4
84
SHORT PAPER
A Facile and Efficient One-Step Conversion of Alcohol Triphenylmethyl
Ethers to the Corresponding Acetates
O
K
ne-step
C
onversi
u
on of
A
lcoho
m
l
Triphenylmethyl
E
i
thers t
k
o Acetates o Kobayashi, Tsutomu Watahiki, Takeshi Oriyama*
Department of Environmental Sciences, Faculty of Science, Ibaraki University, 2-1-1 Bunkyo, Mito 310-8512, Japan
Fax +81(29)2288403; E-mail: tor@mx.ibaraki.ac.jp
Received 26 December 2002
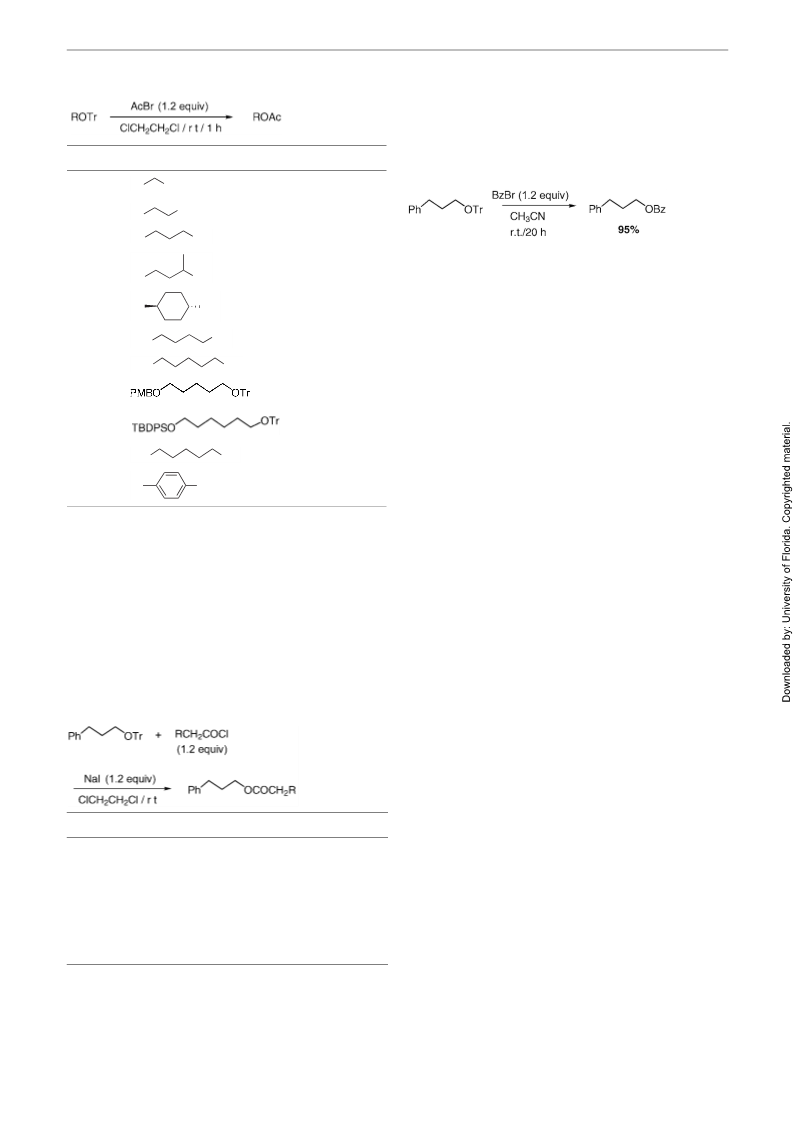
To establish the optimal reaction conditions, acylation of
-triphenylmethoxy-3-phenylpropane as a model sub-
strate using acetyl bromide was carried out, and the effect
Abstract: Alcohol triphenylmethyl (trityl) ethers were readily and
efficiently transformed into the corresponding acetates by reaction
with acetyl bromide. Triphenylmethyl ethers can also be trans-
1
formed into the corresponding substituted acetates in high yields by of solvents was investigated. Among the solvents tested,
the use of various substituted acetyl chlorides combined with sodi- 1,2-dichloroethane was found to be the most suitable sol-
um iodide.
vent for this one-pot transformation and the desired ace-
Key words: one-step conversion of protecting groups, trityl ethers, tate was obtained quantitatively.
acetates, acetyl bromide, sodium iodide
The scope of this direct conversion of trityl ethers into the
corresponding acetates was examined under the optimal
conditions: 1.2 equivalents of acetyl bromide in 1,2-
Alcohol triphenylmethyl (trityl) ethers are significant
functional groups and protecting groups. They have been
dichloroethane at room temperature for 1 hour. The repre-
sentative results are summarized in Table 1. Trityl ethers
of primary and secondary alcohols were transformed into
the corresponding acetates in excellent yields (Runs 1–5).
Successful results were obtained by using distilled acetyl
bromide, which was considered to contain scarcely any
HBr. When we used commercially available acetyl bro-
mide without purification, the corresponding acetate was
obtained in lower yield (Run 3; 100→86%). In the case of
the trityl ether substrates. having other functional groups
such as a benzoate, benzyl ether, p-methoxybenzyl ether,
1
widely used for the protection of hydroxyl functions, es-
pecially in carbohydrate chemistry. While trityl ethers are
generally hydrolyzed under acidic reaction conditions to
2
afford the parent alcohols, acetates are stable under sim-
ilar acidic conditions. On the other hand, several years
ago, we initiated a program focused on the one-step con-
3
version of a hydroxyl protecting group into another. As
one part of this investigation, we presented that an acetyl
bromide was a very effective acylation reagent for the
one-step conversion of tetrahydropyranyl (THP) ethers
6
and t-butyldiphenylsilyl (TBDPS) ether, the trityl ether
4
into the corresponding acetates. This direct conversion of
moieties were transformed into the corresponding ace-
tates selectively (Runs 6–9). The bis-trityl ether of an ali-
phatic 1,5-diol was readily converted into the
corresponding bis-acetate in 93% yield using 2.5 equiva-
lents of acetyl bromide (Run 10). Furthermore, phenolic
trityl ethers were also successfully transformed into the
corresponding acetates in good yields (Run 11). In this
case, p-bromophenol was also obtained in 12% yield.
THP ethers to the corresponding acetates in one pot pro-
ceeds smoothly and efficiently without a catalyst. More-
over, substituted acetates were also obtained from
substituted acetyl chlorides with an equimolar amount of
sodium iodide. In contrast, Bergmeier et al. have recently
reported the direct conversion of trityl ethers into ace-
5
tates. This reaction was performed by the treatment of
trityl ethers with 2 equivalents of acetyl chloride. Longer
reaction time is needed to attain higher yield of acetates.
Therefore, Bergmeier’s report led us to study the use of
acetyl bromide in a fast direct conversion of alcohol trityl
ethers to acetates.
To study the generality of this reaction, we examined a re-
action of the 1-triphenylmethoxy-3-phenylpropane with
1
.2 equivalents of acetyl chloride. After the reaction mix-
ture was stirred for 24 hours at room temperature, usual
work-up afforded 3-phenylpropyl acetate in moderate
yield (54%). As mentioned earlier in this text, sodium io-
dide accelerates the acylation of THP ethers by reaction
with acetyl chloride. After evaluation of the effect of ad-
ditives, it was revealed that 1.2 equivalents of sodium io-
dide functioned as an excellent promoter in this one-step
conversion into acetate, and that the corresponding acetate
was obtained in 98% yield (Table 2, Run 1). Other suc-
cessful examples, of the reaction of 3-phenylpropanol
trityl ether with substituted acetyl chlorides in the pres-
ence of sodium iodide are collected in Table 2. Various
substituted acetates were obtained in good to excellent
yields by the combination of the corresponding substitut-
Herein, we wish to report the protocol for the one-step
conversion of trityl ethers into the corresponding acetates.
This transformation allows catalyst-free acylation of trityl
ethers under mild conditions, and several functional
groups including benzyl ether, p-methoxybenzyl ether,
ester survive the reaction. Furthermore, this method is a
clean synthesis from the viewpoint of atom economy.
Synthesis 2003, No. 4, Print: 18 03 2003.
Art Id.1437-210X,E;2003,0,04,0484,0486,ftx,en;F07702SS.pdf.
©
Georg Thieme Verlag Stuttgart · New York
ISSN 0039-7881

 Kobayashi, Kumiko
Kobayashi, Kumiko
 Watahiki, Tsutomu
Watahiki, Tsutomu
 Oriyama, Takeshi
Oriyama, Takeshi