S. B. Waghmode et al. / Tetrahedron Letters 42 (2001) 3145–3147
3147
Table 3. Acylation of 2-phenylethanol with various car-
boxylic acids over Na–K–ETS-10
Acknowledgements
Nos Acylating
agent
Conversion
Othersa
Acylation
selectivityb
S.B.W. and V.V.T. thank CSIR for research fellow-
ships.
1
2
Acetic acid
Propanoic
acid
97.8
99.4
0.8
1.4
99.2
98.5
References
3
4
5
Octanoic acid 96.8
Nonanoic acid 90.7
Decanoic acid 76.3
6.0
10.2
12.7
93.8
89.8
87.3
1. Philippou, A.; Anderson, M. W. J. Catal. 2000, 189,
395.
2. Philippou, A.; Naderi, M.; Rocha, J.; Anderson, M. W.
Catal. Lett. 1998, 53, 221.
3. Waghmode, S. B.; Das, T. K.; Vetrivel, R.; Sivasanker,
S. J. Catal. 1999, 185, 265.
Reaction conditions: temperature=120°C; substrate=5 mmol; acetic
acid=4 mL; other acids=5 mmol; and duration of run=8 h.
a Mainly C-alkylated products.
b Selectivity=(mol% expected product/mol% conversion)×100.
4. (a) Horton, D. Org. Synth. 1973, 5, 1; (b) Zhanov, R. I.;
Zhenodaova, S. M. Synthesis 1975, 222; (c) Hofle, G.;
Steglich, V.; Vorbruggen, H. Angew. Chem., Int. Ed.
Engl. 1978, 17, 569; (d) Scriven, E. F. V. Chem. Soc.
Rev. 1983, 12, 129.
5. Baker, R. H.; Bordwell, F. G. Org. Synth. Coll. Vol.
1955, 3, 141.
6. Iqbal, J.; Srivastava, R. R. J. Org. Chem. 1992, 57,
2001.
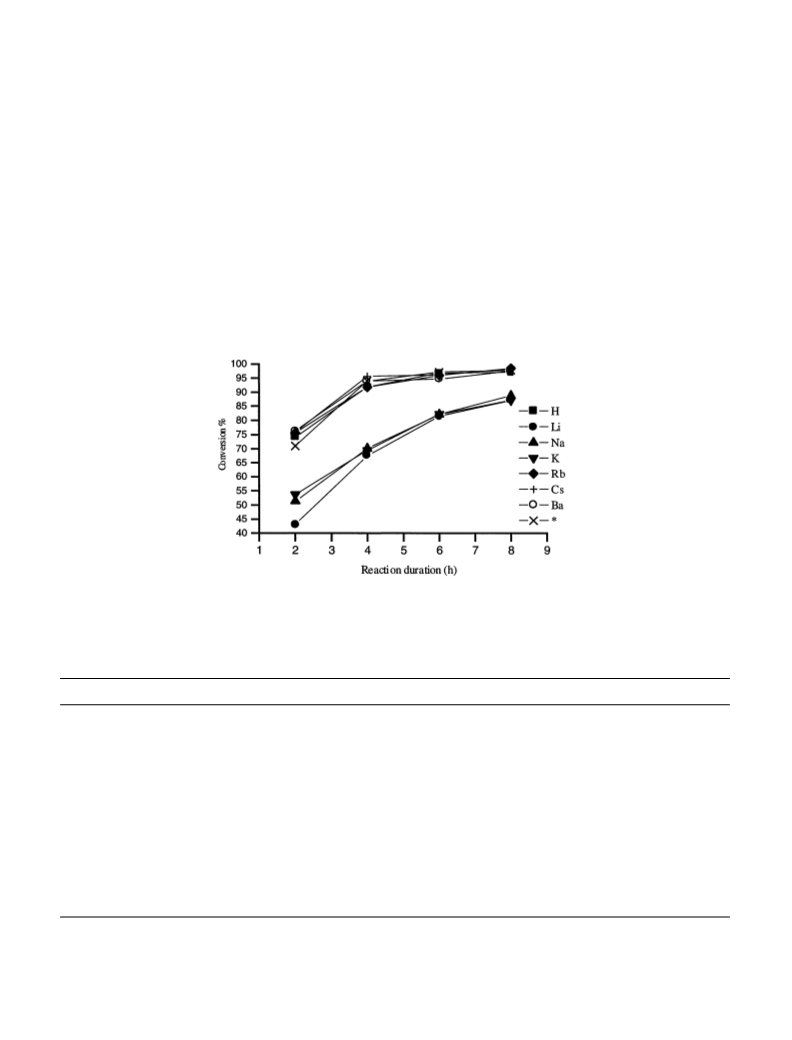
In conclusion, ETS-10 molecular sieves are useful in the
acylation of primary and secondary alcohols with acetic
acid and long chain carboxylic acids (C8–C10). Alcohol
conversion increases with the basicity of the catalyst.
General procedure for catalyst preparation
ETS-10 was prepared according to the procedure
reported earlier.17 The Si/Ti ratio was 5.61. The ETS-10
sample was converted into different ion-exchanged
forms by exchanging thrice with the required metal
chloride solution (20 mL of 1 M solution/g of catalyst
at 80°C for 3 h). After washing with demineralized
water, filtering and drying, the M-ETS-10 samples
(M=Li, Na, K, Rb, Cs and Ba) were calcined at 500°C
for 4 h. Powder X-ray diffraction data of the calcined
samples (collected on Rigaku Model D-MAX III VC)
revealed the samples to be highly crystalline. Elemental
analyses were done by atomic adsorption spectroscopy
(AAS).
7. Okano, T.; Miyamoto, K.; Kiji, J. Chem. Lett. 1995,
245.
8. (a) Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto,
H. J. Am. Chem. Soc. 1995, 117, 4413; (b) Ishihara, K.;
Kubota, M.; Yamamoto, H. Synlett 1996, 265; (c) Bar-
rett, A. G. M.; Braddock, D. C. Chem. Commun. 1997,
351; (d) Ishihara, K.; Kubota, M.; Kurihara, H.;
Yamamoto, H. J. Org. Chem. 1998, 61, 4560; (e) Zhao,
H.; Pendri, A.; Greenwald, R. B. J. Org. Chem. 1998,
63, 7559.
9. (a) Procopiou, P. A.; Baugh, S. P. D.; Flack, S. S.;
Inglis, G. G. A. J. Chem. Soc., Chem. Commun. 1996,
2625; (b) Procopiou, P. A.; Baugh, S. P. D.; Flack, S.
S.; Inglis, G. G. A. J. Org. Chem. 1998, 63, 2342.
10. Chandrasekhar, S.; Ramachander, T.; Takhi, M. Tetra-
hedron Lett. 1998, 39, 3263.
General procedure for the acylation reaction
All the reactions were carried out in the liquid phase in
a round bottom flask fitted with a cold water con-
denser. Substrate (5 mmol), acetic acid (4 mL) (in the
case of long chain C8–C10 acids, a stoichiometric
amount was used) and 50 mg of catalyst were refluxed
at 120°C for 8 h. Conversions have been calculated on
the basis of alcohol used. The progress of the reaction
was monitored by a gas chromatography (HP-5880)
using a capillary column (HP-5, cross-linked methyl–
silicone gum, 50 m×0.5 mm). After completion, the
reaction mixture was quenched with a saturated solu-
tion of NaHCO3 and extracted with ethyl acetate. The
organic layer was washed with water and brine and the
solvent was evaporated in vacuo to give almost pure
products.
11. Chauhan, K. K.; Frost, C. G.; Love, I.; Waite, D.
Synlett 1999, 1743.
12. Saravanan, P.; Singh, V. K. Tetrahedron Lett. 1999, 40,
2611.
13. Orita, A.; Tanahashi, C.; Kakuda, A.; Otera, J. Angew.
Chem., Int. Ed. Engl. 2000, 39, 877.
14. Li, A.-I.; Li, T.-S.; Ding, T.-H. J. Chem. Soc., Chem.
Commun. 1997, 1389.
15. Narender, N.; Srinivasu, P.; Kulkarni, S. J.; Raghavan,
K. V. Synth. Commun. 2000, 30, 1887.
16. Kammoun, N.; Le Bigot, Y.; Delmas, M.; Boutevin, B.
Synth. Commun. 1997, 27, 2777.
17. Das, T.; Chandwadkar, A. J.; Sivasanker, S. J. Chem.
Soc., Chem. Commun. 1996, 1105.
.
.

 Waghmode, Suresh B.
Waghmode, Suresh B.
 Thakur, Vinay V.
Thakur, Vinay V.
 Sudalai, Arumugam
Sudalai, Arumugam
 Sivasanker, Subramanian
Sivasanker, Subramanian