Organic Letters
Letter
7153−7154. (g) Kobayashi, S.; Ishitani, H.; Ueno, M. J. Am. Chem.
Soc. 1998, 120, 431−432. (h) Xue, S.; Yu, S.; Deng, Y.; Wulff, W. D.
Angew. Chem., Int. Ed. 2001, 40, 2271−2274. (i) Kobayashi, S.;
Matsubara, R.; Nakamura, Y.; Kitagawa, H.; Sugiura, M. J. Am. Chem.
L. Angew. Chem., Int. Ed. 2016, 55, 48−57. For other representative
examples, see: (b) Zhu, S.; Niljianskul, N.; Buchwald, S. L. J. Am.
Chem. Soc. 2013, 135, 15746−15749. (c) Zhu, S.; Niljianskul, N.;
Buchwald, S. L. Nat. Chem. 2016, 8, 144−150. (d) Wang, H.; Yang, J.
C.; Buchwald, S. L. J. Am. Chem. Soc. 2017, 139, 8428−8431.
(e) Guo, S.; Yang, J. C.; Buchwald, S. L. J. Am. Chem. Soc. 2018, 140,
15976−15984. (f) Ichikawa, S.; Zhu, S.; Buchwald, S. L. Angew.
Chem., Int. Ed. 2018, 57, 8714−8718. (g) Dai, X.-J.; Engl, O. D.;
́
Soc. 2003, 125, 2507−2515. (j) Cordova, A. Acc. Chem. Res. 2004, 37,
102−112. (k) List, B. J. Am. Chem. Soc. 2000, 122, 9336−9337.
(l) Matsunaga, S.; Yoshida, T.; Morimoto, H.; Kumagai, N.;
Shibasaki, M. J. Am. Chem. Soc. 2004, 126, 8777−8785. (m) West-
ermann, B.; Neuhaus, C. Angew. Chem., Int. Ed. 2005, 44, 4077−4079.
For a representative example of asymmetric Mannich reactions to
obtain β-amino acids, see: (n) Wenzel, A. G.; Jacobsen, E. N. J. Am.
Chem. Soc. 2002, 124, 12964−12965. For a representative example of
asymmetric Mannich reactions to obtain β-amino aldehyde, see:
(o) Chen, Y. M.; Yoshida, M.; MacMillan, D. W. C. J. Am. Chem. Soc.
2006, 128, 9328−9329.
́
Leon, T.; Buchwald, S. L. Angew. Chem., Int. Ed. 2019, 58, 3407−
3411. (h) Ichikawa, S.; Dai, X.-J.; Buchwald, S. L. Org. Lett. 2019, 21,
4370−4373.
(11) (a) Shi, S.-L.; Wong, Z. L.; Buchwald, S. L. Nature 2016, 532,
353−356. (b) Bandar, J. S.; Ascic, E.; Buchwald, S. L. J. Am. Chem.
Soc. 2016, 138, 5821−5824. (c) Yang, Y.; Perry, I. B.; Lu, G.; Liu, P.;
Buchwald, S. L. Science 2016, 353, 144−150.
(12) For representative examples of the use of unprotected allylic
alcohols in nonenantioselective hydroamination, see: (a) Nakamura,
Y.; Ohta, T.; Oe, Y. Chem. Commun. 2015, 51, 7459−7462. (b) Ma,
W.; Zhang, X.; Fan, J.; Liu, Y.; Tang, W.; Xue, D.; Li, C.; Xiao, J.;
Wang, C. J. Am. Chem. Soc. 2019, 141, 13506−13515.
(13) (a) Bandar, J. S.; Pirnot, M. T.; Buchwald, S. L. J. Am. Chem.
Soc. 2015, 137, 14812−14818. (b) Thomas, A. A.; Speck, K.;
Kevlishvili, I.; Lu, Z.; Liu, P.; Buchwald, S. L. J. Am. Chem. Soc. 2018,
140, 13976−13984. (c) Lu, G.; Liu, R. Y.; Yang, Y.; Fang, C.;
Lambrecht, D. S.; Buchwald, S. L.; Liu, P. J. Am. Chem. Soc. 2017, 139,
16548−16555.
(14) Under the optimized reaction conditions, the starting material
allylic alcohol bearing thiophene (1f) was fully converted. We
speculated that the decomposition of 1f could proceed via a
dearomatization process caused by the 1,3-shift of copper to the 3
position of a thiophene ring.
(4) For representative examples, see: (a) Kochi, T.; Tang, T. P.;
Ellman, J. A. J. Am. Chem. Soc. 2002, 124, 6518−6519. (b) Kochi, T.;
Tang, T. P.; Ellman, J. A. J. Am. Chem. Soc. 2003, 125, 11276−11282.
For representative examples of diastereoselective reduction methods
of N-sulfinyl β-amino ketones, see: (c) Davis, F. A.; Gaspari, P. M.;
Nolt, B. M.; Xu, P. J. Org. Chem. 2008, 73, 9619−9626.
(5) For representative examples of enantioselective reduction
methods of β-amino ketones to obtain γ-amino alcohols, see:
(a) Liu, D.; Gao, W.; Wang, C.; Zhang, X. Angew. Chem., Int. Ed.
2005, 44, 1687−1689. (b) Hu, Q.; Zhang, Z.; Liu, Y.; Imamoto, T.;
Zhang, W. Angew. Chem., Int. Ed. 2015, 54, 2260−2264. For an
example of asymmetric synthesis of γ-amino alcohols from β-
ketoenamides, see: (c) Geng, H.; Zhang, W.; Chen, J.; Hou, G.;
Zhou, L.; Zou, Y.; Wu, W.; Zhang, X. Angew. Chem., Int. Ed. 2009, 48,
6052−6054. For a representative example of diastereoselective
reduction methods of β-amino ketones to obtain γ-amino alcohols,
see: (d) Verkade, J. M. M.; Quaedflieg, P. J. L. M.; Verzijl, G. K. M.;
Lefort, L.; van Delft, F. L.; de Vries, J. G.; Rutjes, F. P. J. T. Chem.
Commun. 2015, 51, 14462−14464. (e) Bartoli, G.; Bartolacci, M.;
Cortese, M.; Marcantoni, E.; Massaccesi, M.; Pela, R.; Sambri, L. Eur.
J. Org. Chem. 2004, 2004, 2359−2366. (f) Barluenga, J.; Aguilar, E.;
Fustero, S.; Olano, B.; Viado, A. L. J. Org. Chem. 1992, 57, 1219−
1223. (g) Hayashi, Y.; Urushima, T.; Shin, M.; Shoji, M. Tetrahedron
2005, 61, 11393−11404. (h) Keck, G. E.; Truong, A. P. Org. Lett.
2002, 4, 3131−3134. For other examples of diastereoselective
reduction methods to obtain γ-amino alcohols, see: (i) Menche, D.;
Arikan, F.; Li, J.; Rudolph, S. Org. Lett. 2007, 9, 267−270.
(6) Spreider, P. A.; Haydl, A. M.; Heinrich, M.; Breit, B. Angew.
Chem., Int. Ed. 2016, 55, 15569−15573.
(7) For representative examples, see: (a) Xie, Y.; Yu, K.; Gu, Z. J.
Org. Chem. 2014, 79, 1289−1302. (b) Lee, J. S.; Kim, D.; Lozano, L.;
Kong, S. B.; Han, H. Org. Lett. 2013, 15, 554−557. (c) Broustal, G.;
Ariza, X.; Campagne, J.-M.; Garcia, J.; Georges, Y.; Marinetti, A.;
Robiette, R. Eur. J. Org. Chem. 2007, 2007, 4293−4297.
(8) For representative examples of C−H amination-based stereo-
selective synthesis of γ-amino alcohols, see: (a) Rice, G. T.; White, M.
C. J. Am. Chem. Soc. 2009, 131, 11707−11711. (b) Paradine, S. M.;
White, M. C. J. Am. Chem. Soc. 2012, 134, 2036−2039. (c) Qi, X.;
Rice, G. T.; Lall, M. S.; Plummer, M. S.; White, M. C. Tetrahedron
2010, 66, 4816−4826. (d) Nahra, F.; Liron, F.; Prestat, G.; Mealli, C.;
Messaoudi, A.; Poli, G. Chem. - Eur. J. 2009, 15, 11078−11082.
(e) Zalatan, D. N.; Du Bois, J. J. Am. Chem. Soc. 2008, 130, 9220−
9221. (f) Milczek, E.; Boudet, N.; Blakey, S. Angew. Chem., Int. Ed.
2008, 47, 6825−6828. (g) Liang, J.-L.; Yuan, S.-X.; Huang, J.-S.; Yu,
W.-Y.; Che, C.-M. Angew. Chem., Int. Ed. 2002, 41, 3465−3468.
(9) For an example of asymmetric synthesis of γ-amino alcohols by
hydroxy-directed aziridination and aziridine hydrosilylation, see:
(a) Zhang, Y.-Q.; Bohle, F.; Bleith, R.; Schnakenburg, G.; Grimme,
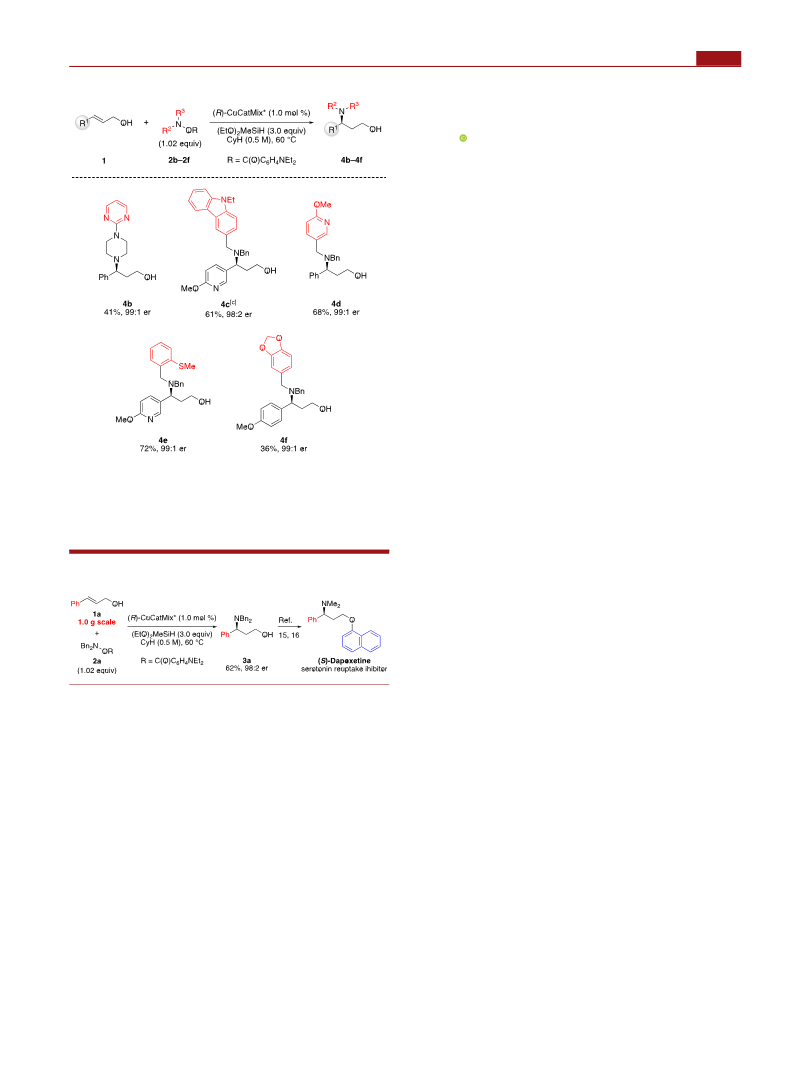
(15) For representative examples for asymmetric synthesis of (S)-
́
dapoxetine, see: (a) Torre, O.; Gotor-Fernandez, V.; Gotor, V.
Tetrahedron: Asymmetry 2006, 17, 860−866. (b) Sasikumar, M.;
Nikalje, M. D. Synth. Commun. 2012, 42, 3061−3067. (c) Kim, S. J.;
Jeon, T. H.; Min, I. S.; Kim, I. S.; Jung, Y. H. Tetrahedron Lett. 2012,
53, 3680−3682. (d) Kang, S.; Lee, H.-K. J. Org. Chem. 2010, 75, 237−
240.
(16) For representative examples of methods to cleave the benzyl
protecting groups, see: (a) Takata, T.; Hirano, K.; Miura, M. Org. Lett.
2019, 21, 4284−4288. (b) Lemoine, R. C.; Petersen, A. C.; Setti, L.;
Baldinger, T.; Wanner, J.; Jekle, A.; Heilek, G.; deRosier, A.; Ji, C.;
Rotstein, D. M. Bioorg. Med. Chem. Lett. 2010, 20, 1674−1676.
(c) Cociorva, O. M.; Li, B.; Nomanbhoy, T.; Li, Q.; Nakamura, A.;
Nakamura, K.; Nomura, M.; Okada, K.; Seto, S.; Yumoto, K.;
Liyanage, M.; Zhang, M. C.; Aban, A.; Leen, B.; Szardenings, A. K.;
Rosenblum, J. S.; Kozarich, J. W.; Kohno, Y.; Shreder, K. R. Bioorg.
Med. Chem. Lett. 2011, 21, 5948−5951. (d) Ok, D.; Fisher, M. H.;
Wyvratt, M. J.; Meinke, P. T. Tetrahedron Lett. 1999, 40, 3831−3834.
(e) Saito, F.; Trapp, N.; Bode, J. W. J. Am. Chem. Soc. 2019, 141,
5544−5554.
̈
S.; Gansauer, A. Angew. Chem., Int. Ed. 2018, 57, 13528−13532. For
other methods for the asymmetric synthesis of 1,3-amino alcohols,
see: (b) Aschwanden, P.; Geisser, R. W.; Kleinbeck, F.; Carreira, E.
M. Org. Lett. 2005, 7, 5741−5742.
(10) For a minireview on the copper-catalyzed asymmetric
hydroamination, see: (a) Pirnot, M. T.; Wang, Y.-M.; Buchwald, S.
D
Org. Lett. XXXX, XXX, XXX−XXX

 Ichikawa, Saki
Ichikawa, Saki
 Buchwald, Stephen L.
Buchwald, Stephen L.