Chemistry Letters Vol.34, No.8 (2005)
1153
In summary, we have developed a new and practical reac-
tion system for oxidative homo-coupling of alkyl Grignard re-
agents possessing ꢀ-hydrogens. The reaction is efficiently cata-
lyzed by 1 mol % of silver tosylate in the presence of inexpensive
1,2-dibromoethane as a stoichiometric oxidant under mild and
ligandless conditions.
1/2 R−R
RMgX
RMgX
AgOTs
Ag−R
A
Ag
B
AgBr
C
1/2 BrCH2CH2Br
This work was partially supported by a Grant-in-Aid for
Scientific Research, the Ministry of Education, Japan. T. N.
thanks the Japan Society for the Promotion of Science for the
award of a fellowship for graduate students.
1/2 C2H4
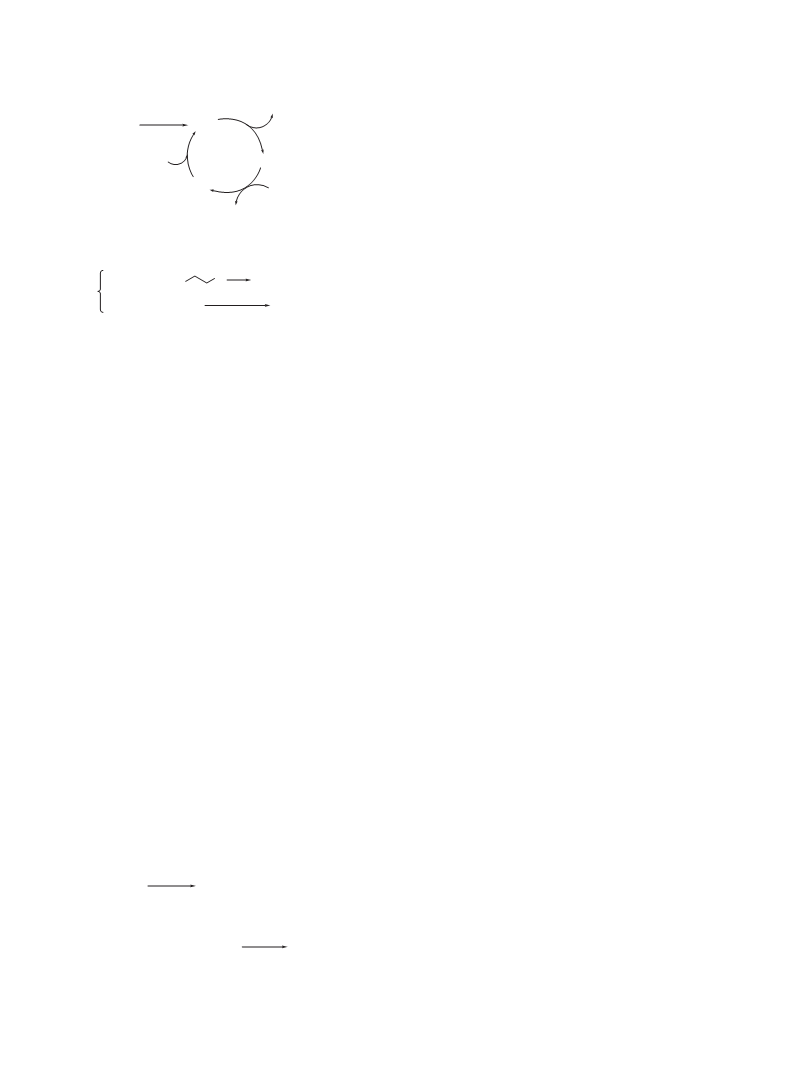
Scheme 1. Plausible mechanism for silver-catalyzed homo-cou-
pling.
Br
(i) R−MgBr
(ii) R−MgBr
+
+
+ C2H4 + MgBr2
R−Br
References and Notes
Br
1
Recent reviews on oxidative homo-coupling reactions, see: a) Alkynyl–
alkynyl coupling: P. Siemsen, R. C. Livingston, and F. Diederich, Angew.
Chem., Int. Ed., 39, 2632 (2000). b) Aryl–aryl coupling: J. Hassan, M.
Ag (catalyst)
+
R−R
MgBr2
R−Br
´
Sevignon, C. Gozzi, E. Schultz, and M. Lemaire, Chem. Rev., 102, 1359
(2002).
a) V. V. R. Rao, C. V. Kumar, and D. Devaprabhakara, J. Organomet.
Chem., 179, C7 (1979). b) Z. Z. Song and H. N. C. Wong, J. Org. Chem.,
Scheme 2. Alternative mechanism for silver-catalyzed homo-cou-
pling.
2
which would be expected to readily undergo ꢀ-hydrogen elimi-
nation giving styrene, gave a high yield of the homo-coupling
product 3e (Entry 5). The reactions of a ꢀ-disubstituted
Grignard reagent 1g and secondary alkyl Grignard reagents 1h
and 1i are slow at room temperature resulting in low yields of
the homo-coupling products, but the yields were improved to
some extent by heating the reaction at 50 ꢀC (Entries 7–12).
A plausible mechanism for the present silver-catalyzed
homo-coupling is shown in Scheme 1. The reaction of an alkyl
Grignard reagent (R–MgX) with the precatalyst AgOTs forms
an alkyl–silver species (Ag–R, A), which undergoes dispropor-
tionation to give homo-coupling product (R–R) and a silver(0)
species B.9 Oxidation of B with 1,2-dibromoethane10 giving
silver bromide (Ag–Br, C) followed by transmetallation of an
alkyl group from magnesium to silver regenerates A to carry
the catalytic cycle.
Based on the report by Kochi6c that a silver salt catalyzes the
coupling reaction of BuMgBr with BuBr giving octane (79%),
one would propose an alternative mechanism where the alkyl
Grignard reagent (R–MgBr) is converted into alkyl bromide
(R–Br) on reacting with 1,2-dibromoethane and the coupling
of R–MgBr with R–Br in the presence of a silver catalyst pro-
duces R–R (Scheme 2). This mechanism is ruled out by the con-
trol experiments shown in Eqs 1 and 2. Thus, Oct–MgBr (1a)
was allowed to react with Hex–Br and 1,2-dibromoethane (2)
in the presence of AgOTs (1 mol %) in THF at room temperature
to give a high yield (86%) of Oct–Oct but only a trace amount
(2%) of Oct–Hex (Eq 1), indicating that the homo-coupling of
alkyl Grignard reagent in the presence of 2 is much faster than
the cross-coupling between the Grignard reagent and alkyl bro-
mide. It was also demonstrated by the reaction of Oct–MgBr
with 1,2-dibromoethane (2) (Eq 2) that the oxidation of alkyl
Grignard reagent with 2 giving alkyl bromide is slow.
´
59, 33 (1994). c) M. Moreno-Man˜as, M. Perez, and R. Pleixats, J. Org.
Chem., 61, 2346 (1996). d) K. A. Smith, E. M. Campi, W. R. Jackson,
S. Marcuccio, C. G. M. Naeslund, and G. B. Deacon, Synlett, 1997,
131. e) D. J. Koza and E. Carita, Synthesis, 2002, 2183. f) G. W. Kabalka
and L. Wang, Tetrahedron Lett., 43, 3067 (2002). g) L. M. Klingensmith
and N. E. Leadbeater, Tetrahedron Lett., 44, 765 (2003). h) H. Yoshida, Y.
Yamaryo, J. Ohshita, and A. Kunai, Tetrahedron Lett., 44, 1541 (2003).
´
i) S. Punna, D. D. Dıaz, and M. G. Finn, Synlett, 2004, 2351. j) S.-K. Kang,
T.-H. Kim, and S.-J. Pyun, J. Chem. Soc., Perkin Trans. 1, 1997, 797.
k) S. Yamaguchi, S. Ohno, and K. Tamao, Synlett, 1997, 1199. l) S.
Kanemoto, S. Matsubara, K. Oshima, K. Utimoto, and H. Nozaki, Chem.
Lett., 1987, 5. m) V. Farina, B. Krishnan, D. R. Marshall, and G. P. Roth,
J. Org. Chem., 58, 5434 (1993). n) E. Shirakawa, Y. Nakao, Y. Murota,
and T. Hiyama, J. Organomet. Chem., 670, 132 (2003). o) A. S. Demir,
¨
O. Reis, and M. Emrullahoglu, J. Org. Chem., 68, 10130 (2003).
3
4
5
The combination of a palladium catalyst with an ꢁ-halo carbonyl com-
pound has been reported for the homo-coupling of alkylzinc reagents,
but the yields are not always high for those bearing ꢀ-hydrogen atoms:
A. Lei and X. Zhang, Org. Lett., 4, 2285 (2004).
For the homo-coupling of alkyl Grignard reagents with stoichiometric
oxidants: a) A. MacKillop, L. F. Elsom, and C. E. Taylor, J. Am. Chem.
Soc., 90, 2423 (1968). b) T. Nishiyama, T. Seshita, H. Shodai, K. Aoki,
H. Kameyama, and K. Komura, Chem. Lett., 1996, 549.
´
Recent reviews on alkyl–alkyl coupling reactions: a) D. J. Cardenas,
Angew. Chem., Int. Ed., 38, 3018 (1999). b) T.-Y. Luh, M.-k. Leung,
and K.-T. Wong, Chem. Rev., 100, 3187 (2000). c) D. J. Cardenas,
´
Angew. Chem., Int. Ed., 72, 384 (2003). d) M. R. Netherton and G. C.
Fu, Adv. Synth. Catal., 346, 1525 (2004). e) A. C. Frisch and M. Beller,
Angew. Chem., Int. Ed., 44, 674 (2005).
a) J. H. Gardner and P. Borgstrom, J. Am. Chem. Soc., 51, 3375 (1929).
b) M. Tamura and J. K. Kochi, J. Am. Chem. Soc., 93, 1483 (1971).
c) M. Tamura and J. Kochi, Synthesis, 1971, 303. d) M. Tamura and
J. K. Kochi, Bull. Chem. Soc. Jpn., 45, 1120 (1972). e) G. M. Whitesides
and F. D. Gutowski, J. Org. Chem., 41, 2882 (1976).
Although Tamura and Kochi reported that LiNO3, CH3NO3, and NO2 act
as re-oxidant for the silver-promoted oxidative homo-coupling of EtMgBr
and n-BuMgBr, turnover numbers are quite low (TN = < ca. 3.3): See
Ref. 6d.
T. Nagano and T. Hayashi, Org. Lett., 7, 491 (2005); See also: G. Cahiez,
C. Chaboche, F. Mahuteau-Betzer, and M. Ahr, Org. Lett., 7, 1943 (2005).
Disproportionation of an alkylsilver(I) complex giving alkyl–alkyl cou-
pling product and silver(0) has been reported: G. M. Whitesides, D. E.
Bergbreiter, and P. E. Kendall, J. Am. Chem. Soc., 96, 2806 (1974).
6
7
8
9
Oct−MgBr (1a)
(1.00 mmol)
AgOTs
+
10 The use of 1,2-dichloroethane for oxidation of Pd(0) to Pd(II): a) T.
Tsubomura, A. Itsuki, M. Homma, and K. Sakai, Chem. Lett., 1994,
661. b) K. Ohrai, K. Kondo, M. Sodeoka, and M. Shibasaki, J. Am. Chem.
(1 mol %)
THF, rt,
30 min
Hex−Br
(1.00mmol)
+
+
+
Hex−Br
(1)
(2)
Oct−Oct
Oct−Hex
3
0.43mmol 0.02 mmol 0.93 mmol
Br(CH2)2Br (2)
´
Soc., 116, 11737 (1994). c) S. Aıt-Mohand, F. Henin, and J. Muzart,
¨
2% yield
93% rec.
86% yield
(1.00mmol)
Tetrahedron Lett., 36, 2473 (1995). d) G. Rothenberg, S. Humbel, and
J. Muzart, J. Chem. Soc., Perkin Trans. 2, 2001, 1998, and references cited
therein; One electron oxidation of Cr(II) to Cr(III) by vicinal dibromides
has been reported: D. M. Singleton and J. K. Kochi, J. Am. Chem. Soc., 89,
6547 (1967).
nocat.
THF, rt,
30 min
Oct−MgBr (1a)
+
OctBr
0.14mmol
14% yield
Br(CH2)2Br (2)
(1.00 mmol)
(1.20 mmol)
Published on the web (Advance View) July 16, 2005; DOI 10.1246/cl.2005.1152

 Nagano, Takashi
Nagano, Takashi
 Hayashi, Tamio
Hayashi, Tamio