48
J. Mondal et al. / Journal of Molecular Catalysis A: Chemical 350 (2011) 40–48
4. Conclusion
[38] S.A. Patel, K.N. Patel, S. Sinha, B.V. Kamath, J. Mol. Catal. A: Chem. 332 (2010)
70–75.
[39] J. Dupont, R.F. de Souza, P.A.Z. Suarez, Chem. Rev. 102 (2002) 3667–3691.
[40] O. Metin, F. Durap, M. Aydemir, S. Ozkar, J. Mol. Catal. A: Chem. 337 (2011)
39–44.
[41] N. Azizi, F. Aryanasab, L. Torkiyan, A. Ziyaei, M.R. Saidi, J. Org. Chem. 71 (2006)
3634–3635.
In summary we have developed an environment-friendly
catalytic system based on functionalized mesoporous polymer
material grafted with Pd nanoparticle at its surface. Heck C–C
coupling reaction for a variety of aryl and heteroaryl iodides,
bromides and chlorides undergoes very efficiently using this meso-
porous polymer supported palladium catalyst in water as reaction
medium. The method provides green protocol that water as a
solvent/reaction medium prevents environmental concerns. The
catalyst can be recycled without loss of efficiency and thus the strat-
egy described herein has high potential in eco-friendly catalysis.
[42] M. Lamblin, L. Nassar-Hardy, J.C. Hierso, E. Fouquet, F.X. Felpin, Adv. Synth.
[43] D. Chandra, A. Bhaumik, J. Mater. Chem. 19 (2009) 1901–1907.
[44] A.V. Gaikwad, G. Rothenberg, Phys. Chem. Chem. Phys. 8 (2006) 3669–3675.
[45] Q. Liu, J.C. Bauer, R.E. Schaak, J.H. Lunsford, Angew. Chem. Int. Ed. 47 (2008)
6221–6224.
[46] 1H and 13C NMR chemical shifts for different coupling products reported in
Tables 1, 2 and 3.;
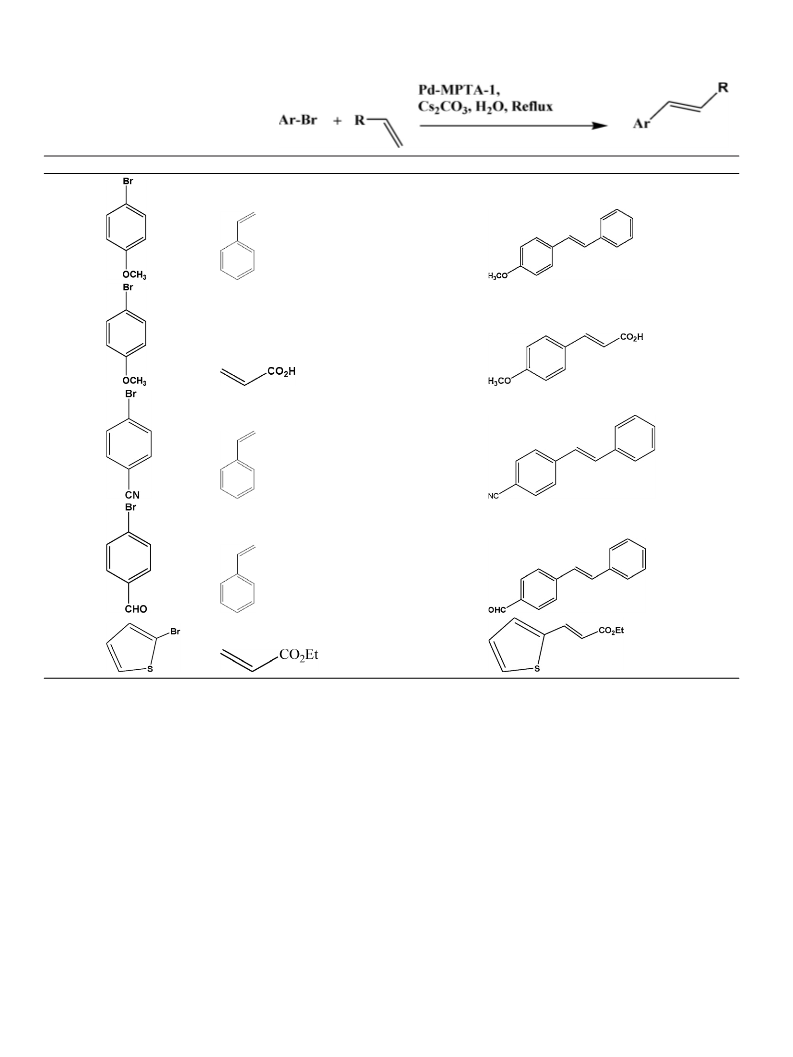
trans-stilbene (table* 1, entry 1): White solid; 1H NMR (300 MHz, CDCl3) ı
7.44 (4H, d, J = 6 Hz), 7.30 (5H, t), 7.19 (3H, t); 13C NMR (300 MHz, CDCl3) ı
137.4, 128.8, 128.2, 127.7, 126.6.;
Acknowledgements
trans-cinnamic acid (table* 1, entry 3): White solid; 1H NMR (500 MHz, CDCl3)
ı 7.74 (1H, d, J = 20 Hz), 7.49 (2H, d, J = 2 Hz), 7.42–7.22 (3H, m), 6.36 (1H, d,
117.4.;
AB wishes to thank DST New Delhi for providing instrumental
facilities through the Nano Mission Initiatives. JM and AM thank
CSIR, New Delhi for their respective senior research fellowships.
trans-4-methoxycinnamic acid (table* 1, entry 4): White solid; 1H NMR (300
MHz, CDCl3) ı 7.70 (1H, d, J = 18 Hz), 7.45 (2H, d, J = 9 Hz), 6.86 (2H, d, J = 9 Hz),
6.27 (1H, d, J = 15 Hz), 3.77 (3H, s); 13C NMR (300 MHz, CDCl3) ı 171.9, 161.8,
146.7, 130.1, 126.9, 114.6, 55.5.;
References
n-Butyl trans-cinnamate;1; (table* 1, entry 5): Colorless oily liquid; 1H NMR
(300 MHz, CDCl3) ı 7.63 (1H, d, J = 15 Hz), 7.46 (2H, d, J = 9.3 Hz), 7.30–7.22 (3H,
m), 6.39 (1H, d, J = 18 Hz), 4.15 (2H, t), 1.66–1.59 (2H, m), 1.37–1.30 (2H, m),
0.91 (3H, t); 13C NMR (300 MHz, CDCl3) ı 167.2, 144.7, 134.5, 129.2, 128.3,
127.9, 118.3, 64.5, 30.8, 19.3, 13.8.;
[1] R.F. Heck Jr., J.P. Nolley, J. Org. Chem. 37 (1972) 2320–2322.
[2] R.F. Heck, Acc. Chem. Res. 12 (1979) 146–151.
[3] A.F. Littke, G.C. Fu, Angew. Chem. Int. Ed. 41 (2002) 4176–4211.
[4] C.F.J. Barnard, Organometallics 27 (2008) 5402–5422.
[5] G.P. McGlacken, L.M. Bateman, Chem. Soc. Rev. 38 (2009) 2447–2464.
[6] H. Kawada, M. Iwamoto, M. Utsugi, M. Miyano, M. Nakada, Org. Lett. 6 (2004)
4491–4494.
[7] G.A. Molander, F. Dehmel, J. Am. Chem. Soc. 126 (2004) 10313–10318.
[8] R. Szabo, M.D. Crozet, P. Vanelle, Synthesis 127 (2008) 127–135.
[9] L.J. Jeffrey, R. Sarpong, Tetrahedron Lett. 50 (2009) 1969–1972.
[10] L.F. Tietze, G. Kettschau, U. Heuschert, G. Nordmann, Chem. Eur. J. 7 (2001)
368–373.
[11] M. Beller, A. Tafesh, W.A. Herrmann, German Patent DE 19503/119 (1996).
[12] S. Durgadas, V.K. Chatare, K. Mukkanti, S. Pal, Appl. Organomet. Chem. 24 (2010)
680–684.
[13] G.T. Crisp, Chem. Soc. Rev. 27 (1998) 427–436.
[14] F. Diederich, P.J. Stang (Eds.), Metal-Catalyzed Cross-Coupling Reactions, Wiley,
New York, 1998.
n-Butyl trans-4-methyl cinnamate (table* 1, entry 6): Pale yellow liquid, 1
H
NMR (300 MHz, CDCl3) ı 7.69 (1H, d, J = 18 Hz), 7.43 (2H, d, J = 12 Hz), 7.26 (2H,
d, J = 18 Hz), 6.42 (2H, d, J = 15 MHZ), 4.23 (2H, t), 2.39 (3H, s), 1.71–1.64 (2H,
m), 1.48–1.38 (2H, m), 0.995.(3H, t).;
trans-4-nitrocinnamic acid;1; (table* 1, entry 7): White solid, 1H NMR (300
MHz, DMSO-d6) ı 8.25 (2H, d, J = 9 Hz), 7.99 (2H, d, J = 9 Hz), 7.71 (1H, d,
J = 15 Hz), 6.76 (1H, d, J = 15 Hz); 13C NMR (300 MHz, DMSO-d6) ı 167.5, 148.5,
141.8, 141.3, 129.8, 124.4, 124.2.;
trans-4-methoxystilbene (table* 1, entry 8): White solid, 1H NMR (500 MHz,
CDCl3) ı 7.09 (1H, d, J = 9 Hz), 7.001 (1H, d, J = 9.6 Hz), 6.919 (2H, d, J = 8.5 Hz),
7.508 (2H, d, J = 9 Hz), 7.473 (2H, d, J = 8.5 Hz), 7.368 (2H, t), 7.260 (1H, t), 3.838
(3H, s); 13C NMR (500 MHz, CDCl3) ı 159.4, 137.7, 130.3, 128.7, 128.3, 127.8,
127.3, 126.7, 126.3, 114.2, 55.4.;
trans-4-methylstilbene (table* 1, entry 9): 1H NMR (300 MHz, CDCl3) ı 7.43
(2H, d, J = 7.2 Hz), 7.34 (2H, d, J = 6 Hz), 7.10 (2H, d, J = 9 Hz), 7.32 (3H, t), 7.18
(2H, d, J = 6 Hz), 2.28.(3H,;1; s).;
[15] D. Astruc, F. Lu, J.R. Aranzaes, Angew. Chem. Int. Ed. 44 (2005) 7852–7872.
[16] N.T.S. Phan, M. Van Der Sluys, C.W. Jones, Adv. Synth. Catal. 348 (2006)
609–679.
trans-4-nitrostilbene (table* 1, entry 10): Pale yellow solid, 1H NMR (300
MHz, CDCl3) ı 8.23 (2H, d, J = 6 Hz), 7.65 (2H, d, J = 9 Hz), 7.56 (2H, d, J = 6 Hz),
7.42-7.33 (4H, m), 7.17 (1H, d, J = 18 Hz); 13C NMR (300 MHz, CDCl3) ı 146.8,
143.9, 133.2, 133.4, 128.9, 128.9, 127.1, 126.9, 126.3, 124.2.;
[17] G.J. Hutchings, J. Mater. Chem. 19 (2009) 1222–1235.
[18] Y. Kume, K. Qiao, D. Tomida, C. Yokoyama, Catal. Commun.
369–375.
9 (2008)
trans-4-methylcinnamic acid (table* 1, entry 11): White solid, 1H NMR (300
MHz, CDCl3) ı 7.79 (1H, d, J = 20 Hz), 7.46 (2H, d, J = 10 Hz), 7.26 (2H, d, J = 20 Hz),
6.42 (1H, d, J = 15 Hz), 2.98 (3H, s); 13C NMR (300 MHz, CDCl3) ı 172.5, 147.2,
141.4, 131.5, 129.8, 128.5, 116.3, 21.6.;
[19] F. Lang, H.R.A. May, B.L. Iversen, B.D. Chandler, J. Am. Chem. Soc. 125 (2003)
14832–14836.
[20] K. Sarkar, M. Nandi, M. Islam, T. Mubarak, A. Bhaumik, Appl. Catal. A: Gen. 352
(2009) 81–86.
[21] R. Bernini, S. Cacchi, G. Fabrizi, G. Forte, F. Petrucci, A. Prastaro, S. Niembro, A.
Shafir, A. Vallribera, Green Chem. 12 (2010) 150–158.
[22] S. Ungureanu, H. Deleuze, O. Babot, M.F. Achard, C. Sanchez, M.I. Popa, R. Backov,
Appl. Catal. A: Gen. 390 (2010) 51–58.
[23] L.M. Neal, H.E. Hagelin-Weaver, J. Mol. Catal. A: Chem. 284 (2008) 141–148.
[24] K.E. Price, D.T. McQuade, Chem. Commun. (2005) 1714–1716.
[25] D.D. Das, A. Sayari, J. Catal. 246 (2007) 60–65.
[26] J. Demel, S.E. Sujandi, J. Park, P. Cejka, Stepnicka, J. Mol. Catal. A: Chem. 302
(2009) 28–35.
[27] S.L. Jain, B.S. Rana, B. Singh, A.K. Sinha, A. Bhaumik, M. Nandi, B. Sain, Green
Chem. 12 (2010) 374–377.
[28] A. Modak, J. Mondal, V.K. Aswal, A. Bhaumik, J. Mater. Chem. 20 (2010)
8099–8106.
[29] K.M. Parida, D. Rath, S.S. Dash, J. Mol. Catal. A: Chem. 318 (2010) 85–93.
[30] Y. Meng, D. Gu, F. Zhang, Y.F. Shi, L. Cheng, D. Feng, Z.X. Wu, Z.X. Chen, Y. Wan,
A. Stein, D. Zhao, Chem. Mater. 18 (2006) 4447–4464.
[31] D. Chandra, B.K. Jena, C.R. Raj, A. Bhaumik, Chem. Mater. 19 (2007) 6290–6296.
[32] M. Nandi, R. Gangopadhyay, A. Bhaumik, Micropor. Mesopor. Mater. 109 (2008)
239–247.
[33] J. Schuster, R. Koehn, A. Keilbach, M. Doeblinger, H. Amenitsch, T. Bein, Chem.
Mater. 21 (2009) 5754–5762.
[34] Y.L. Zhang, S. Wei, F.J. Liu, Y.C. Du, S. Liu, Y.Y. Ji, T. Yokoi, T. Tatsumi, F.S. Xiao,
Nano Today 4 (2009) 135–142.
[35] J.K. Cho, R. Najman, T.W. Dean, O. Ichihara, C. Muller, M. Bradley, J. Am. Chem.
Soc. 128 (2006) 6276–6277.
[36] B. Karimi, D. Enders, Org. Lett. 8 (2006) 1237–1240.
[37] H. Zhao, G.M. Zheng, W.Y. Hao, M.Z. Cai, Appl. Organomet. Chem. 24 (2010)
92–98.
3-pyridyl styrene (table* 1, entry 12): Brown colored solid, 1H NMR (300 MHz,
CDCl3) ı 8.69 (1H, s), 8.46 (1H, d, J = 3 Hz), 7.78 (1H, d, J = 9 Hz), 7.50 (2H, d,
J = 6 Hz), 7.37 (2H, t), 7.29–7.21 (2H, m), 7.15 (1H, d, J = 18 Hz), 7.04 (1H, d,
J = 18 Hz); 13C NMR (300 MHz, CDCl3) ı 148.4, 136.5, 132.9, 132.6, 130.8, 128.7,
128.1, 126.6, 124.7, 123.5.;
trans-3-fluorostilbene (table* 1, entry 13): Brownish white solid, 1H NMR
(300 MHz, CDCl3) ı 7.55 (2H, d, J = 9 Hz), 7.41-7.30 (7H, m), 7.11 (1H, d, J = 6 Hz),
6.97 (1H, s); 13C NMR (300 MHz, CDCl3) ı 161.7, 139.9, 136.9, 130.4, 128.8,
128.1, 127.6, 126.8, 126.6, 114.6, 113.0.;
trans-4-formylstilbene (table* 2, entry 4): White solid, 1H NMR (300 MHz,
DMSO-d6) ı 9.96 (1H, s), 7.90 (2H, d, J = 9 Hz), 7.81 (2H, d, J = 9 Hz), 7.65 (2H,
136.4, 135.0, 131.9, 129.9, 128.7, 128.3, 127.2, 126.9, 126.9.;
trans-4-cyanostilbene (table* 2, entry 3): Grey solid, 1H NMR (300 MHz,
CDCl3) ı 7.18 (1H, d, J = 12 Hz), 7.25 (1H, d, J = 3 Hz), 7.40–7.31 (2H, m), 7.81
(1H, t), 7.69–7.51 (6H, m); 13C NMR (300 MHz, CDCl3) ı 141.9, 136.4, 132.6,
132.0, 129.0, 128.7, 126.9, 126.8, 119.1, 110.6.;
2-thiophenyl ethylacrylate (table* 2, entry 5): Brown colored liquid, 1H NMR
(300 MHz, CDCl3) ı 7.21 (2H, d, J = 6 Hz), 7.04 (1H, t), 6.90 (1H, d, J = 18 Hz), 6.66
(1H, d, J = 9 Hz), 3.75 (2H, q), 1.25 (3H, t); 13C NMR (300 MHz, CDCl3) ı 167.2,
144.7, 134.5, 130.3, 129.2, 128.1, 127.9, 64.5, 13.8.
[47] S. Mukhopadhyay, G. Rothenberg, A. Joshi, M. Baidossi, Y. Sasson, Adv. Synth.
Catal. 344 (2002) 348–354.
[48] Z. Zhang, Z. Zha, C. Gan, C. Pan, Y. Zhou, Z. Wang, M.-M. Zhou, J. Org. Chem. 71
(2006) 4339–4342.
[49] C. Amatore, A. Jutand, Acc. Chem. Res. 33 (2000) 314–321.
[50] J.A. Fritz, J.P. Wolfe, Tetrahedron 64 (2008) 6838–6852.
[51] J.G. de Vries, Dalton Trans. (2006) 421–429.

 Mondal, John
Mondal, John
 Modak, Arindam
Modak, Arindam
 Bhaumik, Asim
Bhaumik, Asim