Typical procedure for the reaction of indium enolates and
carbonyl compounds
CH(t-Bu)), 3.20–3.30 (1 H, m, OH), 4.13 (2 H, q, J 7.2,
OCH2Me), 3.90–3.98 (1 H, m, CHOH); minor isomer: δH 0.88
(3 H, t, J 6.9, Me), 1.07 (9 H, s, 3 × Me), 1.25–1.32 (15 H, m,
(CH2)6 and OCH2Me), 2.21 (1 H, d, J 1.8, CH(t-Bu)), 3.20–3.30
(1 H, m, OH), 4.20 (2 H, q, J 7.2, OCH2Me), 3.82–3.89 (1 H,
m, CHOH) (Found C, 70.62; H, 12.08. C16H32O3 requires C,
70.54; H, 11.84%).
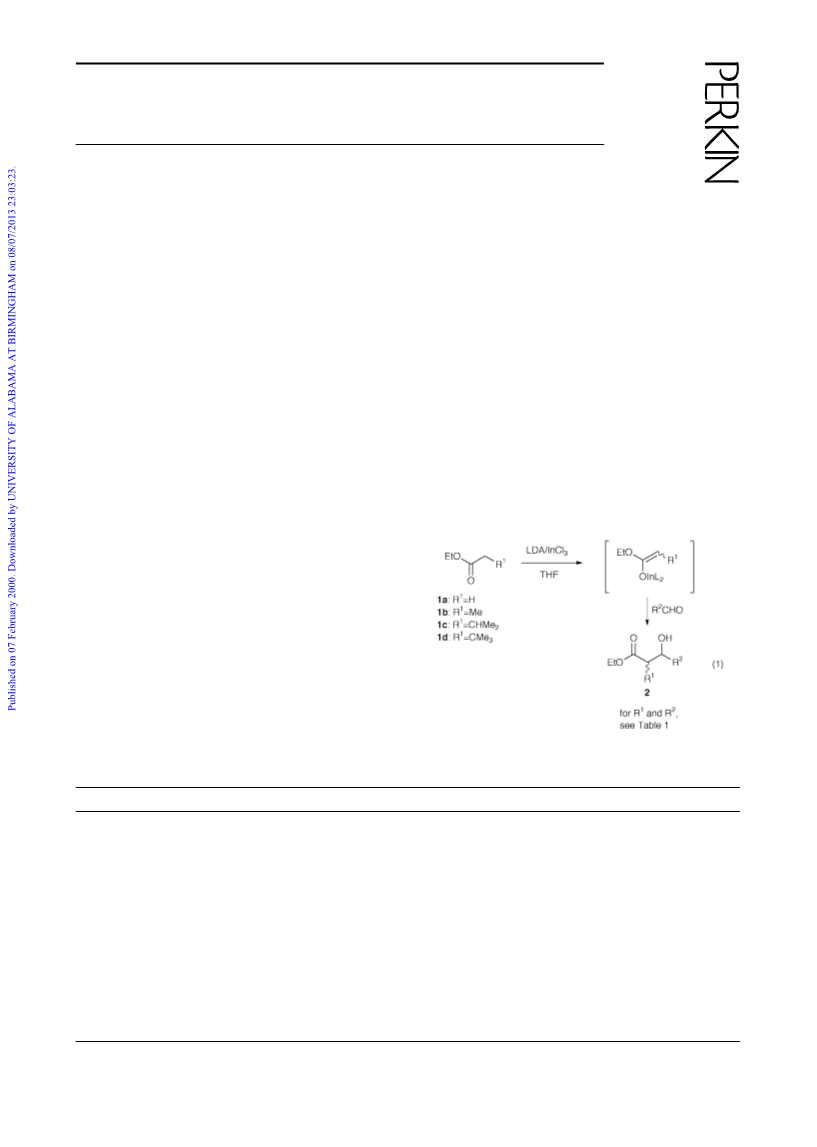
The following reaction (entry 3 in Table 1) represents the
general procedure. Lithium dicyclohexylamide, prepared from
dicyclohexylamine (0.6 mL, 3.0 mmol) and n-butyllithium
(1.6 M, 2.0 mL, 3.1 mmol) in THF (4 mL) was added to a
stirred solution of anhydrous indium trichloride (221 mg, 1.0
mmol) and ethyl propanoate (344 µL, 3.0 mmol) in THF (6 mL)
at Ϫ78 ЊC. The mixture was stirred for 10 min, after which
time benzaldehyde (152 µL, 1.5 mmol) was added. The reaction
mixture was warmed to room temperature and left overnight.
The reaction was quenched with 1 M HCl (6 mL), and the
product was extracted with diethyl ether. The organic extracts
were washed with water and brine, and concentrated. The
residue was purified by column chromatography on silica gel
(elution with hexane–EtOAc, 9:1) to give ethyl 3-hydroxy-2-
methyl-3-phenylpropanoate (2c)6 (245 mg, 79%; syn:anti,
66:34).
Ethyl (E)-2-tert-butyl-3-hydroxy-5-phenylpent-4-enoate (2l).
(Entry 13 in Table 1): νmax(film)/cmϪ1: 3450, 2960, 1724, 1630,
1600, 1580, 1496, 1480, 1445, 1396, 1368, 1330, 1254, 1194,
1152, 1112, 1100, 1072, 1028, 970, 748, 696; a mixture of syn
and anti isomers: δH 1.11 (9 H, s, 3 × Me), 1.12 (9 H, s, 3 × Me),
1.20 (3 H, t, J 7.1, OCH2Me), 1.23 (3 H, t, J 7.2, OCH2Me),
2.42 (1 H, d, J 3.6, CH(t-Bu)), 2.57 (1 H, d, J 7.8, CH(t-Bu)),
3.46 (1 H, br s, OH), 4.08 (2 H, q, J 7.1, OCH2Me), 4.16 (2 H,
q, J 7.2, OCH2Me), 4.59–4.69 (1 H, m, CHOH), 6.18 (1 H, dd,
J 15.9 and 5.4, PhCH᎐CH), 6.38 (1 H, dd, J 15.9 and 7.8,
᎐
PhCH᎐CH), 6.59 (1 H, d, J 15.9, PhCH᎐), 6.63 (1 H, d, J 15.9,
The products 2a,2 2b,2 2e,7 2f,8 2g9 and 2j9 are known
compounds.
᎐
᎐
PhCH᎐), 7.20–7.42 (5 H, m, Ph) (Found C, 74.09; H, 8.94.
᎐
C17H24O3 requires C, 73.88; H, 8.75%).
The products 4a,10 4b,11 4c,12 and 4d13 were known com-
pounds.
Ethyl
3-hydroxy-3-(2-hydroxyphenyl)-2-methylpropanoate
(2d). (Entry 5 in Table 1): νmax(film)/cmϪ1: 3370, 2990, 1706,
1610, 1582, 1490, 1458, 1378, 1344, 1242, 1190, 1100, 1052,
1020, 930, 860, 754, 732; major isomer: δH 1.26 (3 H, d, J 7.2,
Me), 1.27 (3 H, t, J 7.2, Me), 3.02 (1 H, qd, J 9.0 and 7.2,
CHMe), 4.18 (2 H, q, J 7.2, OCH2Me), 4.18 (1 H, d, J 3.6,
CHOH), 4.89 (1 H, dd, J 9.0 and 3.6, CHOH), 6.80–6.99 (3 H,
m, aromatic), 7.13–7.23 (1 H, m, aromatic), 7.88 (1 H, d, J 1.8,
OH); minor isomer: δH 1.04 (3 H, d, J 7.2, Me), 1.31 (3 H, t,
J 7.2, Me), 2.81 (1 H, qd, J 7.2 and 3.2, CHMe), 4.21 (1 H, d,
J 2.6, CHOH), 4.23 (2 H, q, J 7.2, OCH2Me), 5.33 (1 H, dd,
J 3.2 and 2.6, CHOH), 6.80–6.99 (3 H, m, aromatic), 7.13–7.23
(1 H, m, aromatic), 8.53 (1 H, s, OH) (Found: C, 64.49; H, 7.01.
C12H16O4 requires C, 64.27; H, 7.19%).
The reaction of the lithium enolate of ethyl propanoate with
benzaldehyde
Lithium dicyclohexylamide, prepared from dicyclohexylamine
(0.6 mL, 3.0 mmol) and n-butyllithium (1.6 M, 2.0 mL,
3.1 mmol) in THF (4 mL) was added to a stirred solution
of ethyl propanoate (344 µL, 3.0 mmol) and benzaldehyde
(152 µL, 1.5 mmol) in THF (6 mL), at Ϫ78 ЊC. The reaction
mixture was stirred for 3 h at this temperature and was
subsequently quenched with saturated aqueous ammonium
chloride. After the white precipitate had been removed by
filtration, the product was extracted with diethyl ether. The
organic extracts were washed with water and brine, and con-
centrated. The residue was purified by column chromatography
on silica gel (elution with hexane–dichloromethane, 1:1 and
then dichloromethane) to give 2c (198 mg, 63% yield; syn:anti,
50:50).
Ethyl 3-hydroxy-2-isopropyldecanoate (2h). (Entry 9 in
Table 1): νmax(film)/cmϪ1: 3450, 2940, 1725, 1462, 1368, 1350,
1238, 1180, 1160, 1120, 1096, 1030, 720; a mixture of syn
and anti isomers: δH 0.85–1.03 (9 H, m, 3 × Me), 1.25–1.31
(15 H, m, (CH2)6 and OCH2Me), 2.11 (1 H, m, CH(i-Pr),
minor isomer), 2.16 (1 H, m, CHMe2), 2.33 (1 H, dd, J 6.9
and 6.0, CH(i-Pr), major isomer), 2.60–2.80 (1 H, br s,
OH), 3.70–3.80 (1 H, m, CHOH, minor isomer), 3.86–3.92
(1 H, m, CHOH, major isomer), 4.13–4.22 (2 H, m, OCH2Me)
(Found: C, 69.61; H, 11.93. C15H30O3 requires C, 69.72; H,
11.70%).
The reaction of the lithium enolate of methyl bromoacetate
with benzaldehyde was carried out similarly, and oxirane 4a
(35% yield, cis:trans, 27:73) and methyl 2-bromo-3-hydroxy-3-
phenylpropanoate14 (26% yield, syn 100%) were obtained.
The self-coupling reaction of 5 [eqn. (3)]
To a stirred mixture of anhydrous indium trichloride (221 mg,
1.0 mmol) and 5 (597 mg, 3.0 mmol) in THF (6 mL), lithium
dicyclohexylamide prepared from dicyclohexylamine (0.6 mL,
3.0 mmol) and n-butyllithium (1.6 M, 2.0 mL, 3.1 mmol) in
THF (4 mL), was added at Ϫ78 ЊC. The mixture was stirred for
10 min and benzaldehyde (152 µL, 1.5 mmol) was added. The
reaction mixture was warmed to room temperature and the
reaction was continued at room temperature for 3 h. The
mixture was quenched with 1 M HCl (6 mL), and the product
was extracted with diethyl ether. The organic extracts were
washed with water and brine, and concentrated. The residue
was purified by column chromatography on silica gel (elution
with hexane–EtOAc, 9:1) to give 4-bromo-2,3-epoxy-1,3-
diphenylbutan-1-one (6)15 (462 mg, 97%, cis:trans, 64:36). The
unreacted benzaldehyde was recovered.
Ethyl (E)-3-hydroxy-2-isopropyl-5-phenylpent-4-enoate (2i).
(Entry 10 in Table 1): νmax(film)/cmϪ1: 3450, 2960, 1725, 1600,
1578, 1495, 1460, 1445, 1390, 1370, 1300, 1240, 1180, 1160,
1098, 1026, 968, 750, 694; a mixture of syn and anti isomers:
δH 0.97 (3 H, d, J 6.6, Me), 1.00 (3 H, d, J 6.9, Me), 1.02 (3 H,
d, J 6.9, Me), 1.08 (3 H, d, J 6.6, Me), 1.22 (3 H, t, J 7.2,
OCH2Me), 1.25 (3 H, t, J 7.2, OCH2Me), 2.11–2.24 (1 H,
m, OH), 2.11–2.24 (1 H, m, CHMe2), 2.32 (1 H, dd, J 8.4 and
4.8, CH(i-Pr)), 2.53 (1 H, dd, J 6.9 and 6.6, CH(i-Pr)), 3.09
(1 H, s, OH), 4.12 (2 H, q, J 7.2, OCH2Me), 4.15 (2 H, q,
J 7.2, OCH2Me), 4.50–4.62 (1 H, m, CHOH), 6.18 (1 H, dd,
J 15.9 and 5.4, PhCH᎐CH), 6.35 (1 H, dd, J 15.9 and 7.2,
᎐
PhCH᎐CH), 6.64 (1 H, d, J 15.9, PhCH᎐), 6.66 (1 H, d, J 15.9,
᎐
᎐
PhCH᎐), 7.20–7.41 (5 H, m, Ph) (Found C, 72.53; H, 8.53.
᎐
C16H22O3 requires C, 73.25; H, 8.45%).
The cross-coupling reaction of 5 with benzaldehyde [eqn. (4)]
Ethyl 2-tert-butyl-3-hydroxydecanoate (2k). (Entry 12 in
Table 1): νmax(film)/cmϪ1: 3460, 2950, 1730, 1710, 1465, 1398,
1370, 1348, 1300, 1260, 1218, 1200, 1155, 1096, 1026, 720;
major isomer: δH 0.88 (3 H, t, J 6.9, Me), 1.08 (9 H, s, 3 × Me),
1.25–1.32 (15 H, m, (CH2)6 and OCH2Me), 2.31 (1 H, d, J 8.4,
This reaction was performed as described above by changing
the addition of lithium dicyclohexylamide to a mixture of
indium trichloride,
5 and benzaldehyde. 2,3-Epoxy-1,3-
diphenylpropan-1-one (7)16 (258 mg, 77%, trans 100%) was
obtained.
J. Chem. Soc., Perkin Trans. 1, 2000, 825–828
827

 Hirashita, Tsunehisa
Hirashita, Tsunehisa
 Kinoshita, Kenji
Kinoshita, Kenji
 Yamamura, Hatsuo
Yamamura, Hatsuo
 Kawai, Masao
Kawai, Masao
 Araki, Shuki
Araki, Shuki