Dalton Transactions
Paper
(3 × 5 mL) and dried over MgSO4. Nonane was added as an
internal standard (0.06 mL). The conversion and yield of the
cross coupling reactions were determined by GC-MS.
Acknowledgements
We acknowledge the National University of Singapore, the
Ministry of Education (WBS No. R-143-000-361-112) and
the Agency for Science, Technology and Research (A*Star) of
Singapore (WBS No. R-143-000-426-305) for financial support.
We thank Dr L. L. Koh, G. K. Tan and Y. M. Hong for X-ray
diffractometry assistance.
X-ray crystallography
Diffraction measurements were conducted at 100(2)–293(2) K
on a Bruker AXS APEX CCD diffractometer using Mo Kα radi-
ation (λ = 0.71073 Å). The data were corrected for Lorentz and
polarization effects with the SMART suite of programs and for
absorption effects with SADABS.12 Structure solutions and
refinements were performed by using the programs
SHELXS-9713a and SHELXL-97.13b The structures were solved
by direct methods to locate the heavy atoms, followed by differ-
ence maps for the light non-hydrogen atoms. Anisotropic
thermal parameters were refined for the rest of the non-hydro-
gen atoms. Hydrogen atoms were placed geometrically and
refined isotropically.
Notes and references
1 (a) Metal-Catalyzed Cross-Coupling Reactions, ed. A. De
Meijere and F. Diederich, Wiley-VCH, Weinheim, 2nd edn,
2004; (b) Transition Metals for Organic Synthesis, ed.
M. Beller and C. Bolm, Wiley-VCH, Weinheim, 2nd edn,
2004; (c) T.-Y. Luh, M.-K. Leung and K.-T. Wong, Chem. Rev.,
2000, 100, 3187; (d) H. B. Li, C. Seechurn and T. J. Colacot,
ACS Catal., 2012, 2, 1147; (e) M. Pagliaro, V. Pandarus,
R. Ciriminna, F. Beland and P. D. Cara, ChemCatChem,
2012, 4, 432; (f) C. Seechurn, M. O. Kitching, T. J. Colacot
and V. Snieckus, Angew. Chem., Int. Ed., 2012, 51, 5062.
2 (a) S. Lin and T. Agapie, Synlett, 2011, 1; (b) W. M. Czaplik,
M. Mayer, J. Cvengroš and A. J. von Wangelin, ChemSusCat,
2009, 2, 396; (c) B. D. Sherry and A. Fürstner, Acc. Chem.
Res., 2008, 41, 1500; (d) D. J. Cárdenas, Angew. Chem., Int.
Ed., 2003, 42, 384; (e) M. R. Netherton and G. C. Fu, Adv.
Synth. Catal., 2004, 346, 1525; (f) A. C. Frisch and
M. Beller, Angew. Chem., Int. Ed., 2005, 44, 674;
(g) O. Vechorkin, V. Proust and X. L. Hu, J. Am. Chem. Soc.,
2009, 131, 9756.
3 (a) D. Noda, Y. Sunada, T. Hatakeyama, M. Nakamura and
H. Nagashima, J. Am. Chem. Soc., 2009, 131, 6078;
(b) M. Nakamura, K. Matsuo, S. Ito and E. Nakamura,
J. Am. Chem. Soc., 2004, 126, 3686; (c) G. Cahiez, V. Habiak,
C. Duplais and A. Moyeux, Angew. Chem., Int. Ed., 2007, 46,
4364; (d) L. Ackermann, A. R. Kapdi and C. Schulzke, Org.
Lett., 2010, 12, 2298; (e) R. B. Bedford, D. W. Bruce,
R. M. Frost and M. Hird, Chem. Commun., 2005, 4161;
(f) R. B. Bedford, M. Betham, D. W. Bruce,
A. A. Danopoulos, R. M. Frost and M. Hird, J. Org. Chem.,
2006, 71, 1104; (g) H. Gao, C. Yan, X. Tao, Y. Xia, H. Sun,
Q. Shen and Y. Zhang, Organometallics, 2010, 29, 4189;
(h) K. Bica and P. Gaertner, Org. Lett., 2006, 8, 733;
(i) R. R. Chowdhury, A. K. Crane, C. Fowler, P. K. Wong and
C. M. Kozak, Chem. Commun., 2008, 94; ( j) R. B. Bedford,
D. W. Bruce, R. M. Frost, J. W. Goodby and M. Hird, Chem.
Commun., 2004, 2822; (k) A. Fürstner and R. Martin, Chem.
Lett., 2005, 34, 624; (l) A. Fürstner, R. Martin, H. Krause,
G. Seidel, R. Goddard and C. W. Lehmann, J. Am. Chem.
Soc., 2008, 130, 8773; (m) R. Martin and A. Fürstner, Angew.
Chem., Int. Ed., 2004, 43, 3955.
Conclusions
We have demonstrated the catalytic activity of dinuclear Ni(II)
amine–pyrazolyl complexes towards aryl/alkyl Grignard
reagents coupling with alkyl halides. The nature of the tether
function on the central amine moiety of the hybrid ligand
strongly affects the cross coupling selectivity and activity.
Complex 1, which contains the ligand with a pendant furan
arm, gives the best catalytic efficacy. Compared with other cata-
lyst systems,5 1 is efficient for both sp3–sp3 and sp3–sp2 types
of cross coupling in the presence of TMEDA under milder reac-
tion conditions (r.t.) with lower catalyst loading. The
TMEDA-Mg compound (4) isolated from the catalytic reaction
mixture cannot replace TMEDA’s role in the catalysis. The
TMEDA-Ni compound (5) obtained from the mixture of 1,
PhMgBr and TMEDA shows low selectivity towards the cross
coupling product. Furthermore, the 5/free ligand L1, L2 or L3
system cannot represent the catalyst generated from the
1/TMEDA system. Nevertheless, the current catalytic data
clearly show both the promoting nature of the TMEDA and the
role of the tripodal ligand. ESI-MS has been used to detect the
possible catalytic active species related to TMEDA and the tri-
podal ligand, a conclusive result cannot yet be obtained.
However, it is noteworthy that the ESI-MS spectra of the
mixture of Grignard reagent and 1 showed the presence of the
Ni(II) and Mg(II) bimetallic species supported by the tripodal
hybrid ligand L1, thus suggesting the intermetallic coordi-
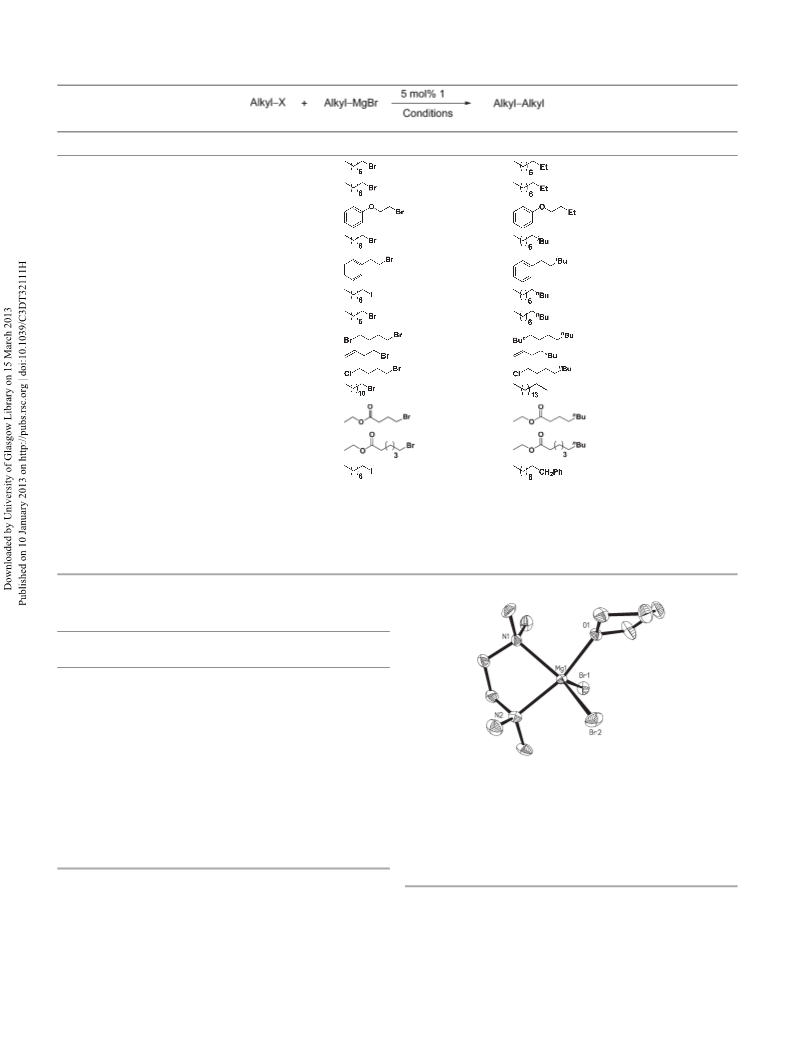
nation effect of a hybrid ligand. Together with the X-ray crystal-
lographic data, these point to a stabilising role of an additive
donor such as TMEDA towards the Grignard reagent, the Ni(II)
catalyst, as well as the possible bimetallic intermediate that
holds the key to the aryl/alkyl transfer across the metals.
Current research in our laboratory is directed at the design
of suitable intermetallic complexes supported by suitable
hybrid ligands that can serve as single-site catalysts for cross-
coupling reactions.
4 K. G. Dongol, H. Koh, M. Sau and C. L. L. Chai, Adv. Synth.
Catal., 2007, 349, 1015.
5 (a) J. Terao, H. Watanabe, A. Ikumi, H. Kuniyasu and
N. Kambe, J. Am. Chem. Soc., 2002, 124, 4222;
This journal is © The Royal Society of Chemistry 2013
Dalton Trans., 2013, 42, 5150–5158 | 5157

 Xue, Fei
Xue, Fei
 Zhao, Jin
Zhao, Jin
 Hor, T. S. Andy
Hor, T. S. Andy