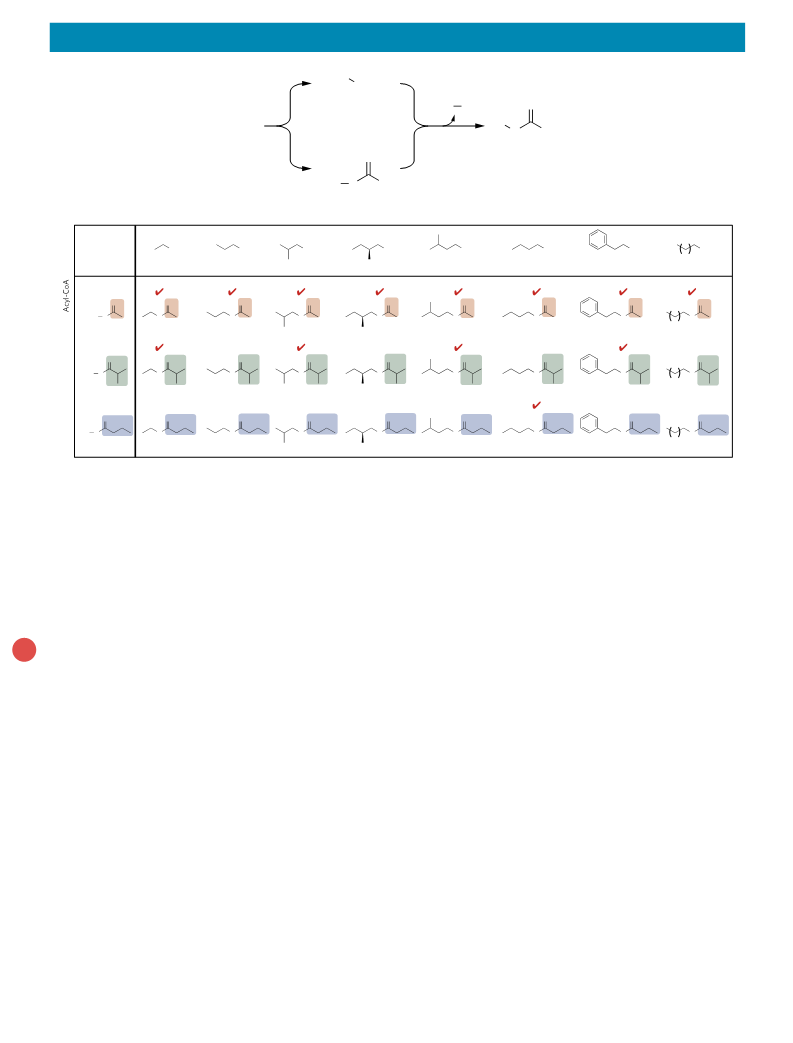
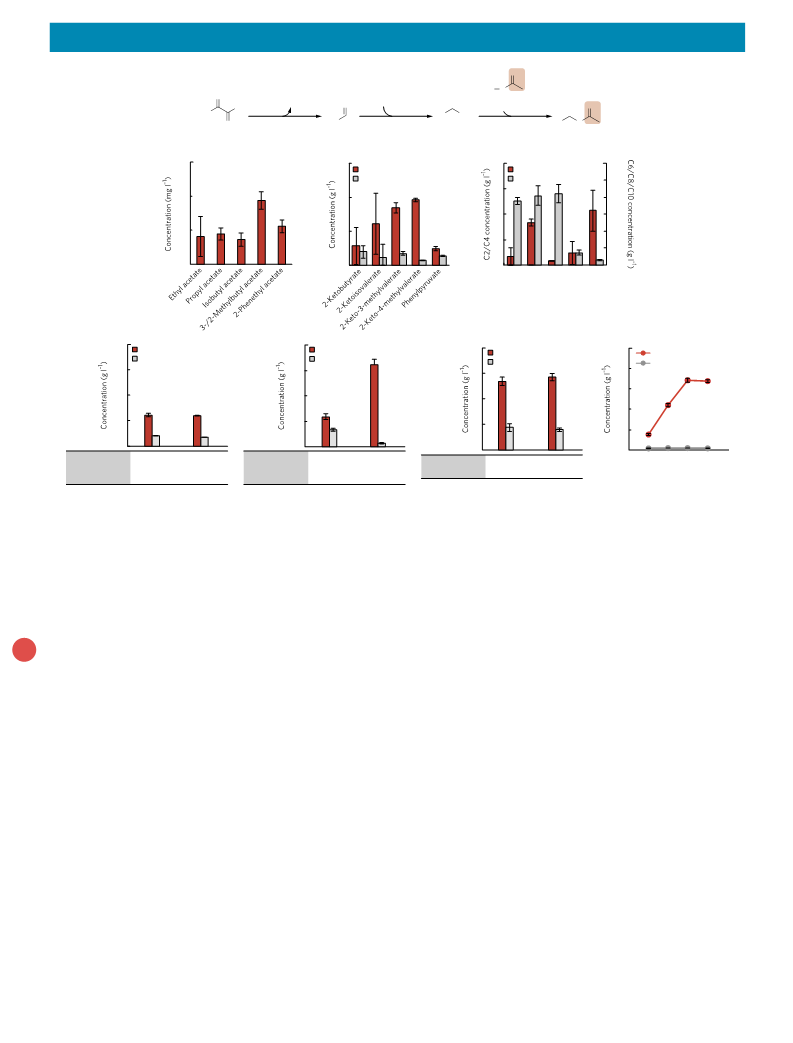
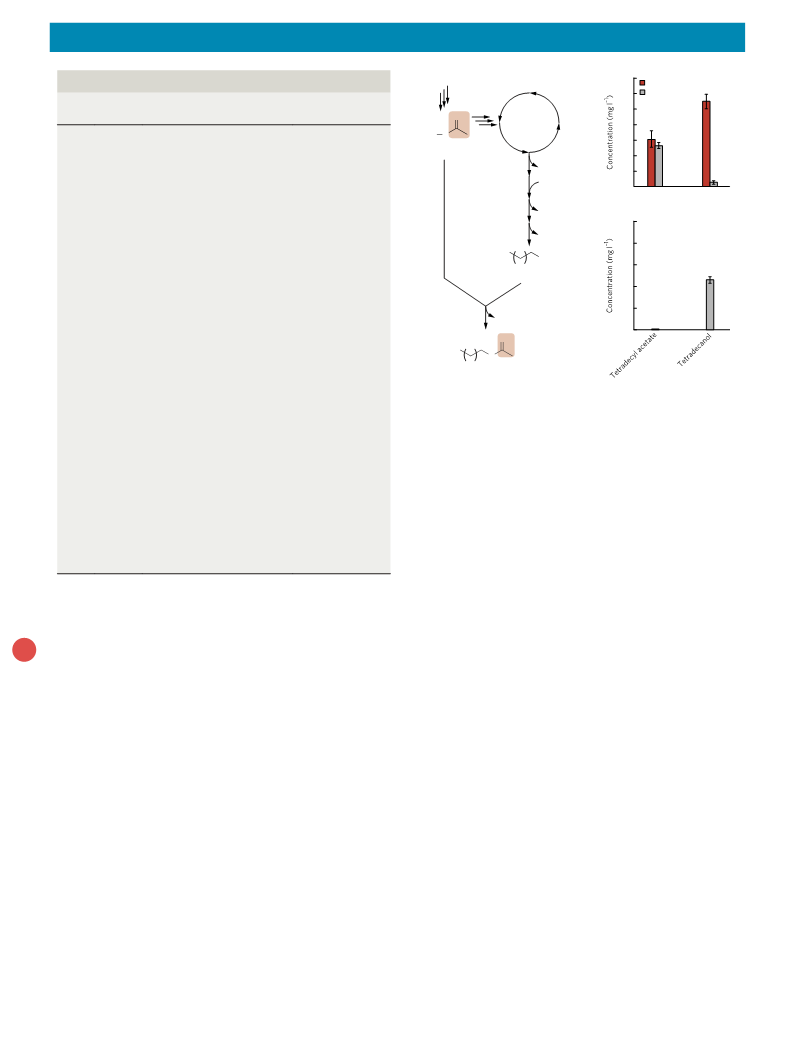
article
references
28. Lin, F., Das, D., Lin, X.N. & Marsh, E.N. Aldehyde-forming fatty acyl-CoA
reductase from cyanobacteria: expression, purification and characterization of
the recombinant enzyme. FEBS J. 280, 4773–4781 (2013).
1. Beekwilder, J. et al. Functional characterization of enzymes forming volatile
esters from strawberry and banana. Plant Physiol. 135, 1865–1878 (2004).
2. Verstrepen, K.J. et al. Expression levels of the yeast alcohol acetyltransferase
genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of
volatile esters. Appl. Environ. Microbiol. 69, 5228–5237 (2003).
3. Iwasaki, T., Maegawa, Y., Ohshima, T. & Mashima, K. in Kirk-Othmer
Encyclopedia of Chemical Technology Vol. 10, 497–516 (John Wiley & Sons,
New Jersey, 2012).
29. Schirmer, A., Rude, M.A., Li, X., Popova, E. & del Cardayre, S.B. Microbial
biosynthesis of alkanes. Science 329, 559–562 (2010).
30. Mooney, B.P., Miernyk, J.A. & Randall, D.D. e complex fate of α-ketoacids.
Annu. Rev. Plant Biol. 53, 357–375 (2002).
31. Rodriguez, G.M. & Atsumi, S. Isobutyraldehyde production from
Escherichia coli by removing aldehyde reductase activity. Microb. Cell Fact.
11, 90 (2012).
4. Liu, Y.J., Lotero, E. & Goodwin, J.G. Effect of water on sulfuric acid catalyzed
esterification. J. Mol. Catal. Chem. 245, 132–140 (2006).
32. de la Plaza, M., Fernandez de Palencia, P., Pelaez, C. & Requena, T.
Biochemical and molecular characterization of α-ketoisovalerate
decarboxylase, an enzyme involved in the formation of aldehydes from amino
acids by Lactococcus lactis. FEMS Microbiol. Lett. 238, 367–374 (2004).
33. Bastian, S. et al. Engineered ketol-acid reductoisomerase and alcohol
dehydrogenase enable anaerobic 2-methylpropan-1-ol production at
theoretical yield in Escherichia coli. Metab. Eng. 13, 345–352 (2011).
34. Baez, A., Cho, K.M. & Liao, J.C. High-flux isobutanol production using
engineered Escherichia coli: a bioreactor study with in situ product removal.
Appl. Microbiol. Biotechnol. 90, 1681–1690 (2011).
35. Atsumi, S. et al. Engineering the isobutanol biosynthetic pathway in
Escherichia coli by comparison of three aldehyde reductase/alcohol
dehydrogenase genes. Appl. Microbiol. Biotechnol. 85, 651–657 (2010).
36. Connor, M.R., Cann, A.F. & Liao, J.C. 3-Methyl-1-butanol production
in Escherichia coli: random mutagenesis and two-phase fermentation.
Appl. Microbiol. Biotechnol. 86, 1155–1164 (2010).
5. Nelson, D.L. & Cox, M.M. Lehninger Principles of Biochemistry 5th edn.
(Sara Tenney, New York, 2008).
6. Stergiou, P.Y. et al. Advances in lipase-catalyzed esterification reactions.
Biotechnol. Adv. 31, 1846–1859 (2013).
7. Dhake, K.P., akare, D.D. & Bhanage, B.M. Lipase: a potential biocatalyst for
the synthesis of valuable flavour and fragrance ester compounds. Flavour
Fragrance J. 28, 71–83 (2013).
8. Rabinovitch-Deere, C.A., Oliver, J.W., Rodriguez, G.M. & Atsumi, S. Synthetic
biology and metabolic engineering approaches to produce biofuels. Chem.
Rev. 113, 4611–4632 (2013).
9. Bornscheuer, U.T. et al. Engineering the third wave of biocatalysis. Nature
485, 185–194 (2012).
10. Fujii, T. et al. Molecular cloning, sequence analysis, and expression of the
yeast alcohol acetyltransferase gene. Appl. Environ. Microbiol. 60, 2786–2792
(1994).
11. Rowland, O. & Domergue, F. Plant fatty acyl reductases: enzymes generating
fatty alcohols for protective layers with potential for industrial applications.
Plant Sci. 193–194, 28–38 (2012).
37. Belas, R. et al. Bacterial bioluminescence: isolation and expression of the
luciferase genes from Vibrio harveyi. Science 218, 791–793 (1982).
38. Meighen, E.A. Bacterial bioluminescence: organization, regulation, and
application of the lux genes. FASEB J. 7, 1016–1022 (1993).
39. Ferrández, A., Garcia, J.L. & Diaz, E. Genetic characterization and
expression in heterologous hosts of the 3-(3-hydroxyphenyl)propionate
catabolic pathway of Escherichia coli K-12. J. Bacteriol. 179, 2573–2581
(1997).
12. Rontani, J.F., Bonin, P.C. & Volkman, J.K. Production of wax esters during
aerobic growth of marine bacteria on isoprenoid compounds. Appl. Environ.
Microbiol. 65, 221–230 (1999).
13. Lenneman, E.M., Ohlert, J.M., Palani, N.P. & Barney, B.M. Fatty alcohols for
wax esters in Marinobacter aquaeolei VT8: two optional routes in the wax
biosynthesis pathway. Appl. Environ. Microbiol. 79, 7055–7062 (2013).
14. Reiser, S. & Somerville, C. Isolation of mutants of Acinetobacter calcoaceticus
deficient in wax ester synthesis and complementation of one mutation with
a gene encoding a fatty acyl coenzyme A reductase. J. Bacteriol. 179,
2969–2975 (1997).
40. Lee, S.J., Ko, J.H., Kang, H.Y. & Lee, Y. Coupled expression of MhpE
aldolase and MhpF dehydrogenase in Escherichia coli. Biochem. Biophys. Res.
Commun. 346, 1009–1015 (2006).
41. Hester, K., Luo, J., Burns, G., Braswell, E.H. & Sokatch, J.R. Purification of
active E1α2β2 of Pseudomonas putida branched-chain-oxoacid
dehydrogenase. Eur. J. Biochem. 233, 828–836 (1995).
15. Holtzapple, E. & Schmidt-Dannert, C. Biosynthesis of isoprenoid wax ester in
Marinobacter hydrocarbonoclasticus DSM 8798: identification and
characterization of isoprenoid coenzyme A synthetase and wax ester
synthases. J. Bacteriol. 189, 3804–3812 (2007).
16. Barney, B.M., Wahlen, B.D., Garner, E., Wei, J. & Seefeldt, L.C. Differences in
substrate specificities of five bacterial wax ester synthases. Appl. Environ.
Microbiol. 78, 5734–5745 (2012).
17. Stöveken, T., Kalscheuer, R., Malkus, U., Reichelt, R. & Steinbuchel, A. e
wax ester synthase/acyl coenzyme A:diacylglycerol acyltransferase from
Acinetobacter sp. strain ADP1: characterization of a novel type of
acyltransferase. J. Bacteriol. 187, 1369–1376 (2005).
18. Atsumi, S., Hanai, T. & Liao, J.C. Non-fermentative pathways for synthesis
of branched-chain higher alcohols as biofuels. Nature 451, 86–89 (2008).
19. Shen, C.R. et al. Driving forces enable high-titer anaerobic 1-butanol
synthesis in Escherichia coli. Appl. Environ. Microbiol. 77, 2905–2915 (2011).
20. Bond-Watts, B.B., Bellerose, R.J. & Chang, M.C. Enzyme mechanism as a
kinetic control element for designing synthetic biofuel pathways. Nat. Chem.
Biol. 7, 222–227 (2011).
21. Inokuma, K., Liao, J.C., Okamoto, M. & Hanai, T. Improvement of
isopropanol production by metabolically engineered Escherichia coli using gas
stripping. J. Biosci. Bioeng. 110, 696–701 (2010).
22. Dellomonaco, C., Clomburg, J.M., Miller, E.N. & Gonzalez, R. Engineered
reversal of the β-oxidation cycle for the synthesis of fuels and chemicals.
Nature 476, 355–359 (2011).
23. Saerens, S.M. et al. e Saccharomyces cerevisiae EHT1 and EEB1 genes
encode novel enzymes with medium-chain fatty acid ethyl ester synthesis and
hydrolysis capacity. J. Biol. Chem. 281, 4446–4456 (2006).
24. Lilly, M. et al. e effect of increased yeast alcohol acetyltransferase and
esterase activity on the flavour profiles of wine and distillates. Yeast 23,
641–659 (2006).
25. Mason, A.B. & Dufour, J.P. Alcohol acetyltransferases and the significance of
ester synthesis in yeast. Yeast 16, 1287–1298 (2000).
26. Fujii, T., Yoshimoto, H. & Tamai, Y. Acetate ester production by
Saccharomyces cerevisiae lacking the ATF1 gene encoding the alcohol
acetyltransferase. J. Ferment. Bioeng. 81, 538–542 (1996).
42. Hester, K.L., Luo, J. & Sokatch, J.R. Purification of Pseudonomas Putida
branched-chain keto acid dehydrogenase E1 component. Methods Enzymol.
324, 129–138 (2000).
43. Alonso-Gutierrez, J. et al. Metabolic engineering of Escherichia coli for
limonene and perillyl alcohol production. Metab. Eng. 19, 33–41 (2013).
44. Park, Y.C., Shaffer, C.E. & Bennett, G.N. Microbial formation of esters.
Appl. Microbiol. Biotechnol. 85, 13–25 (2009).
45. van den Berg, C., Heeres, A.S., Van der Wielen, L.A. & Straathof, A.J.
Simultaneous Clostridial fermentation, lipase-catalyzed esterification, and
ester extraction to enrich diesel with butyl butyrate. Biotechnol. Bioeng. 110,
137–142 (2013).
acknowledgments
This work was supported by University of California–Davis startup fund and the
Hellman fellowship to S.A. G.M.R. is supported by a US National Institutes of Health
Biotechnology Training Grant Fellowship (T32-GM008799) and a Sloan Fellowship.
Y.T. is supported by Japan Society for the Promotion of Science postdoctoral fellowship
for research abroad. We would like to thank R. Luu and R.E. Parales (University of
California–Davis) for graciously providing genomic DNA from P. putida g7 and
M. Kato and S.-J. Lin (University of California–Davis) for providing genomic DNA from
S. cerevisiae BY4742. We also thank M.D. Toney and C.A. Rabinovitch-Deere (University
of California–Davis) for critical reading of the manuscript. Finally, we thank S. Desai for
technical assistance with HPLC analysis and for constructing pAL603.
author contributions
G.M.R., Y.T. and S.A. designed research; G.M.R. and Y.T. performed the experiments;
G.M.R., Y.T. and S.A. analyzed data; and G.M.R., Y.T. and S.A. wrote the paper.
Competing financial interests
The authors declare no competing financial interests.
additional information
html. Correspondence and requests for materials should be addressed to S.A.
27. Howard, T.P. et al. Synthesis of customized petroleum-replica fuel molecules
by targeted modification of free fatty acid pools in Escherichia coli. Proc. Natl.
Acad. Sci. USA 110, 7636–7641 (2013).
265

 Rodriguez, Gabriel M
Rodriguez, Gabriel M
 Tashiro, Yohei
Tashiro, Yohei
 Atsumi, Shota
Atsumi, Shota