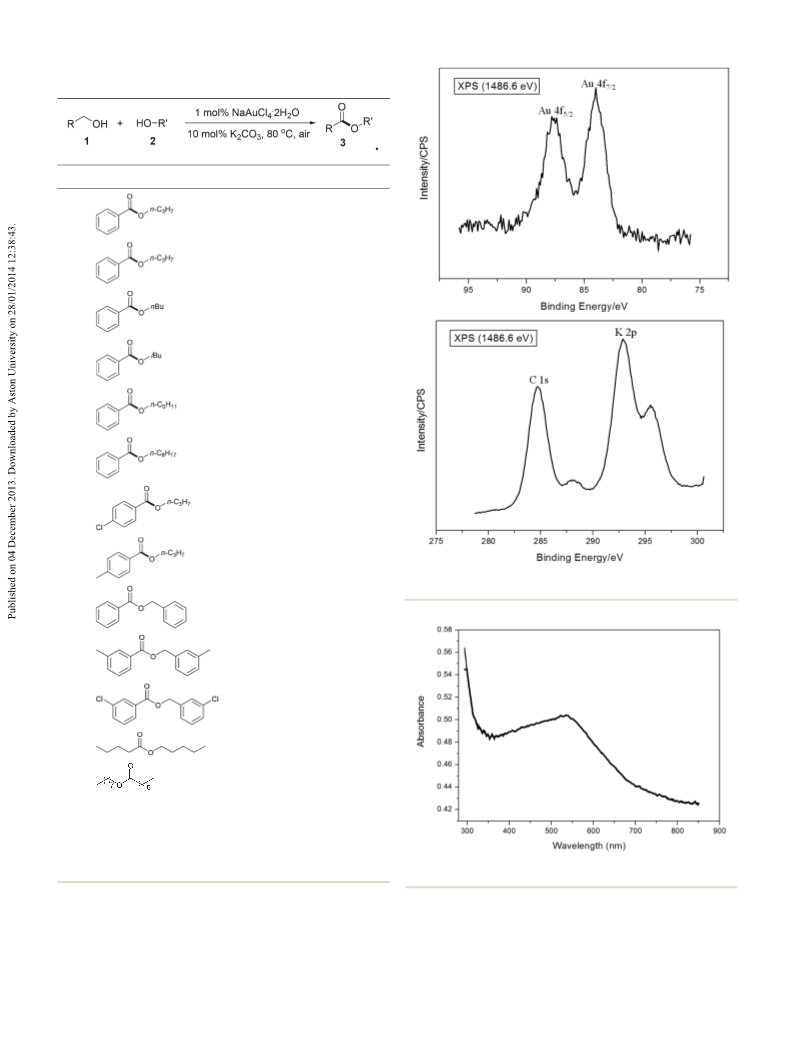
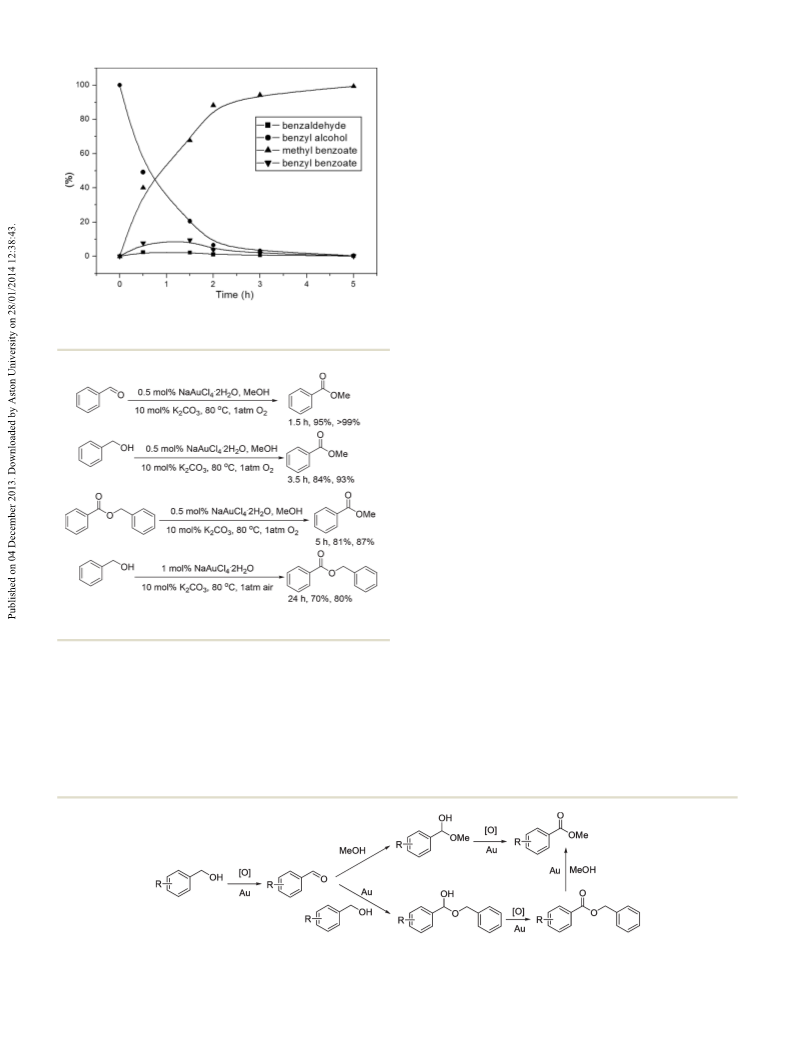
Green Chemistry
Paper
especially useful for practical applications in organic synthesis.
Further work on other applications of the present catalytic
system is currently ongoing in our laboratory.
Tetrahedron, 2005, 61, 5915; (e) N. N. Karade and
D. B. Huple, Synlett, 2005, 2039–2042.
9 (a) T. Hayashi, T. Inagaki, N. Itayama and H. Baba, Catal.
Today, 2006, 117, 210–213; (b) I. S. Nielsen, E. Taarning,
K. Egeblad, R. Madsen and C. H. Christensen, Catal. Lett.,
2007, 116, 35–40; (c) E. Taarning, A. T. Madsen,
J. M. Marchetti, K. Egeblad and C. H. Christensen, Green
Chem., 2008, 10, 408–414; (d) E. Taarning, I. S. Nielsen,
K. Egeblad, R. Madsen and C. H. Christensen, Chem-
SusChem, 2008, 1, 75–78; (e) T. Ishida, M. Nagaoka, T. Akita
and M. Haruta, Chem.–Eur. J., 2008, 14, 8456–8460;
(f) F. Z. Su, J. Ni, H. Sun, Y. Cao, H. Y. He and K. N. Fan,
Chem.–Eur. J., 2008, 14, 7131–7135; (g) S. K. Klitgaard,
A. T. D. Riva, S. Helveg, R. M. Werchmeister and
C. H. Christensen, Catal. Lett., 2008, 126, 213–217;
(h) O. Casanova, S. Iborra and A. Corma, J. Catal., 2009,
265, 109–116; (i) R. L. Oliveira, P. K. Kiyohara and
L. M. Rossi, Green Chem., 2009, 11, 1366–1370;
( j) H. Miyamura, T. Yasukawa and S. Kobayashi, Green
Chem., 2010, 12, 776–778; (k) X. Wang, G. Zhao, H. Zou,
Y. Cao, Y. Zhang, R. Zhang, F. Zhang and M. Xian, Green
Chem., 2011, 13, 2690–2695; (l) L. A. Parreira,
N. Bogdanchikova, A. Pestryakov, T. A. Zepeda,
I. Tuzovskaya, M. H. Farias and E. V. Gusevskaya, Appl.
Catal., A, 2011, 397, 145–152; (m) Y. Hao, Y. Chong, S. Li
and H. Yang, J. Phys. Chem. C, 2012, 116, 6512–6519;
(n) G. L. Brett, P. J. Miedziak, N. Dimitratos, J. A. Lopez-
Sanchez, N. F. Dummer, R. Tiruvalam, C. J. Kiely,
D. W. Knight, S. H. Taylor, D. J. Morgan, A. F. Carley and
G. J. Hutchings, Catal. Sci. Technol., 2012, 2, 97–104;
(o) P. Liu, C. Li and E. J. M. Hensen, Chem.–Eur. J., 2012,
18, 12122–12129; (p) V. V. Costa, M. Estrada, Y. Demidova,
I. Prosvirin, V. Kriventsov, R. F. Cotta, S. Fuentes,
A. Simakov and E. V. Gusevskaya, J. Catal., 2012, 292, 148–
156.
Acknowledgements
Financial support from the National Basic Research Program
of China (2009CB623505) is gratefully acknowledged.
Notes and references
1 J. Otera, Esterification: methods, reactions, and applications,
Wiley-VCH, Weinheim, 2003.
2 (a) R. C. Larock, Comprehensive Organic Transformations: A
Guide to Functional Group Preparations, Wiley-VCH,
New York, 2nd edn, 1999; (b) N. Iranpoor, H. Firouzabadi
and D. Khalili, Org. Biomol. Chem., 2010, 8, 4436–4443;
(c) N. Nowrouzi, A. M. Mehranpour and J. A. Rad, Tetrahe-
dron, 2010, 66, 9596–9601; (d) J. Otera, Acc. Chem. Res.,
2004, 37, 288–296; (e) M. Hatano, Y. Furuya, T. Shimmura,
K. Moriyama, S. Kamiya, T. Maki and K. Ishihara, Org. Lett.,
2011, 13, 426–429.
3 (a) G.-J. Brink, I. W. C. E. Arends and R. A. Sheldon, Chem.
Rev., 2004, 104, 4105–4123; (b) Y. Yoshida, K. Murakami,
H. Yorimitsu and K. Oshima, J. Am. Chem. Soc., 2010, 132,
9236–9239.
4 F. Luo, C. D. Pan and J. Cheng, Synlett, 2012, 357–366.
5 (a) K. Ekoue-Kovi and C. Wolf, Chem.–Eur. J., 2008, 14,
6302–6315; (b) W.-J. Yoo and C.-J. Li, Tetrahedron Lett.,
2007, 48, 1033–1035; (c) W.-J. Yoo and C.-J. Li, J. Org.
Chem., 2006, 71, 6266–6268; (d) B. Xu, X. Liu, J. Haubrich
and C. M. Friend, Nat. Chem., 2010, 2, 61–65; (e) X.-F. Wu
and C. Darcel, Eur. J. Org. Chem., 2009, 1144–1147.
6 (a) B. R. Travis, M. Sivakumar, G. O. Hollist and B. Borhan, 10 (a) S. Gowrisankar, H. Neumann and M. Beller, Angew.
Org. Lett., 2003, 5, 1031–1034; (b) B. E. Maki and
K. A. Scheidt, Org. Lett., 2008, 10, 4331–4334;
(c) R. Gopinath, B. Barkakaty, B. Talukdar and B. K. Patel,
Chem., Int. Ed., 2011, 50, 5139–5143; (b) C. Liu, J. Wang,
L. Meng, Y. Deng, Y. Li and A. Lei, Angew. Chem., Int. Ed.,
2011, 50, 5144–5148.
J. Org. Chem., 2003, 68, 2944–2947; (d) W. J. Yoo and 11 J. Liu, H. B. Wang, H. Zhang, X. J. Wu, H. Zhang,
C. J. Li, Tetrahedron Lett., 2007, 48, 1033–1035;
(e) K. R. Reddy, M. Venkateshwar, C. U. Maheswari and
Y. Deng, Z. Yang and A. W. Lei, Chem.–Eur. J., 2009, 15,
4437–4445.
S. Prashanthi, Synth. Commun., 2010, 40, 186–195; 12 R. Nakamura, Y. Obora and Y. Ishii, Adv. Synth. Catal.,
(f) J. S. Foot, H. Kanno, G. M. P. Giblin and R. J. K. Taylor,
Synthesis, 2003, 1055–1064 and references cited therein.
7 (a) A. J. A. Watson and J. M. J. Williams, Science, 2010, 329,
2009, 351, 1677–1684.
13 N. Yamamoto, Y. Obora and Y. Ishii, J. Org. Chem., 2011,
76, 2937–2941.
635–636; (b) G. E. Dobereiner and R. H. Crabtree, Chem. 14 L. Kaganovsky, D. Gelman and K. R. Braun, J. Organomet.
Rev., 2010, 110, 681–703; (c) G. Guillena, D. J. Ramón and Chem., 2010, 695, 260–266.
M. Yus, Chem. Rev., 2010, 110, 1611–1641; (d) E. Emer, 15 S. D. Sarkar, S. Grimme and A. Studer, J. Am. Chem. Soc.,
R. Sinisi, M. G. Capdevila, D. Petruzziello, F. De Vincentiis
2010, 132, 1190–1191.
and P. G. Cozzi, Eur. J. Org. Chem., 2011, 647–666.
16 Y. Yamamoto, Adv. Synth. Catal., 2010, 352, 478–492.
8 (a) J. N. Milocanovic, M. Vasojevic and S. Gojkovic, J. Chem. 17 E. Terazzi, L. Guénée, P. Y. Morgantini, G. Bernardinelli,
Soc., Perkin Trans. 2, 1991, 1231–1233; (b) C. E. McDonald,
L. E. Nice and N. B. Nestor, Tetrahedron Lett., 1993, 34,
B. Donnio, D. Guillon and C. Piguet, Chem.–Eur. J., 2007,
13, 1674–1691.
2741–2744; (c) G. A. Hiegel and C. B. Gilley, Synth. 18 (a) J. Zhang, G. Leitus, Y. Ben-David and D. Milstein, J. Am.
Commun., 2003, 33, 2003–2009; (d) N. Mori and H. Togo,
Chem. Soc., 2005, 127, 10840–10841; (b) C. Gunanathan,
This journal is © The Royal Society of Chemistry 2014
Green Chem.

 Wang, Lianyue
Wang, Lianyue
 Li, Jun
Li, Jun
 Dai, Wen
Dai, Wen
 Lv, Ying
Lv, Ying
 Zhang, Yi
Zhang, Yi
 Gao, Shuang
Gao, Shuang