PAPER
Large-Scale Acetylation of Alcohols and Diols Using Bismuth Triflate
2093
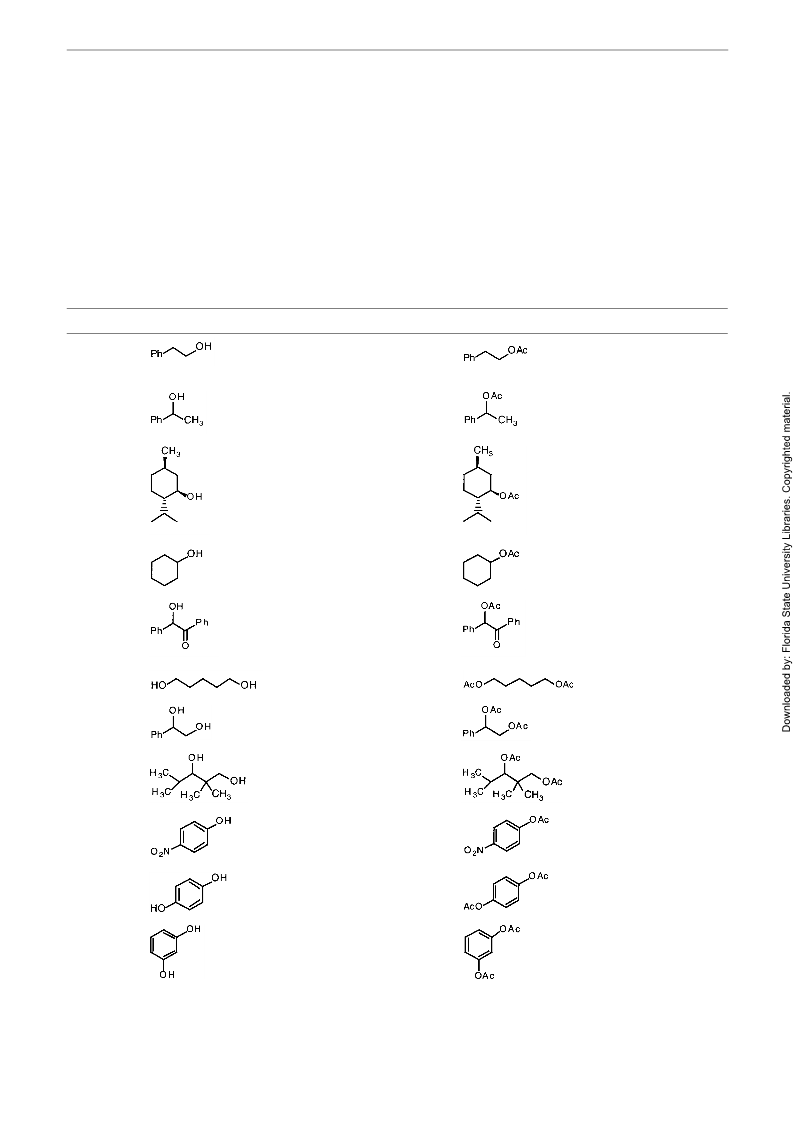
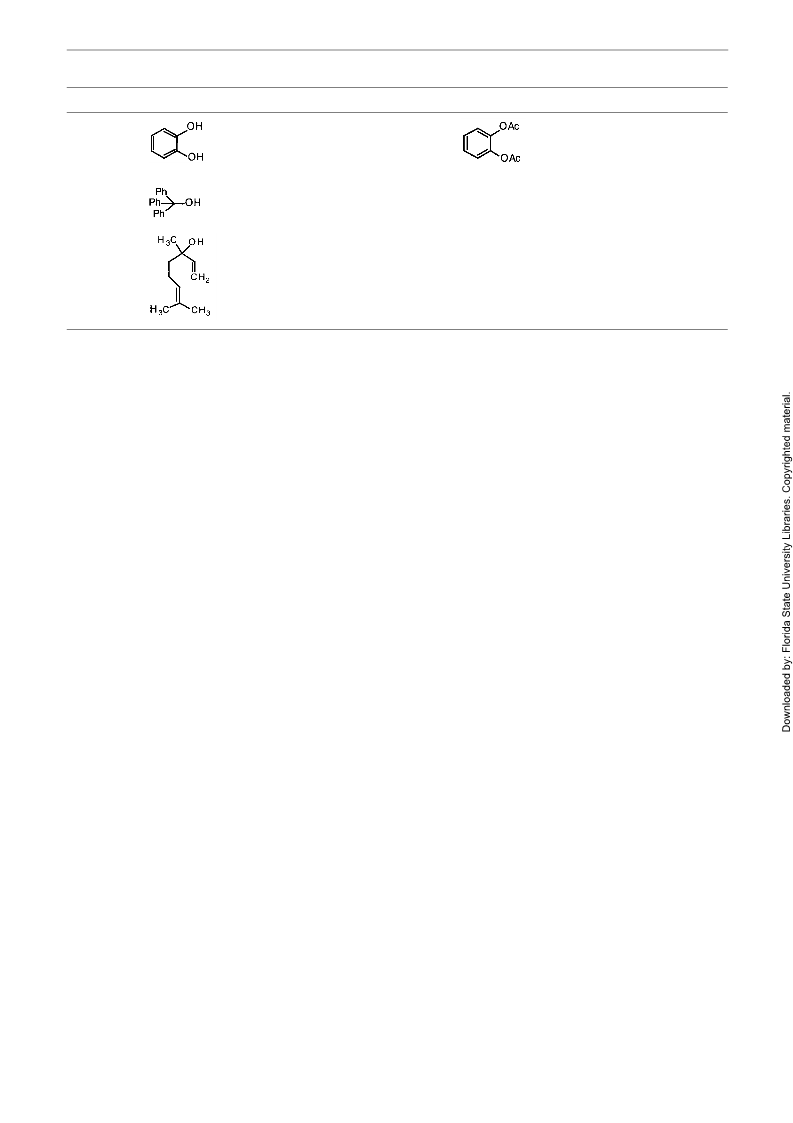
Table Bismuth Triflate-Catalyzed acetylation of Alcohols in MeCNa (continued)
Entry
12f
Alcohol
Timeb
Product
Yield (%)c,d
85
15 min
13g
14
12 h
12 h
NR
NR
a Reagent grade MeCN was used.
b All reactions were run at r.t. and reaction progress was monitored by TLC.
c Yields refer to that of isolated product.
d The crude product in each case was deemed to be of sufficient (> 98%) purity by 1H and 13C NMR spectroscopy and hence was not purified
further.
e Reaction was carried out using 1 mol% catalyst.
f 3.0 Equivalents of Ac2O were used for all diols.
g No reaction was observed even when the mixture was heated at reflux for 3 h.
(2) Stork, G.; Takahashi, T.; Kawamoto, I.; Suzuki, T. J. Am.
Chem. Soc. 1978, 100, 8272.
In summary, this work demonstrates a practical prepara-
tive method for the acetylation of 1° and 2° alcohols and
diols. The advantages of bismuth triflate include (1) ease
of handling, (2) low cost, (3) fast reaction rates, (4) cata-
lytic in nature and (5) insensitivity to air and small
amounts of moisture, eliminating the need for anhydrous
solvents.
(3) (a) Steglich, W.; Höfle, G. Angew. Chem., Int. Ed. Engl.
1969, 8, 981. (b) Review: Höfle, G.; Steglich, W.;
Vorbrüggen, H. Angew. Chem., Int. Ed. Engl. 1978, 17, 569.
(4) Kumareswaran, R.; Gupta, A.; Vankar, Y. D. Synth.
Commun. 1997, 27, 277.
(5) Vedejs, E.; Daugulis, O. J. Org. Chem. 1996, 61, 5702.
(6) Ishihara, K.; Kubota, M.; Yamamoto, H. Synlett 1996, 265.
(7) Miyashita, M.; Shiina, I.; Miyoshi, S.; Mukaiyama, T. Bull.
Chem. Soc. Jpn. 1993, 66, 1516.
(8) Iqbal, J.; Srivastava, R. R. J. Org. Chem. 1992, 57, 2001.
(9) (a) Sn(OTf)2: Mukaiyama, T.; Shiina, I.; Miyashita, M.
Chem. Lett. 1992, 625. (b) Cu(OTf)2: Saravanan, P.; Singh,
V. K. Tetrahedron Lett. 1999, 40, 2611. (c) In(OTf)3:
Chauhan, K. K.; Frost, C. G.; Love, L.; Waite, D. Synlett
1999, 1743.
sec-Phenethyl Acetate; Typical Procedure
To a stirred solution of sec-phenethyl alcohol (8.00 g, 0.0655 mol)
in MeCN (80 mL) were added Ac2O (10.0 g, 0.0982 mol) and bis-
muth triflate (42.9 mg, 6.55 ¥ 10–5 mol, 0.1 mol%). After 1 h,
MeCN was removed on a rotary evaporator and 10% aq Na2CO3 so-
lution (40 mL) was added. The mixture was extracted with Et2O (3
¥ 30 mL) and the combined organic extracts were washed with aq
Na2CO3 solution until basic. The organic layer was washed further
with brine (15 mL) and dried (Na2SO4). The solvents were removed
on a rotary evaporator to give 9.28 g (86 %) of a colorless liquid,
which was identified to be sec-phenethyl acetate (>98% pure) by 1H
NMR, 13C NMR and IR spectroscopy.
(10) Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto, H. J.
Org. Chem. 1996, 61, 4560.
(11) Garrett, R. L. In Designing Safer Chemicals; Garrett, R. L.;
De Vito, S. C., Eds.; American Chemical Society
Symposium Series 640: Washington DC, 1996, Chap. 1.
(12) (a) Reglinski, J. In Chemistry of Arsenic, Antimony and
Bismuth; Norman, N. C., Ed.; Blackie Academic and
Professional: New York, 1998, 403–440. (b) Marshall, J. A.
Chemtracts 1997, 1064. (c) Suzuki, H.; Ikegami, T.;
Matano, Y. Synthesis 1997, 249. (d) Organobismuth
Chemistry; Suzuki, H.; Matano, Y., Eds.; Elsevier:
Amsterdam, 2001.
Acknowledgement
We wish to acknowledge funding by the National Science Founda-
tion (NSF-RUI Grant 0078881).
(13) (a) Wada, M.; Takegichi, E.; Matsumoto, T. Bull. Chem.
Soc. Jpn. 1991, 64, 990. (b) Le Roux, C.; Gaspard-
Iloughmane, H.; Dubac, J.; Jaud, J.; Vignaux, P. J. Org.
Chem. 1993, 58, 1835.
(14) (a) Labrouillère, M.; Le Roux, C.; Gaspard, H.; Laporterie,
A.; Dubac, J. Tetrahedron Lett. 1997, 38, 8871.
(b) Repichet, S.; Le Roux, C.; Dubac, J.; Desmure, J.-R. Eur.
J. Org. Chem. 1998, 2743.
References
(1) (a) Greene, T. W.; Wuts, P. G. M. Protective Groups in
Organic Synthesis; Wiley: New York, 1999, 3rd ed.
(b) Hanson, J. R. Protecting Groups in Organic Synthesis;
Blackwell Science, Inc: Malden, MA, 1999, 1st ed.
(c) Kocienski, P. J. Protecting Groups; Georg Thieme
Verlag: Stuttgart, 1994, 1st ed.
Synthesis 2001, No. 14, 2091–2094 ISSN 0039-7881 © Thieme Stuttgart · New York

 Carrigan
Carrigan
 Freiberg
Freiberg
 Smith
Smith
 Zerth
Zerth
 Mohan
Mohan