R.K. Sharma, S. Gulati / Journal of Molecular Catalysis A: Chemical 363–364 (2012) 291–303
303
Table 6
[3] J. Otera, Esterification: Methods, Reactions and Applications, Wiley, New York,
2003.
Catalytic reusability test for oxidative esterification of benzaldehyde with
methanol.a
[4] R.C. Larock, Comprehensive Organic Transformations, VCH, New York, 1989.
[5] K.E. Kovi, C. Wolf, Chem. Eur. J. 14 (2008) 6302–6315.
[6] H. Miyamura, T. Yasukawa, S. Kobayashi, Green Chem. 12 (2010) 776–778.
[7] B.S. Bal, Y.W. Childers, H.W. Pinnick, Tetrahedron 11 (1980) 2091–2096.
[8] M. Okimoto, T. Chiba, J. Org. Chem. 53 (1987) 218–220.
[9] R. Gopinath, B.K. Patel, Org. Lett. 2 (2000) 577–579.
[10] B.R. Travis, M. Sivakumar, G.O. Hollist, B. Borhan, Org. Lett. 5 (2003) 1031–1034.
[11] S.P. Chavan, S.W. Danatle, C.A. Gavande, M.S. Venkataraman, C. Praveen, Synlett
2 (2002) 267–268.
[12] T.M.A. Shaikh, L. Emmanuvel, A. Sudalai, Synth. Commun. 37 (2007) 2641–2646.
[13] X.F. Wu, C. Darcel, Eur. J. Org. Chem. 8 (2009) 1144–1147.
[14] Y. Diao, R. Yan, S. Zhang, P. Yang, Z. Li, L. Wang, H. Dong, J. Mol. Catal. A: Chem.
303 (2009) 35–42.
[15] K.R. Reddy, M. Venkateshwar, C.U. Maheshwari, S. Prashanthi, Synth. Commun.
40 (2010) 186–195.
Run
Conversionb (%)
Selectivity (%)
Fresh
100
100
100
100
98
100
100
>99
>99
>99
>99
>99
1
2
3
4
5
6
98
97
a
Reaction conditions: catalyst (20 mg), benzaldehyde (1 mmol), methanol
(5 mmol) and 30% H2O2 (2 mmol), reflux at 60 ◦C for 3 h.
b
Conversion and selectivity were determined by GC.
[16] J.H. Clark (Ed.), Chemistry of Waste Minimization, Chapman & Hall, London,
1995.
resulting solution by atomic absorption method showed no
detectable (<0.01 ppm) manganese. The manganese content was
quantified in duplicate for each sample. This ensured that no leach-
ing of the active supported MnPc-complex occurred. Thus, the
obtained catalytic results derive exclusively from the heteroge-
neous catalyst. It is noteworthy that the work-up of the reaction
mixture is rather simple since the catalyst can be recovered by sim-
pared with the fresh catalyst. The results obtained by recycling of
the catalyst are shown in Table 6. The recycling result suggests het-
erogeneity of the catalyst. The reused catalyst was also subjected to
XRD and FT-IR spectroscopy (Supplementary material). Compari-
son of IR spectra and XRD patterns of fresh and recovered catalysts
depicts that the structural properties of the anchored complex
remains unaltered after the oxidative esterification reaction.
[17] P.T. Anastas, T.C. Williamson (Eds.), Green Chemistry: Frontiers in Benign
Chemical Synthesis and Processes, Oxford University Press, Oxford, 1998.
[18] O.V. Zalomaevaab, A.B. Sorokin, New J. Chem. 30 (2006) 1768–1773.
[19] E. Kockrick, T. Lescouet, E.V. Kudrik, A.B. Sorokin, D. Farrusseng, Chem. Com-
mun. 47 (2011) 1562–1564.
[20] H. Liu, E. Min, Green Chem. 8 (2006) 657–662.
[21] A.B. Sorokin, E.V. Kudrik, Catal. Today 159 (2011) 37–46.
[22] R.K. Sharma, S. Gulati, S. Sachdeva, Green Chem. Lett. Rev. 5 (2012) 83–87.
[23] R.K. Sharma, C. Sharma, Tetrahedron Lett. 51 (2010) 4415–4418.
[24] A.P. Wight, M.E. Davis, Chem. Rev. 102 (2002) 3589–3614.
[25] C. Li, Catal. Rev. 46 (2004) 419–492.
[26] Q.H. Fan, Y.M. Li, A.S.C. Chan, Chem. Rev. 102 (2002) 3385–3466.
[27] D. Brunel, N. Belloq, P. Sutra, A. Cauvel, M. Lasperas, P. Moreau, F. Di Renzo, A.
Galarneau, F. Fajula, Coord. Chem. Rev. 1085 (2008) 178–180.
[28] A. Corma, H. Garcia, Chem. Rev. 102 (2002) 3837–3892.
[29] R. Sharma, S. Dhingra, Designing and Synthesis of Functionalized Silica
Gels and their Applications as Metal Scavengers, Sensors and Catalysts: A
Green Chemistry Approach, LAP Lambert Academic Publishing, Germany,
2011.
[30] R.K. Sharma, D. Rawat, Inorg. Chem. Commun. 17 (2012) 58–63.
[31] R.K. Sharma, C. Sharma, J. Mol. Catal. A: Chem. 332 (2010) 53–58.
[32] R.K. Sharma, D. Rawat, J. Inorg. Organomet. Polym. 20 (2010) 698–705.
[33] R.K. Sharma, D. Rawat, J. Inorg. Organomet. Polym. 21 (2011) 619–626.
[34] R.K. Sharma, A. Pandey, S. Gulati, Appl. Catal. A: Gen. 431–432 (2012) 33–41.
[35] R.K. Sharma, C. Sharma, Catal. Commun. 12 (2011) 327–331.
[36] R.K. Sharma, D. Rawat, G. Gaba, Catal. Commun. 19 (2012) 31–36.
[37] Z. Biyiklioglu, I. Acar, H. Kantekin, Inorg. Chem. Commun. 11 (2008)
630–632.
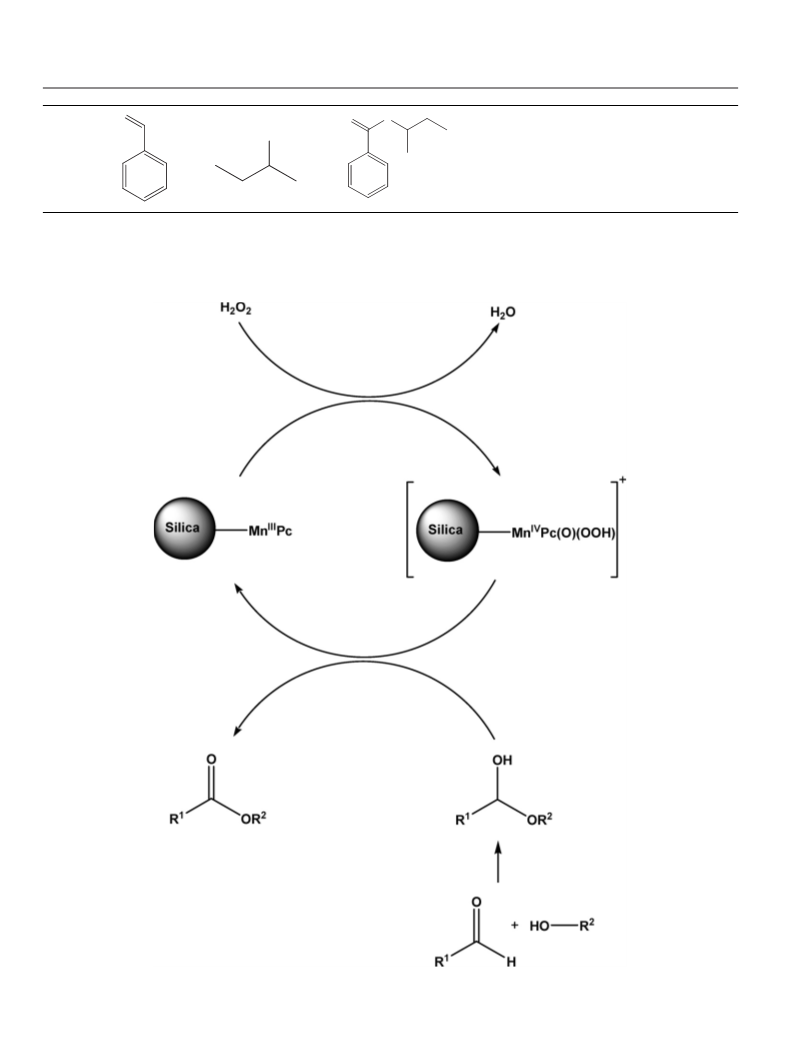
4. Conclusion
We have developed an environmentally benign protocol for the
highly selective oxidative esterification of aldehydes with alcohols
catalyzed by silica supported manganese catalyst using hydrogen
peroxide, an environment friendly oxidant. Catalyst/product sepa-
ration is very simple and the catalyst recovered after the reaction
is reusable as it retains its catalytic performance. Because of its
high recyclability and the use of hydrogen peroxide as the oxidant,
this protocol has environmental and economic advantages over
other supported catalytic systems. In addition, the advantages of
this catalytic system also includes high substrate conversion, short
reaction time, ambient temperature, mild reaction conditions, high
catalytic turnover number which make it a greener alternative for
the direct synthesis of esters from aldehydes.
[38] T.E. Youssef, Polyhedron 29 (2010) 1776–1783.
[39] A.M. Donia, A.A. Atia, W.A. Al-amrani, A.M. El-Nahas, J. Hazard. Mater. 161
(2009) 1544–1550.
[40] R. Kureshy, I. Ahmad, N.H. Khan, S. Abdi, S. Singh, P. Pandia, R. Jasra, J. Catal. 235
(2005) 28–34.
[41] S. Shylesh, A.P. Singh, J. Catal. 228 (2004) 333–346.
[42] A. Bhatt, K. Pathak, R. Jasra, R. Kureshy, N. Khan, S. Abdi, J. Mol. Catal. A: Chem.
244 (2005) 110–117.
[43] S.B. Hartono, S.Z. Qiao, J. Liu, K. Jack, B.P. Ladewig, Z. Hao, G.Q.M. Lu, J. Phys.
Chem. C 114 (2010) 8353–8362.
[44] T. Yokoi, H. Yoshitake, T. Tatsumi, J. Mater. Chem. 14 (2004) 951–957.
[45] M.A.B. Meador, E.F. Fabrizio, F. Ilhan, A. Dass, G. Zhang, P. Vassilaras, J.C. John-
ston, N. Leventis, Chem. Mater. 17 (2005) 1085–1098.
[46] S. Shylesh, A.P. Singh, J. Catal. 244 (2006) 52–64.
[47] A.B. Sorokin, P. Buisson, A.C. Pierre, Mesopor. Micropor. Mater. 46 (2001)
87–98.
[48] E. DeOliveira, C.R. Neri, A.O. Ribeiro, V.S. Garcia, L.L. Costa, A.O. Moura,
A.G.S. Prado, O.A. Serra, Y. Iamamoto, J. Colloid Interface Sci. 323 (2008)
98–104.
Acknowledgement
[49] D.J. Upadhyaya, S.D. Samant, Appl. Catal. A: Gen. 340 (2008) 42–51.
[50] C. Pereira, S. Patricio, A.R. Silva, A.L. Magalhaes, A.P. Carvalho, J. Pires, C. Freire,
J. Colloid Interface Sci. 316 (2007) 570–579.
[51] A.B. Sorokin, A. Tuel, Catal. Today 57 (2000) 45–59.
[52] H.F. Hoefnagels, D. Wu, G. de With, W. Ming, Langmuir 23 (2007) 13158–13163.
[53] J.-L. Liu, S. Xu, B. Yan, Colloids Surf. A: Physicochem. Eng. Aspects 373 (2011)
116–123.
The financial assistance from University Grant Commission and
DU-DST PURSE grant is acknowledged. Due thanks to AIRF, JNU,
Delhi, India for SEM analysis and IISc, Bangalore, India for solid state
NMR measurements.
[54] M. Kruk, M. Jaroniec, Y. Sakamoto, O. Terasaki, R. Ryoo, C.H. Ko, J. Phys. Chem.
B 104 (2000) 292–301.
[55] P. Karandhikar, A.J. Chandwadkar, M. Agashe, N.S. Ramgir, S. Sivasanker, Appl.
Catal. A: Gen. 297 (2006) 220–230.
[56] F.Z. Su, J. Ni, H. Sun, Y. Cao, H.Y. He, K.N. Fan, Chem. Eur. J. 14 (2008) 7131–7135.
[57] Y. Diaoa, R. Yana, S. Zhanga, P. Yang, Z. Li, L. Wang, H. Dong, J. Mol. Catal. A:
Chem. 303 (2009) 35–42.
[58] K.R. Reddy, M. Venkateshwar, C.U. Maheswari, S. Prashanthi, Synth. Commun.
40 (2010) 186–195.
Appendix A. Supplementary data
Supplementary data associated with this article can be
References
[59] C. Marsden, E. Taarning, D. Hansen, L. Johansen, S.K. Klitgaard, K. Egeblad, C.H.
Christensen, Green Chem. 10 (2008) 168–170.
[60] G. Yin, M. Buchalova, A.M. Danby, C.M. Perkins, D. Kitko, J.D. Carter, W.M.
Scheper, D.H. Busch, Inorg. Chem. 45 (2006) 3467–3474.
[1] E.J. Corey, N.W. Gilman, B.E. Ganem, J. Am. Chem. Soc. 90 (1968) 5616–5617.
[2] R. Gopinath, A.R. Paital, B.K. Patel, Tetrahedron Lett. 43 (2002) 5123–5126.

 Sharma
Sharma
 Gulati, Shikha
Gulati, Shikha