E. Białecka-Florjan´czyk et al. / Journal of Molecular Catalysis B: Enzymatic 74 (2012) 241–245
245
Table 1
References
Synthesis of 2-phenylethyl acetate in the presence of pretreated Y. lipolytica KKP
379 cells (20 g) versus commercial lipase from C. antarctica (25 mg).
[1] J. Kim, D.H. Altreuter, D.S. Clark, J.S. Dordick, Jaocs 75 (1998) 1109–1113.
[2] K.J. Verstrepen, G. Derdelinckx, J.P. Dufour, J. Winderickx, J.M. Thevelein, I.S.
Pretorius, F.R. Delvaux, J. Biosci. Bioeng. 96 (2003) 110–118.
[3] U. Krings, R.G. Berger, Appl. Microbiol. Biotechnol. 49 (1998) 1–8.
[4] S. Serra, C. Fuganti, E. Brenna, Trends Biotechnol. 23 (2005) 193–198.
[5] Y.C. Park, C.E.H. Shaffer, G.N. Bennett, Appl. Microbiol. Biotechnol. 85 (2009)
13–25.
[6] M.A. Longo, M.A. Sanromán, Food Technol. Biotechnol. 44 (2006) 253–335.
[7] P. Fickers, J. Destain, P. Thonart, Biotechnol. Agron. Soc., Environ. 12 (2008)
119–130.
Substrate
Product
Yield [%]
Cells of
Y. lipolytica
Commercial lipase
Candida antarctica
Ethyl acetate
Vinyl acetate
2-phenylethyl acetate
2-phenylethyl acetate
73% 4%
78% 7%
79% 13%
57% 9%
[8] A. Houde, A. Kademi, D. Leblanc, Appl. Biochem. Biotechnol. 118 (2004)
155–170.
[9] F. Laboret, R. Perraud, Appl. Biochem. Biotechnol. 82 (1999) 185–199.
[10] H. Nakagawa, S. Watanabe, S. Shimura, K. Kirimura, S. Usami, World J. Microbiol.
Biotechnol. 14 (1998) 219–222.
[11] S. Hari Krishna, S. Divakar, S.G. Prapulla, N.G. Karanth, J. Biotechnol. 87 (2001)
193–201.
[12] C. Lecointe, E. Dubreucq, P. Galzy, Biotechnol. Lett. 18 (1996) 869–874.
[13] A. Larios, H.S. Garcia, R.M. Oliart, G. Valerio-Alfaro, Appl. Microbiol. Biotechnol.
65 (2004) 373–376.
[14] F. Bi, S. Iqbal, A. Ali, M. Ul-Hassan, J. Chem. Soc. Pak. 31 (2009) 485–491.
[15] R.B. Salah, H. Ghamghui, N. Miled, H. Mejdoub, Y. Gargouri, J. Biosci. Bioeng.
103 (2007) 368–372.
[16] D. Bezbradica, D. Mijin, S. Siler-Marinkovic, Z. Knezevic, J. Mol. Catal. B: Enzym.
45 (2007) 97–101.
[17] P. Villeneuve, Biotechnol. Adv. 25 (2007) 515–536.
[18] O. Boutur, E. Dubreucq, P. Galzy, J. Biotechnol. 42 (1995) 23–33.
[19] Y. Ota, K. Gomi, S. Kato, T. Sugiura, Y. Minoda, Agric. Biol. Chem. 46 (1982)
2885–2893.
[20] P. Fickers, A. Marty, M. Nicaud, Biotechnol. Adv. 20 (2011) 632–644.
[21] P. Fickers, P.H. Benetti, Y. Waché, A. Marty, S. Mauersberger, M.S. Smit, J.M.
Nicaud, FEMS Yeast Res. 5 (2005) 527–543.
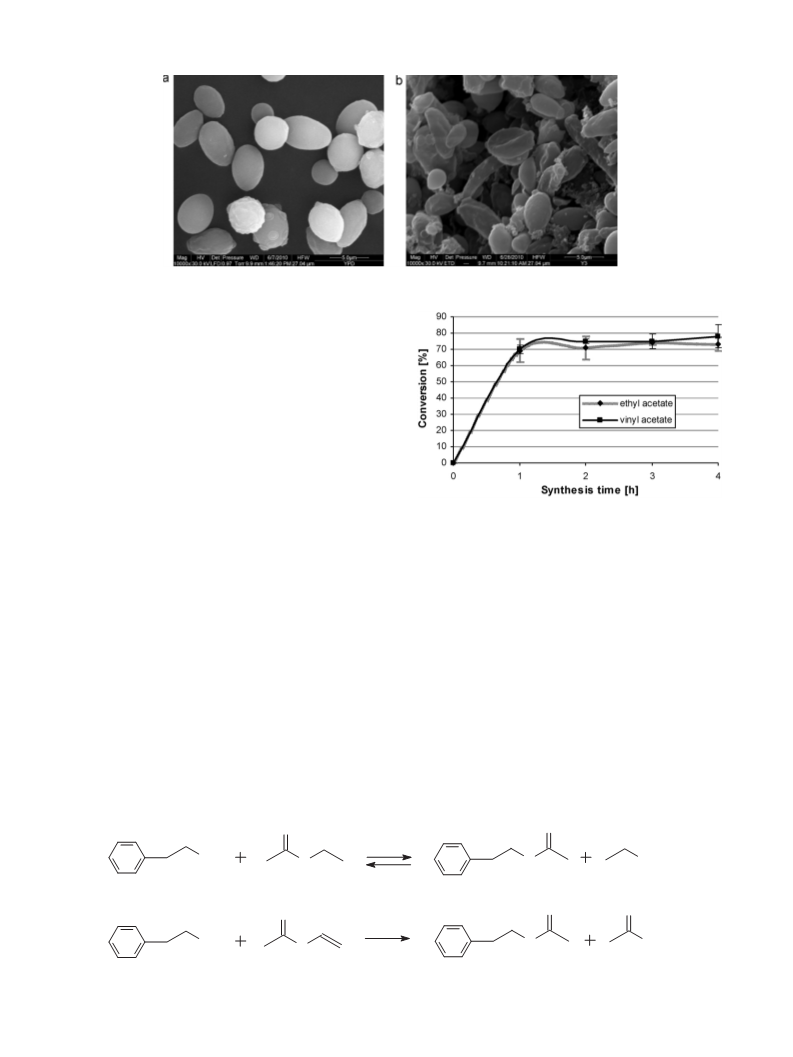
Specifically, in the presence of microbial cells, ester yields were
78% 7%, while for the commercial lipase, only 57% 9% conver-
sion was obtained. Probably, this disparity is due to the influence
is well-known that lipases from Candida cylindracea do not dis-
play any catalytic activity during transesterification with vinyl
acetate, because of the accompanying release of aldehyde with
lysine, resulting in Schiff base formation [38]. Based on the above,
application of whole cell catalysts enable not only comparable, but
sometime significantly better reaction efficiencies.
4. Conclusions
Based on our results, we conclude that Y. lipolytica KKP 379 yeast
cells are effective catalysts for 2-phenylethyl acetate synthesis. Fur-
thermore, for the catalytic activity of these cells the pretreatment
with isooctane is necessary, and the duration of organic solvent
contact by the cells is advantageous and requires further inves-
tigations. Although sonication is an effective method of cell wall
permeabilisation, and allows for better contact between lipolytic
enzymes and their substrates; released enzymes may be deacti-
vated as a result of ultrasound irradiation.
Acyl donor type does not influence the effectiveness of Y. lipoly-
tica biomass catalysed transesterification reactions. Using either
vinyl acetate or ethyl acetate resulted in similar reaction efficien-
cies, and after 2 h, ester conversions reached in both cases 72–78%.
The efficiency of reactions catalysed with yeast biomass were
similar to those catalysed by isolated lipase enzymes. Therefore, our
research strongly suggests that isolated enzymes can be replaced
by biomass for the synthesis of esters, in order to reduce reaction
costs.
[22] E.E. Garc´a, M. Aguedo, N. Gomes, A. Choquet, I. Belo, J.A. Teixeira, J.M. Belin, Y.
Waché, J. Mol. Catal. B: Enzym. 57 (2009) 22–26.
[23] Y. Waché, M. Aguedo, J.M. Nicaud, J.M. Belin, Appl. Microbiol. Biotechnol. 61
(2003) 393–404.
[24] E. Albertazzi, R. Cardillo, S. Servi, G. Zucchi, Biotechnol. Lett. 16 (1994)
491–496.
[25] J. Krzyczkowska, I. Stolarzewicz, E. Białecka-Florjan´ czyk, Monograph Wielok-
ierunkowos´c´ Badan´ w Rolnictwie i Lesnictwie 2 (2009) 665–671 (in Polish).
[26] E. Wehtje, P. Adlercreutz, Biotechnol. Lett. 11 (1997) 537–540.
[27] C. Laane, S. Boeren, K. Vos, C. Veeger, Biotechnol. Bioeng. 30 (1987) 81–87.
[28] G.D. Yadav, A.H. Trivedi, Enzyme Microb. Technol. 32 (2003) 783–789.
[29] F.V. Pereira-Meirelles, M.H. Rocha-Leão, G.L. Sant Anna Jr, Appl. Biochem.
Biotechnol. 63 (1997) 73–85.
[30] E. Białecka-Florjan´ czyk, J. Krzyczkowska, I. Stolarzewicz, Biocatal. Biotrans-
form. 8 (4) (2010) 288–291.
[31] D. Wang, Y. Xu, Y. Teng, Bioprocess Biosyst. Eng. 30 (2007) 147–155.
[32] T.J. Mason, Ultrason. Sonochem. 10 (2003) 175–179.
[33] Y. Chisti, Trends Biotechnol. 21 (2003) 89–93.
[34] A. Kapturowska, I. Stolarzewicz, I. Chmielewska, E. Białecka-Florjan´ czyk,
˙
Zywnos´c´. Nauka. Technologia. Jakos´c´. 4 (77) (2011) 160–171 (in Polish).
[35] E.V. Rokhina, P. Lens, J. Virkutyte, Trends Biotechnol. 27 (2009) 298–306.
[36] T. Chatterjee, D.K. Bhattacharyya, J. Am. Oil Chem. Soc. 75 (1998) 651–655.
[37] A.B. Majumder, B. Singh, D. Dutta, S. Sadhukhan, M.N. Gupta, Bioorg. Med. Chem.
Lett. 16 (2006) 4041–4044.
Acknowledgments
This research was financially supported by the Ministry of Sci-
ence and Higher Education (Grant No. N N209 107639).
[38] C. Torres, C. Otero, Enzyme Microb. Technol. 19 (1996) 594–600.

 Bialecka-Florjanczyk, Ewa
Bialecka-Florjanczyk, Ewa
 Krzyczkowska, Jolanta
Krzyczkowska, Jolanta
 Stolarzewicz, Izabela
Stolarzewicz, Izabela
 Kapturowska, Agata
Kapturowska, Agata