Full Papers
standard, and the yield of phenylacetic acid derivatives was deter-
mined by H NMR spectroscopy using 1-chloro-2,4-dinitrobenzene
as the internal standard. If required, the resulting carboxylic acid
was purified by conventional flash chromatography.
125.23, 116.72, 116.50, 114.65, 114.44, 40.79 ppm; HRMS (ESI): m/z:
calcd for C8H7FO2: 154.0430 [MÀH+]À; found 153.0351.
1
2-(4-Vinylphenyl)acetic acid (2j): Yield 49%; m.p. 83–858C (lit.[5d]
83–868C); 1H NMR (400 MHz, CDCl3): d=3.64 (s, 2H), 5.24 (d, J=
10.9 Hz, 1H), 5.73 (d, J=17.6 Hz, 1H), 6.70 (dd, J=17.6, 10.9 Hz,
1H), 7.26–7.21 (m, 2H), 7.38 ppm (d, J=8.1 Hz, 2H); 13C NMR
(100.6 MHz, CDCl3): d=177.59, 136.91, 136.47, 132.84, 129.69,
126.61, 114.16, 40.82 ppm; HRMS (ESI): m/z: calcd for C10H10O2:
162.0681 [MÀH+]À; found 161.0610.
2-Phenylacetic acid (2a): Yield 77%; m.p. 77–788C (lit.[5d] 77–
798C); 1H NMR (400 MHz, CDCl3): d=3.65 (s, 2H), 7.25–7.45 ppm
(m, 5H); 13C NMR (100.6 MHz, CDCl3): d=177.85, 133.38, 129.52,
128.80, 127.51, 41.16 ppm; HRMS (ESI): m/z: calcd for C8H8O2:
136.0524 [MÀH+]À; found 135.0456.
2-(Naphthalen-2-yl)acetic acid (2k): Yield 78%; m.p. 140–1428C
2-(4-Methoxyphenyl)acetic acid (2b): Yield 68%; m.p. 83–858C
1
(lit.[5d] 140–1428C); H NMR (400 MHz, CDCl3): d=3.81 (s, 2H), 7.36–
1
(lit.[5d] 83–858C); H NMR (400 MHz, CDCl3): d=3.58 (s, 2H), 3.79 (s,
7.50 (m, 3H), 7.71–7.86 ppm (m, 4H); 13C NMR (100.6 MHz, CDCl3):
d=177.78, 133.53, 132.68, 130.81, 128.48, 128.33, 127.82, 127.44,
126.39, 126.10, 41.31 ppm; HRMS (ESI): m/z: calcd for C12H10O2:
186.0681 [MÀH+]À; found 185.0603.
3H), 6.86 (d, 2H), 7.19 ppm (d, 2H); 13C NMR (100.6 MHz, CDCl3):
d=178.26, 158.99, 130.55, 125.45, 114.21, 55.40, 40.26 ppm; HRMS
(ESI): m/z: calcd for C9H10O3: 166.0630 [MÀH+]À; found 165.0548.
2-(p-Tolyl)acetic acid (2c): Yield 74%; m.p. 90–928C (lit.[13a] 90–
1
1,2-Diphenylethane (4a): M.p. 48–508C (lit.[13c] 48–518C); H NMR
1
918C); H NMR (400 MHz, CDCl3): d=2.33 (s, 3H), 3.61 (s, 2H), 7.12–
(400 MHz, CDCl3): d=2.92 (s, 4H), 7.16–7.22 (m, 4H), 7.24–
7.30 ppm (m, 4H); 13C NMR (100.6 MHz, CDCl3): d=141.92, 128.59,
128.47, 126.05, 38.10 ppm; GC–MS calcd for C14H14 182.11; found
182.15 (25.86%), 91.10 (100%).
7.20 (d, 2H), 7.24–7.30 ppm (d, 2H); 13C NMR (100.6 MHz, CDCl3):
d=178.24, 137.19, 130.31, 129.49, 129.37, 40.72, 21.24 ppm; HRMS
(ESI): m/z: calcd for C9H10O2: 150.0681 [MÀH+]À; found 149.0610.
2-(m-Tolyl)acetic acid (2d): Yield 68%; m.p. 62–638C (lit.[13a] 63–
1
648C); H NMR (400 MHz, CDCl3): d=2.34 (s, 3H), 3.61 (s, 2H), 3.79
Computational details
(s, 3H), 7.05–7.20 (m, 3H), 7.22–7.28 ppm (m, 1H); 13C NMR
(100.6 MHz, CDCl3): d=177.81, 138.50, 133.24, 130.25, 128.69,
128.27, 126.51, 41.07, 21.49 ppm; HRMS (ESI): m/z: calcd for
C9H10O2: 150.0681 [MÀH+]À; found 149.0609.
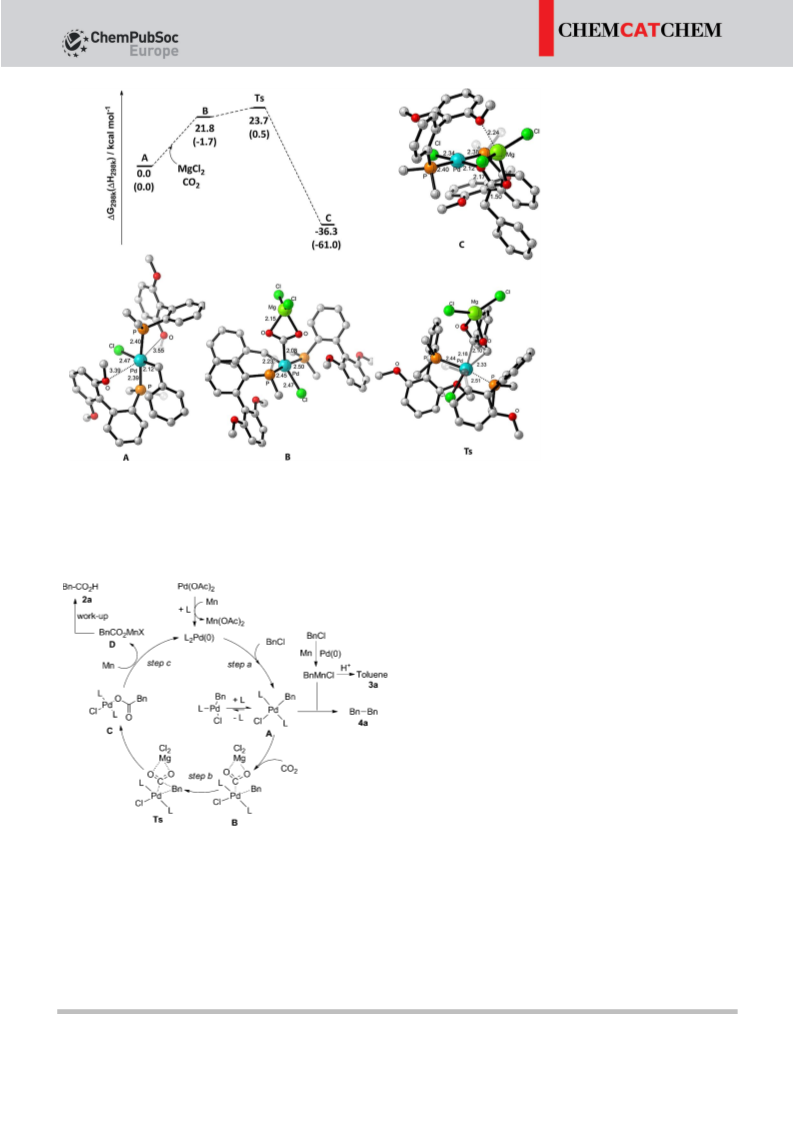
All the calculations were performed by the DFT method[14] by
using the GAUSSIAN 09 package.[15] All the structures reported in
this research were optimized by the M06 method in conjunction
with BSI basis sets, in which BSI signifies basis set LANL2DZ[16a] per-
formed for the Pd atom and basis set 6-31G*[16b] for the other C, H,
O, P, Cl, and Mg atoms. Furthermore, all the final structures were
confirmed by frequency calculations to be energy minima. Energy
calculations were performed at M06/6-311+ +G**[16b–j]/LANL2DZ//
M06/BSI level, in which basis set 6-311+ +G** was employed for
C, H, O, P, Cl, and Mg atoms and basis set LANL2DZ was employed
for the Pd atom. All energy data reported are in kcalmolÀ1, and the
length data were in . Structures were generated using CYLview.[17]
2-[4-(tert-Butyl)phenyl]acetic acid (2e): Yield 66%; m.p. 82–848C
1
(lit.[5d] 83–858C); H NMR (400 MHz, CDCl3): d=1.34 (s, 9H), 1.97 (s,
3
2H), 4.37 (q, J=7.1 Hz, 2H), 7.20–7.28 (m, 2H), 7.34–7.40 ppm (m,
2H); 13C NMR (100.6 MHz, CDCl3): d=177.88, 150.32, 130.40,
129.15, 125.73, 40.67, 31.44 ppm; HRMS (ESI): m/z: calcd for
C12H16O2: 192.1150 [MÀH+]À; found 191.1080.
2-[4-(Ethoxycarbonyl)phenyl]acetic acid (2 f): Yield 88%; m.p.
100–1028C (lit.[13b] 1018C); 1H NMR (400 MHz, CDCl3): d=1.39 (t,
3
3J=7.1 Hz, 2H), 3.71 (s, 2H), 4.37 (q, J=7.1 Hz, 2H), 7.68–7.19 (m,
1H), 8.59–7.65 ppm (m, 1H); 13C NMR (100.6 MHz, CDCl3): d=
176.83, 166.49, 161.71, 138.30, 130.04, 129.78, 129.57, 61.17, 41.03,
14.45 ppm; HRMS (ESI): m/z: calcd for C11H12O4: 208.0736 [MÀH+]À;
found 207.0652.
Acknowledgements
We are grateful to the National Natural Sciences Foundation of
China, the Specialized Research Fund for the Doctoral Program
of Higher Education (20130031110013), the MOE Innovation
Team (IRT13022) of China, and the “111” Project of the Ministry of
Education of China (project No. B06005) for financial support.
2-(4-Acetylphenyl)acetic acid (2g): Yield 86%; m.p. 110–1128C
1
(lit.[5d] 111–1128C); H NMR (400 MHz, CDCl3): d=2.60 (s, 3H), 3.74
(s, 2H), 7.40 (d, 3J=8.6 Hz, 2H), 7.96 ppm (d, 3J=8.3 Hz, 2H).
13C NMR (100.6 MHz, CDCl3): d=197.8, 176.3, 138.6, 136.2, 129.7,
128.7, 40.8, 26.6 ppm; HRMS (ESI): m/z: calcd for C10H10O3:
178.0630 [MÀH+]À; found 177.0560.
Keywords: magnesium · manganese · palladium · phosphane
ligands · reaction mechanisms
2-(2-Chlorophenyl)acetic acid (2h): Yield 92%; m.p. 93–958C
(lit.[5d] 91–938C); 1H NMR (400 MHz, CDCl3): d=3.81 (s, 2H), 7.20–
7.42 ppm (m, 4H); 13C NMR (100.6 MHz, CDCl3): d=177.27, 134.77,
131.76, 131.68, 129.68, 129.12, 127.10, 38.96 ppm; HRMS (ESI): m/z:
calcd for C8H7ClO2: 170.0135; found [MÀH+]À 169.0055,
[MÀCO2ÀH+]À 125.0166.
2-(3-Fluorophenyl)acetic acid (2i): Yield 96%; m.p. 42–448C (lit.[13a]
1
42–438C); H NMR (400 MHz, CDCl3): d=3.65 (s, 3H), 6.98–7.08 (m,
3H), 7.25–7.32 ppm (m, 1H); 13C NMR (100.6 MHz, CDCl3): d=
177.55, 164.16, 161.71, 135.54, 135.46, 130.28, 130.20, 125.26,
ChemCatChem 2015, 7, 3972 – 3977
3976
ꢀ 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Zhang, Shuai
Zhang, Shuai
 Chen, Wei-Qiang
Chen, Wei-Qiang
 Yu, Ao
Yu, Ao
 He, Liang-Nian
He, Liang-Nian