Organic Letters
Letter
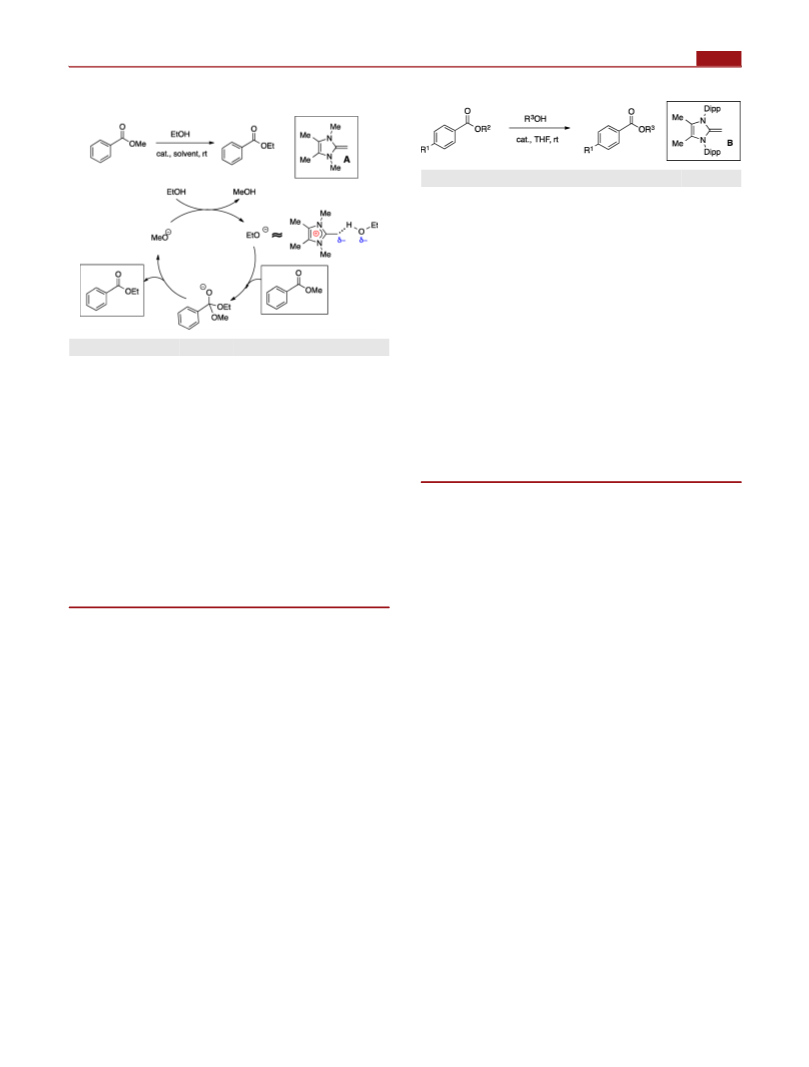
Scheme 4. NHO-Promoted Depolymerization and
Polymerization Reactions of Biodegradable Polyesters
ASSOCIATED CONTENT
Supporting Information
■
*
S
AUTHOR INFORMATION
*
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
The project was supported by the Australian Research Council
Grant DE150100517). M.B. thanks the DAAD and the ERC for
■
(
funding the research exchange to UNSW.
REFERENCES
■
(
(
1) Grondal, C.; Jeanty, M.; Enders, D. Nat. Chem. 2010, 2, 167−178.
2) Dalko, P. I.; Moisan, L. Angew. Chem., Int. Ed. 2004, 43, 5138−
175.
5
(
3) Hopkinson, M. N.; Richter, C.; Schedler, M.; Glorius, F. Nature
014, 510, 485−496.
4) Flanigan, D. M.; Romanov-Michailidis, F.; White, N. A.; Rovis, T.
Chem. Rev. 2015, 115, 9307−9387.
5) (a) Ryan, S. J.; Candish, L.; Lupton, D. W. Chem. Soc. Rev. 2013, 42,
2
(
(
4
5
(
(
906−4917. (b) Grossmann, A.; Enders, D. Angew. Chem., Int. Ed. 2012,
1, 314−325.
6) Crocker, R. D.; Nguyen, T. V. Chem. - Eur. J. 2016, 22, 2208−2213.
7) (a) Wang, Y.-B.; Wang, Y.-M.; Zhang, W.-Z.; Lu, X.-B. J. Am. Chem.
Soc. 2013, 135, 11996−12003. (b) Wang, Y.-B.; Sun, D.-S.; Zhou, H.;
Zhang, W.-Z.; Lu, X.-B. Green Chem. 2015, 17, 4009−4015.
(8) Naumann, S.; Thomas, A. W.; Dove, A. P. Angew. Chem., Int. Ed.
2
015, 54, 9550−9554.
9) Movassaghi, M.; Schmidt, M. A. Org. Lett. 2005, 7, 2453−2456.
10) Grasa, G. A.; Guveli, T.; Singh, R.; Nolan, S. P. J. Org. Chem. 2003,
8, 2812−2819.
11) Nguyen, T. V.; Lyons, D. J. M. Chem. Commun. 2015, 51, 3131−
134.
(12) For a review of organometallic-catalyzed transesterification
reactions, see: Pathak, S. J. Chem. Pharm. Res. 2015, 7 (3), 1780−1786.
13) For a review of enzyme-catalyzed transesterification reactions,
(
(
6
(
̈
close proximity to the expected molecular weights considering
the initiator:monomer ratio and a monomer conversion of
almost 100%, which is suggestive of a living system. These two
polyesters marked the second new type of organopolymerization
reaction promoted by NHOs, thus once again emphasizing the
great potential of N-heterocyclic olefin organocatalysts in
polymer chemistry. Kinetic studies to further understand and
optimize these NHO-assisted polymerization transformations
are currently underway and will be reported in due course.
In conclusion, we have demonstrated that N-heterocyclic
olefins are a novel class of versatile organocatalysts for
transesterification reactions. Because of their strong Brønsted
basicity, NHOs can enhance the nucleophilicity of alcohols for
their reactions with carboxylic esters. This procedure can be
employed in various synthetically important applications such as
depolymerization of PET, production of biodiesel, and ring-
opening polymerization of cyclic esters to give biodegradable
polymers. This work once again demonstrates the bright future
of NHO organocatalysts in synthetic chemistry. We are currently
working on other types of NHO-organocatalyzed chemical
transformations and will endeavor to report these studies in due
course.
3
(
(
(16) Esters bearing epoxide functionality did not survive the reaction
conditions, which can be attributed to the NHO-promoted ring-opening
reaction of epoxide reported in ref 8.
(
17) Grasa, G. A.; Kissling, R. M.; Nolan, S. P. Org. Lett. 2002, 4, 3583−
3586.
(18) For all of the chemical transformations reported in Schemes 3 and
4
, NHO catalyst B was used in preference to catalyst A because of its
superior thermal stability.
19) (a) Fukushima, K.; Coulembier, O.; Lecuyer, J. M.; Almegren, H.
(
A.; Alabdulrahman, A. M.; Alsewailem, F. D.; Mcneil, M. A.; Dubois, P.;
Waymouth, R. M.; Horn, H. W.; Rice, J. E.; Hedrick, J. L. J. Polym. Sci.,
Part A: Polym. Chem. 2011, 49, 1273−1281. (b) Fukushima, K.; Coady,
D. J.; Jones, G. O.; Almegren, H. A.; Alabdulrahman, A. M.; Alsewailem,
F. D.; Horn, H. W.; Rice, J. E.; Hedrick, J. L. J. Polym. Sci., Part A: Polym.
Chem. 2013, 51, 1606−1611.
(20) During the preparation of this article, another study on the ROP
of cyclic esters catalyzed by N-alkyl-NHOs was reported. See: Naumann,
S.; Thomas, A. W.; Dove, A. P. ACS Macro Lett. 2016, 5, 134−138.
D
Org. Lett. XXXX, XXX, XXX−XXX

 Blümel, Marcus
Blümel, Marcus
 Noy, Janina-Miriam
Noy, Janina-Miriam
 Enders, Dieter
Enders, Dieter
 Stenzel, Martina H.
Stenzel, Martina H.
 Nguyen, Thanh V.
Nguyen, Thanh V.