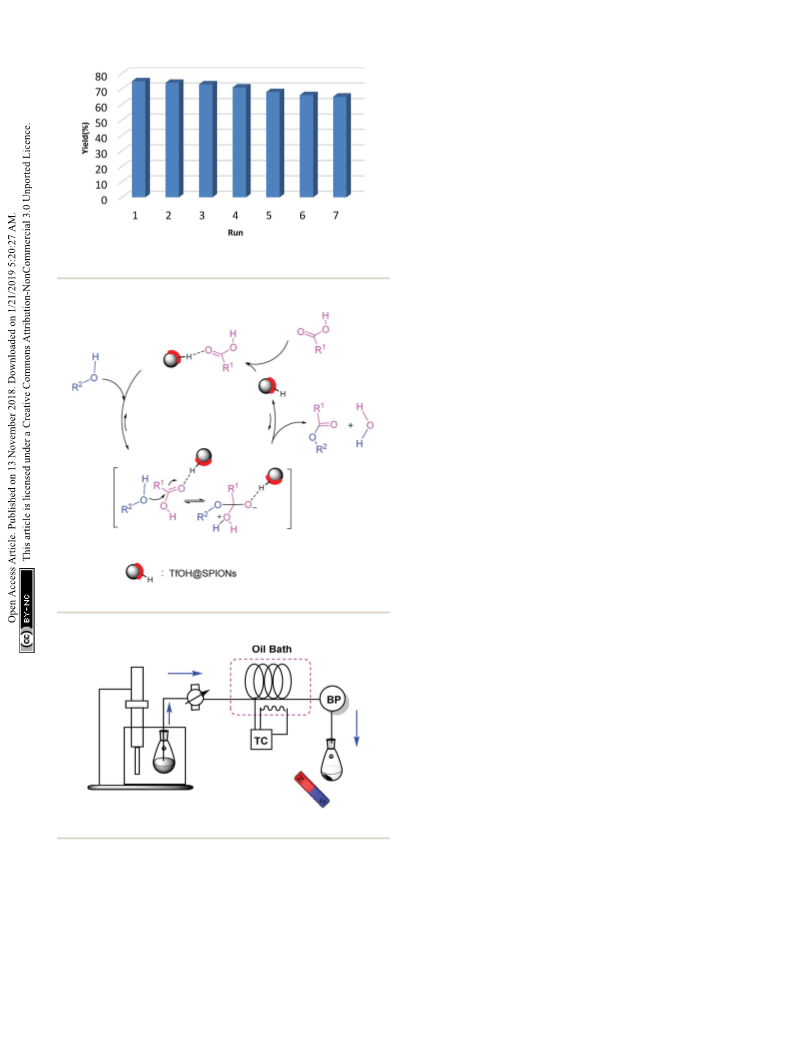
Microreactor designing
The microreactor system used in this study includes an in-house-
made tubing glass reactor which can be implemented in any
chemistry laboratory. The microtube reactor was fabricated in
a glass column (internal diameter, 0.5 mm; internal volume, 0.6
mL; length, 200 cm), which was placed in an oil bath. The
dispersed mixture of substrates and the catalyst was delivered to
the microreactor by a peristaltic pump. A temperature controller
(TC) was used for balancing and setting up heating and a back-
pressure regulator (BPR) for pressure control (Scheme 1).
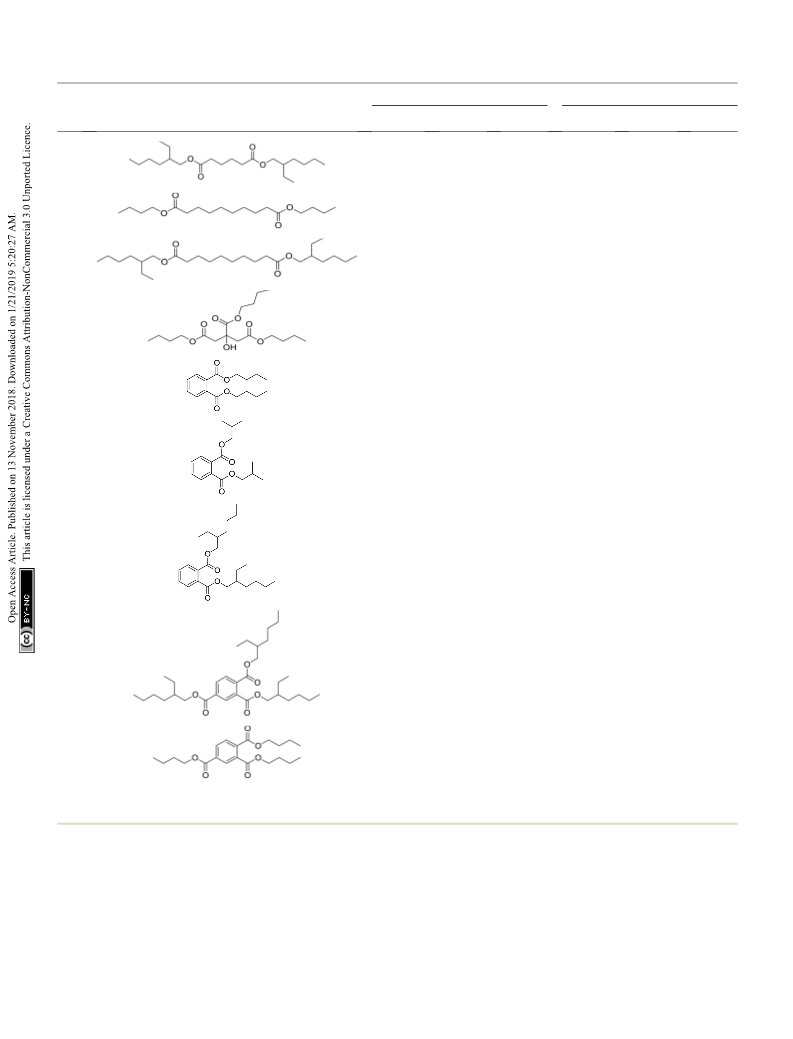
General procedure for synthesis of plasticizers
A sonicated mixture of 1 mL dicarboxylic acid : alcohol or tricar-
boxylic acid : alcohol (1 : 2 and 1 : 3 molar ratio, respectively), and
the catalyst (40 mg, 0.13 mol% based on TfOH) at ambient
temperature and pressure was fed to the microreactor by a peri-
staltic pump. Aerwards, the ow rate of the mixture was adjusted
to the desired value (0.4 mL hÀ1). The reactor was then heated at
90 ꢀC. Aer the residence time was reached (15–95 min), the
discharge was collected out of BPR in a glass vessel equipped with
an external permanent magnet (Scheme 1). The collected catalyst
by the magnet, was washed two times with absolute ethanol (2 Â 1
mL), air-dried, and used directly for the next round. Aer separa-
tion of the catalyst from the resulting crude product, the volatiles
were removed in vacuum. The organic residue was extracted with
ethyl acetate/water (3 Â 5 mL), and the combined organic phase
was dried over anhydrous Na2SO4. Pure products were obtained in
75–96% yields aer removal of solvent under reduced pressure.
22 T. Rodrigues, P. Schneider and G. Schneider, Angew. Chem.,
Int. Ed., 2014, 53, 5750–5758.
23 Q. Deng, R. Shen, Z. Zhao, M. Yan and L. Zhang, Chem. Eng.
J., 2015, 262, 1168–1174.
24 R. Porta, M. Benaglia and A. Puglisi, Org. Process Res. Dev.,
2016, 20, 2–25.
Conflicts of interest
There are no conicts to declare.
25 N. J. W. Straathof, B. J. P. Tegelbeckers, V. Hessel, X. Wang
¨
and T. Noel, Chem. Sci., 2014, 5, 4768–4773.
Acknowledgements
26 D. Cantillo, O. de Frutos, J. A. Rincon, C. Mateos and
C. O. Kappe, Org. Lett., 2014, 16, 896–899.
27 B. Gutmann, D. Cantillo and C. O. Kappe, Angew. Chem., Int.
Ed., 2015, 54, 6688–6728.
28 A. Nagaki, K. Hirose, O. Tonomura, S. Taniguchi, T. Taga,
Sh. Hasebe, N. Ishizuka and J. Yoshida, Org. Process Res.
Dev., 2016, 20, 687–691.
M. H. B. gratefully acknowledges the nancial support through
the startup funds from the University of Arkansas. A. R. K.
gratefully thanks the Center of Excellence of Chemistry and the
Research Council of the University of Isfahan (CECUI) for
nancial support of this work.
29 P. Poechlauer, M. Vorbach, M. Kotthaus, S. Braune,
R. Reintjens, F. Mascarello and G. Kwant, Micro Process
Engineering, Wiley-VCH, Weinheim, 2009, vol. 3.
Notes and references
1 R. Sheldon, Green Chemistry and Catalysis, John Wiley &
Sons, 2015.
This journal is © The Royal Society of Chemistry 2018
RSC Adv., 2018, 8, 37835–37840 | 37839

 Tashi, Maryam
Tashi, Maryam
 Shafiee, Behnaz
Shafiee, Behnaz
 Sakamaki, Yoshie
Sakamaki, Yoshie
 Hu, Ji-Yun
Hu, Ji-Yun
 Heidrick, Zachary
Heidrick, Zachary
 Khosropour, Ahmad R.
Khosropour, Ahmad R.
 Beyzavi, M. Hassan
Beyzavi, M. Hassan