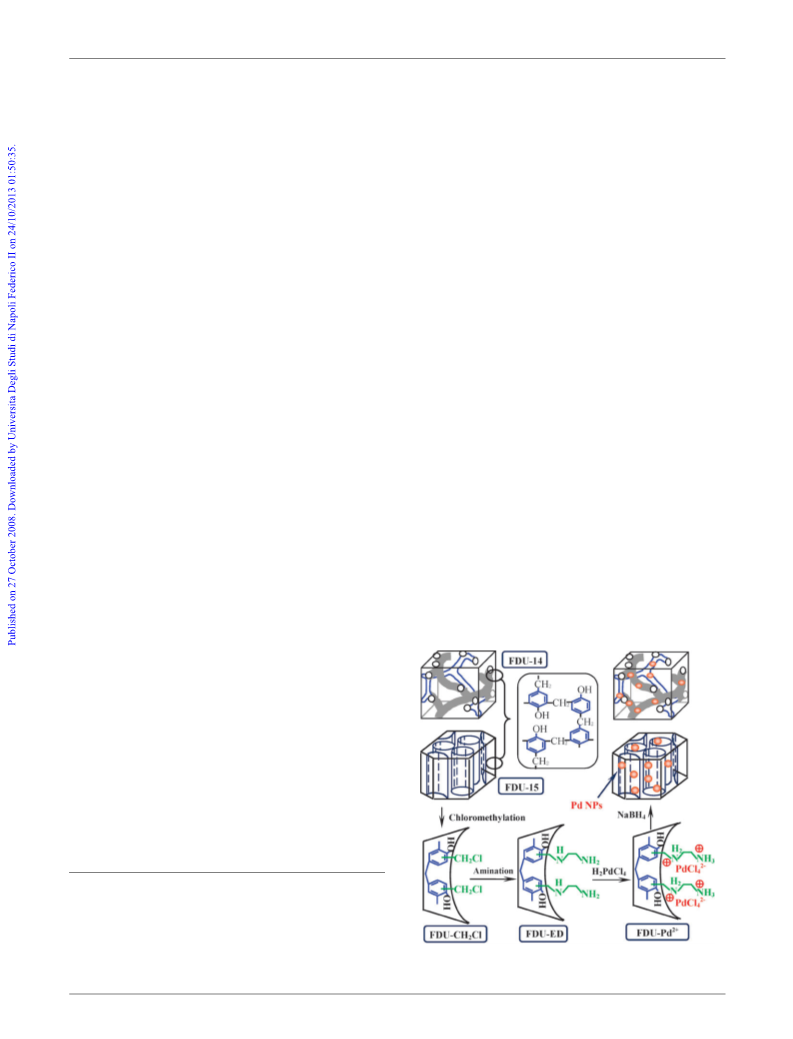
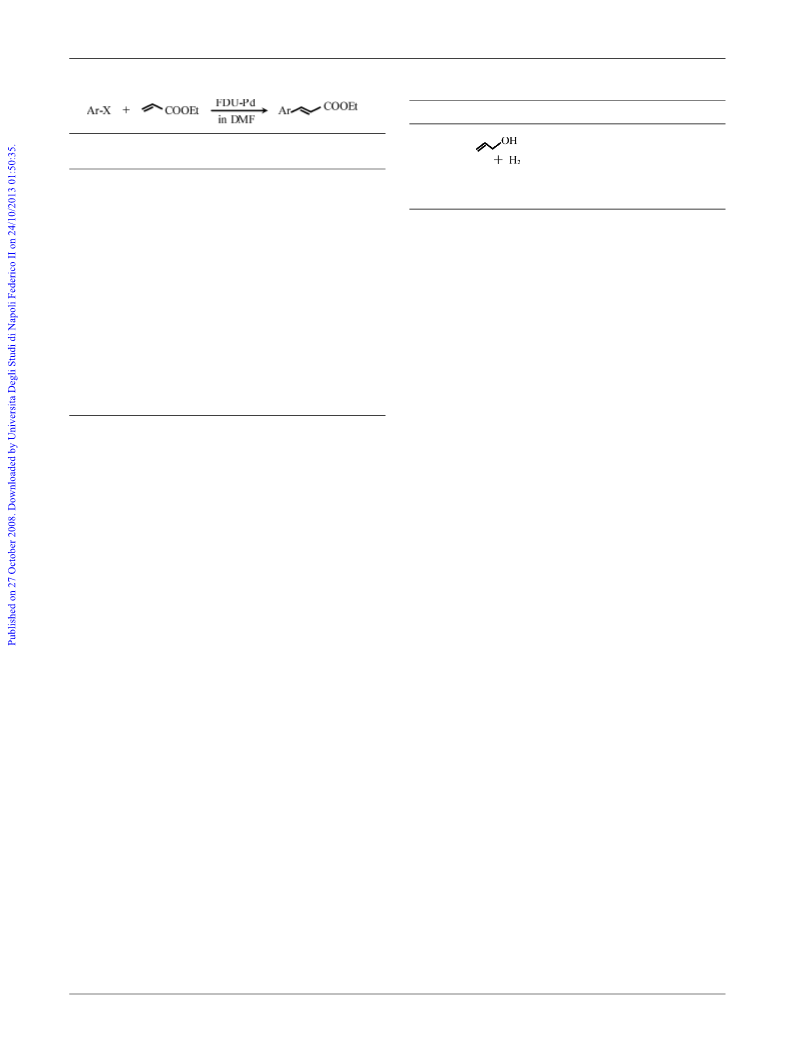
a
Table 1 Catalytic results of Heck reactions
a
Table 2 Catalytic results of allyl alcohol hydrogenation reaction
À1
Cat.
Substrate
Conv. (%) Selectivity (%) TOF/h
b
FDU-Pd
5wt% Pd/C
100
95
83
74
2944
2138
b
Temp./
1C
Conv.
Time/h (%)
À1
Entry Cat.
Substrate
TOF/h
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
FDU-Pd
PhI
PhI
PhI
PhI
PhI
50
86.00 93
6.00 99
1.00 96
0.25 92
0.33 100
0.25 91
0.25 91
1.00 Trace
0.67 98
0.50 99
0.33 95
2.50 96
0.67 95
4.20 96
50.00 72
39
590
3429
13 054
10 642
—
a
Pd = 0.056 mol%, allyl alcohol = 10 mmol, H
b
0 min. Byproduct was acetone.
2
= 1 atm, 30 1C,
FDU-Pd
FDU-Pd
FDU-Pd
FDU-Pd
80
110
130
130
130
130
130
3
extensively used in industry, the FDU-Pd catalyst showed a
significantly higher TOF, allyl alcohol conversion as well as
1-propanol selectivity. In addition, in the kinetic and recycling
experiments, FDU-Pd was confirmed to show a high stability
and reusability for the hydrogenation (Fig. S7 and S8w).
In summary, we have developed diamine-functionalized
mesopolymers which serve as promising scaffolds for disper-
sing and stabilizing Pd NPs. The FDU-Pd catalysts were air-
stable, durable, highly active and reusable in C–C formation
and hydrogenation reactions without leaching of Pd species.
The method explored here can be used as a valid way for
designing and preparing Au, Pt, Ru and Rh nanoparticle
catalysts.
Sixth reuse PhI
b
FDU-Pd
FDU-Pd
FDU-Pd
FDU-Pd
FDU-Pd
FDU-Pd
FDU-Pd
PhI
PhI
—
—
2
+c
2-MeO-PhI 130
3-MeO-PhI 130
4-MeO-PhI 130
2-CF
3-CF
5250
7072
10 179
1372
5090
82
1
1
1
1
1
1
3
-PhI 130
-PhI 130
-PhBr 130
140
3
d
FDU-Pde
FDU-Pd
4-NO
PhBr
2
3
a
Pd = 0.028 mol%, ArX = 10 mmol, ethyl acrylate = 12 mmol,
b
NEt
one month. Not reduced. Pd = 0.28 mol%. Pd = 0.6 mol%,
CO = 12 mmol.
3
= 12 mmol, DMF = 10 mL. After being stored in air for
c
d
e
K
2
3
We thank the financial supports NSFC (20673038), STCSM
08JC1408700, 07QA14017), 973 Program (2006CB202508),
(
time. With the reaction temperature increasing to 130 1C, the
TOF of FDU-Pd reached as high as 10 642 h , a superior
À1
the Research Fund for the Doctoral Program of Higher
Education Research (20070269023), and Shanghai Leading
Academic Discipline Project (B409).
5a,7
activity to other reported Pd-based heterogeneous catalysts.
To gauge the catalytic generality of FDU-Pd, it was further
applied to the Heck reactions of a range of aryl halides with
electron-donating or -withdrawing substituents. It showed an
excellent conversion (495%) for various substrates and was
capable of giving 72% conversion for inactive bromobenzene
Notes and references
1
(a) D. E. De Vos, M. Dams, B. F. Sels and P. A. Jacobs, Chem. Rev.,
002, 102, 3615; (b) J. A. Melero, R. V. Grieken and G. Morales,
2
Chem. Rev., 2006, 106, 3790; (c) S. Angelos, E. Johansson,
J. F. Stoddart and J. I. Zink, Adv. Funct. Mater., 2007, 17, 2261;
2
+
(
entries 9–15). In a control experiment, unreduced FDU-Pd
yield only a trace amount of product even after a prolonged
reaction (entry 8). This means the Pd(0) NPs served as the true
active species for Heck reaction.
(d) M. Vallet-Regı
007, 46, 7548; (e) A. Taguchi and F. Suchu
Mesoporous Mater., 2005, 77, 1.
´
, F. Balas and D. Arcos, Angew. Chem., Int. Ed.,
2
¨
th, Microporous
2
3
(a) F. Zhang, Y. Meng, D. Gu, Y. Yan, C. Yu, B. Tu and D. Zhao,
J. Am. Chem. Soc., 2005, 127, 13508; (b) Y. Meng, D. Gu, F. Zhang,
Y. Shi, H. Yang, Z. Li, C. Yu, B. Tu and D. Zhao, Angew. Chem.,
Int. Ed., 2005, 44, 7053.
(a) H. Li, Z. Bian, J. Zhu, Y. Huo, H. Li and Y. Lu, J. Am. Chem.
Soc., 2007, 129, 4538; (b) C. Yang, H. Lin, B. Zibrowius,
After the workup, the amount of Pd leached into the
reaction mixture was below the detection limit of ICP, and a
hot leaching test clearly showed that the reaction stopped
when the filtrate was further subjected to the same reaction
(
Fig. S5w). After being recycled for six runs, the Pd catalyst
¨
B. Spliethoff, F. Schuth, S. Liou, M. Chu and C. Chen, Chem.
Mater., 2007, 19, 3205; (c) H. Song, R. M. Rioux,
J. D. Hoefelmeyer, R. Komor, K. Niesz, M. Grass, P. Yang and
G. A. Somorjai, J. Am. Chem. Soc., 2006, 128, 3027.
(a) S. Kanaoka, N. Yagi, Y. Fukuyama, S. Aoshima,
H. Tsunoyama, T. Tsukuda and H. Sakurai, J. Am. Chem. Soc.,
showed a remarkable conversion (91%) (Table 1, entry 6), and
remained inside the mesopolymer pores as evidenced by
HRTEM (Fig. S6w). Moreover, FDU-Pd was comparably
active after being stored in air for one month (entry 7). This
feature is obviously different from air- and moisture-sensitive
homogeneous Pd stabilized with the phosphine ligands, also
different from other heterogeneous Pd catalysts such as
4
5
2
9
007, 129, 12060; (b) Y. Hong and A. Sen, Chem. Mater., 2007, 19,
61; (c) N. Erathodiyil, S. Ooi, A. M. Seayad, Y. Han, S. S. Lee and
J. Y. Ying, Chem.–Eur. J., 2008, 14, 3118; (d) M. Weck and
C. W. Jones, Inorg. Chem., 2007, 46, 1865.
(a) K. Okamoto, R. Akiyama, H. Yoshida, T. Yoshida and
S. Kobayashi, J. Am. Chem. Soc., 2005, 127, 2125;
Pd/SiO
2 2 3
, Pd/Al O , Pd/C and Pd/SBA-15 which are reported
to suffer the Pd leaching to a certain extent in the Heck
All these results consistently suggest that FDU-Pd
(
b) H. Miyamura, R. Matsubara, Y. Miyazaki and S. Kobayashi,
Angew. Chem., Int. Ed., 2007, 46, 4151.
6 (a) J. M. Richardson and C. W. Jones, J. Catal., 2007, 251, 80;
b) S. Jana, B. Dutta, R. Bera and S. Koner, Inorg. Chem., 2008, 47,
512; (c) J. D. Webb, S. MacQuarrie, K. McEleney and C. Crudden,
6a,8
reaction.
has the advantages of good reusability and high catalytic
activity. This could be attributed to the confining effect of
mesopores as well as the stabilizing ability of benzene rings for
metal NPs, which prevent the Pd NPs from aggregating into
less active Pd black and from leaching as well.
(
5
J. Catal., 2008, 252, 97.
7 Z. Zhang and Z. Wang, J. Org. Chem., 2006, 71, 7485.
8 (a) B. M. Choudary, S. Madhi, N. S. Chowdari, M. L. Kantam and
B. Sreedhar, J. Am. Chem. Soc., 2002, 124, 14127; (b) S. Noe
C. Luo, C. Pinel and L. Djakovitch, Adv. Synth. Catal., 2007, 349,
128; (c) R. G. Heidenreich, J. G. E. Krauter, J. Pietsch and
¨
l,
Under mild conditions, FDU-Pd catalyzed actively the hydro-
genation of allyl alcohol to produce 1-propanol (Table 2). In
comparison to a commercial 5 wt% Pd/C catalyst which is
1
¨
K. Kohler, J. Mol. Catal. A: Chem., 2002, 182–183, 499.
This journal is ꢀc The Royal Society of Chemistry 2008
Chem. Commun., 2008, 6297–6299 | 6299

 Xing, Rong
Xing, Rong
 Liu, Yueming
Liu, Yueming
 Wu, Haihong
Wu, Haihong
 Li, Xiaohong
Li, Xiaohong
 He, Mingyuan
He, Mingyuan
 Wu, Peng
Wu, Peng