1914
B. R. Kim et al.
LETTER
Cl
Cl
+
N
N
–O
C
O
R'
Cl
Cl
Cl
O
Cl
Cl
Al– Cl
H
Cl
Al– Cl
AlCl3
O
R
alcohol 2
R
O
R
O
+
N
H
N
+
N
C
HO
N
C
Cl
Cl
+
O
R'
O
R'
1
Cl
Cl
N
Cl
Cl
Al– Cl
Cl
O
R
O
N
+
N
R
+
HO
– AlCl3
Cl
N
R'
O
HO
C
ester
3 or 5
O
R'
4,5-dichloropyridazin-
3(2H)-one
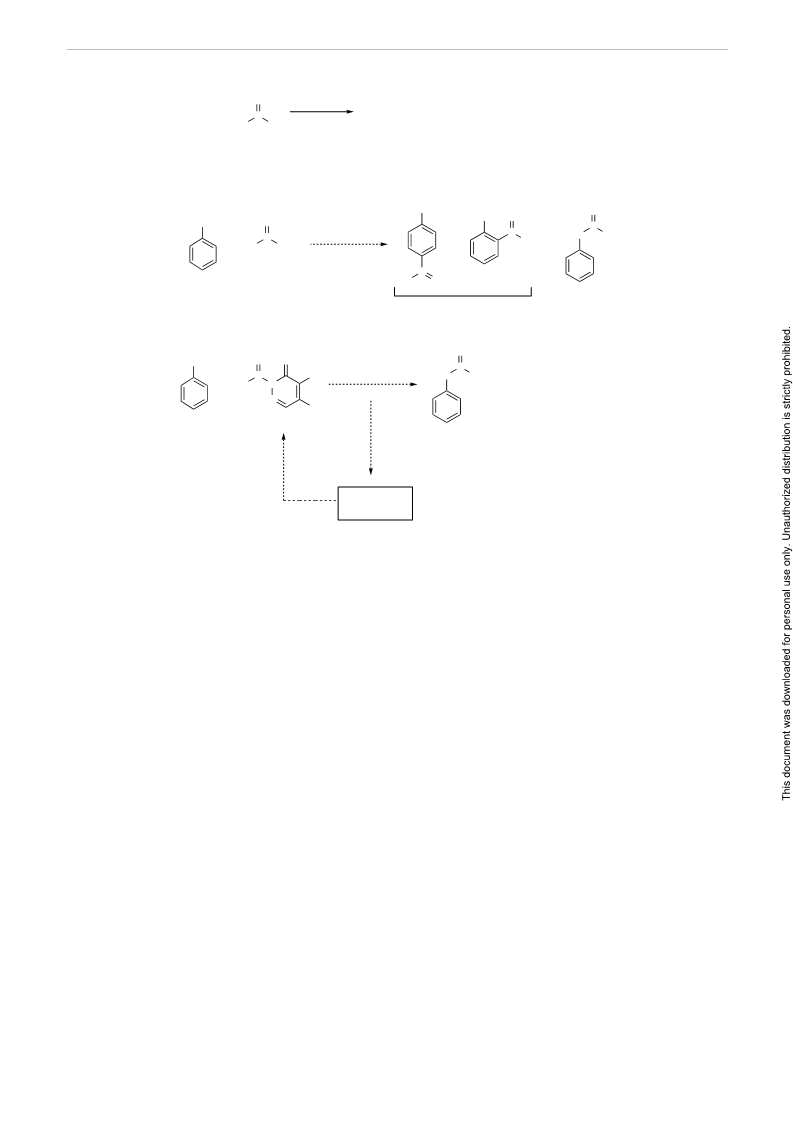
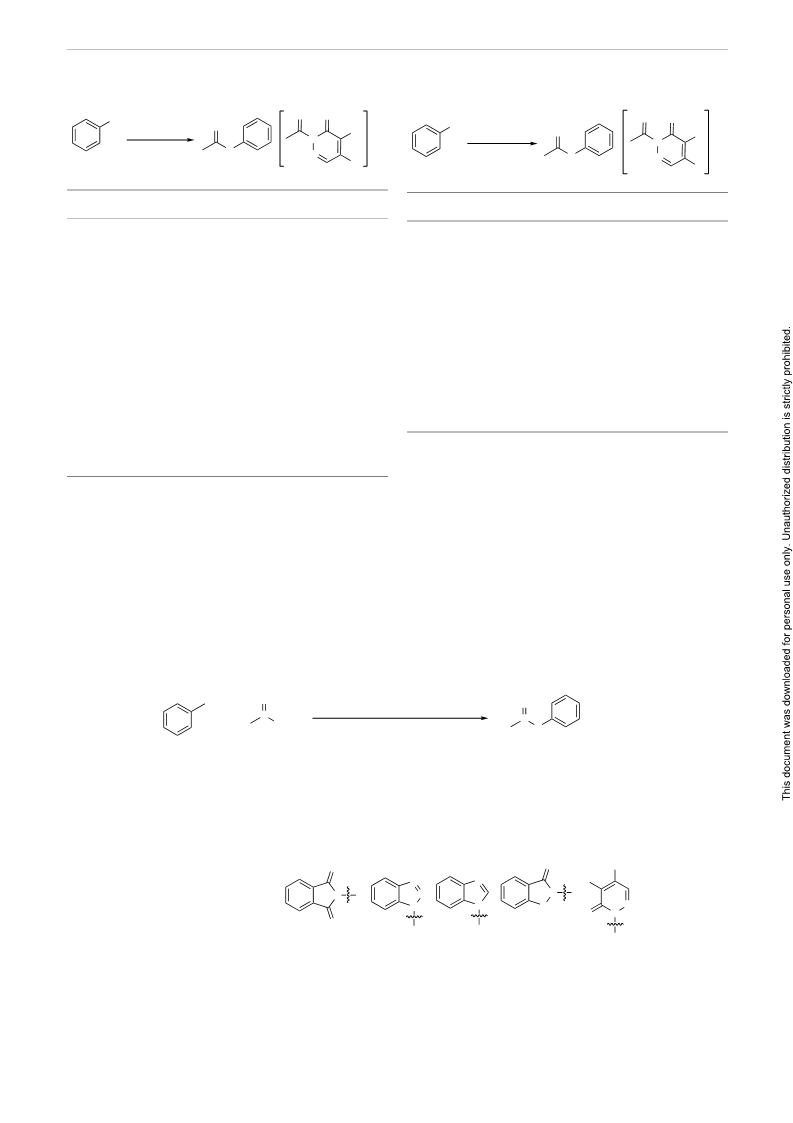
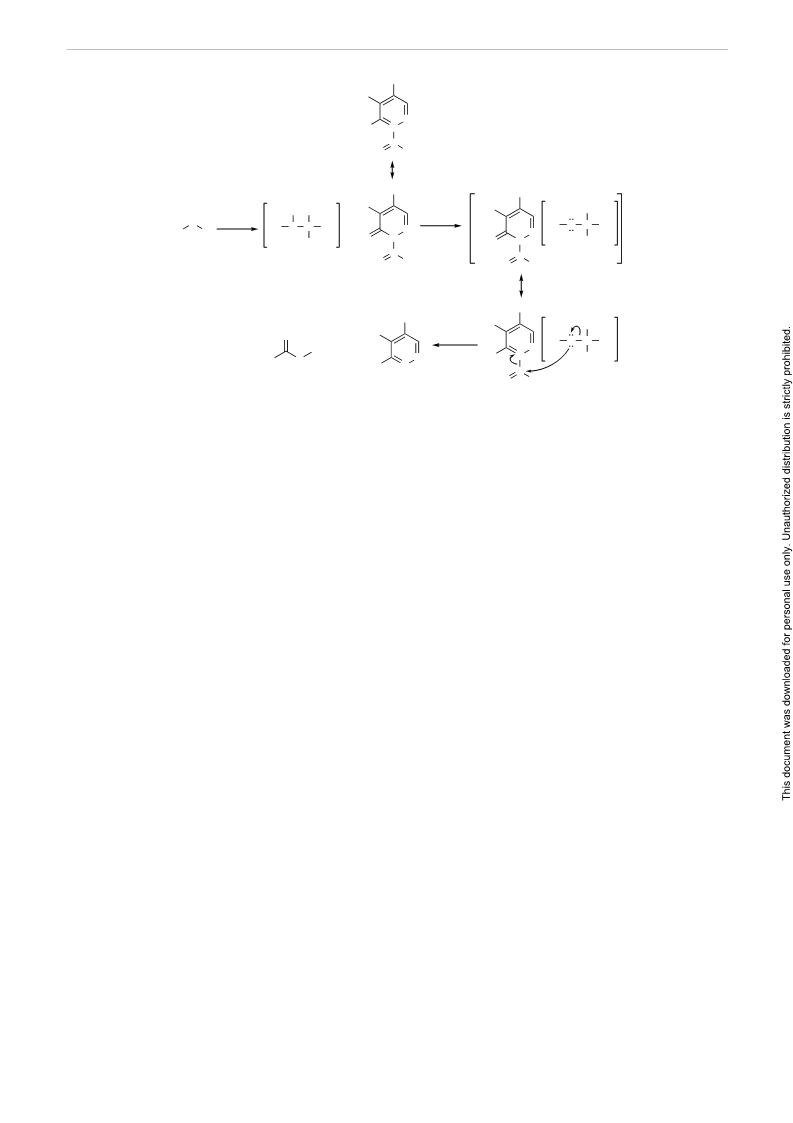
Scheme 4 Plausible mechanism for the esterification of alcohols with 2-acyl-4,5-dichloropyridazin-3(2H)-one in the presence of AlCl3
(4) (a) Hangx, G.; Kwant, G.; Maessen, H.; Markusse, P.;
Urseanu, I. Reaction Kinetics of the Esterification of Ethanol
and Acetic Acid Towards Ethyl Acetate, Deliverable 22,
Intelligent Column Internals for Reactive Separations
(INTINT), Technical Report to the European Commission,
(2001). (b) Zhang, G.-S. Synth. Commun. 1998, 28, 1159.
(5) (a) Ishihara, K.; Nakayama, M.; Ohara, S.; Yamamoto, H.
Tetrahedron 2002, 58, 8179. (b) Ishihara, K.; Nakagawa, S.;
Sakakura, A. J. Am. Chem. Soc. 2005, 127, 4168. (c) Chen,
C.-T.; Munot, Y. S. J. Org. Chem. 2005, 70, 8625.
J. Org. Chem. 2004, 69, 1822. (m) Wakasugi, K.; Iida, A.;
Misaki, T.; Nishii, Y.; Tanabe, Y. Adv. Synth. Catal. 2003,
345, 1209.
(8) For some examples, see refs. 7d and 7j–m. See also: Anastas,
P. T.; Kirchhoff, M. M. Acc. Chem. Res. 2002, 35, 686.
(9) Constable, D. J. C.; Dunn, P. J.; Hayler, J. D.; Humphrey, G.
R.; Leazer, J. L.; Linderman, R. J. Jr.; Lorenz, K.; Manley,
J.; Pearlman, B. A.; Wells, A.; Zaks, A.; Zhang, T. Y. Green
Chem. 2007, 9, 411.
(10) (a) Sartori, G.; Maggi, R. Chem. Rev. 2006, 106, 1077.
(b) Chaube, V. D.; Moreau, P.; Finiels, A.; Ramaswamy, A.
V.; Singh, A. P. J. Mol. Catal. A: Chem. 2001, 174, 255.
(c) Oulsanjo, M. S.; Cartledge, T.; Shah, K.; Ahmed, S. Lett.
Drug Des. Discovery 2011, 8, 253. (d) Hocking, M. B.
J. Chem. Technol. Biotechnol. 1980, 30, 626.
(11) (a) Kim, B. R.; Sung, G. H.; Ryu, K. E.; Kim, J. J.; Yoon, Y.
J. Bull. Korean Chem. Soc. 2013, 34, 3410. (b) Kim, B. R.;
Sung, G. H.; Lee, S. G.; Yoon, Y. J. Tetrahedron 2013, 69,
3234.
(12) (a) Sung, G. H.; Kim, B. R.; Lee, S. G.; Kim, J. J.; Yoon, Y.
J. Curr. Org. Chem. 2012, 16, 852. (b) Lee, S. G.; Kim, J. J.;
Kim, H. K.; Kweon, D. H.; Kang, Y. J.; Cho, S. D.; Kim, S.
K.; Yoon, Y. J. Curr. Org. Chem. 2004, 8, 1463. (c) Kang,
Y. J.; Chung, H.-A.; Kim, J. J.; Yoon, Y. J. Synthesis 2002,
733.
(13) (a) Chung, H.-A.; Kim, J. J.; Cho, S. D.; Lee, S. G.; Yoon,
Y. J. J. Heterocycl. Chem. 2002, 39, 685. (b) Cho, S. D.;
Park, Y. D.; Kim, J. J.; Joo, W. H.; Shiro, M.; Esser, L.;
Falck, J. R.; Ahn, C.; Shin, D. S.; Yoon, Y. J. Tetrahedron
2004, 60, 3763.
(14) (a) Synthesis of 3 and 5a–d; General Procedure: To a
solution of alcohol 2 (1 equiv, 2 mmol) in THF (20 mL),
AlCl3 (1 equiv) was added followed by stirring for 1 min. 2-
Acyl-4,5-pyridazin-3(2H)-one 1 (1 equiv) was then added
and the mixture was stirred at room temperature until the
alcohol was consumed. The reaction was quenched by the
addition of H2O (20 mL), and the mixture was stirred for 10
min at room temperature. After the addition of CH2Cl2 (40
mL), the organic layer was separated and dried over
(d) Chakraborti, A. K.; Singh, B.; Chankeshwara, S. V.;
Patel, A. R. J. Org. Chem. 2009, 74, 5967. (e) Barrett, A. G.
M.; Braddock, D. C. Chem. Commun. 1997, 351.
(f) Srinivas, K. V. N. S.; Mahender, I.; Das, B. Synthesis
2003, 2479. (g) Sharghi, H.; Hosseini Sarvari, M.
Tetrahedron 2003, 59, 3627. (h) Moumne, R.; Lavielle, S.;
Karoyan, P. J. Org. Chem. 2006, 71, 3332.
(6) For selected examples, see: (a) Mackay, L. G.; Wylie, R. S.;
Sanders, J. K. J. Am. Chem. Soc. 1994, 116, 3141.
(b) Gianneschi, N. C.; Nguyen, S. T.; Mirkin, C. A. J. Am.
Chem. Soc. 2005, 127, 1644. (c) Yoon, H. J.; Mirkin, C. A.
J. Am. Chem. Soc. 2008, 130, 11590. (d) Neises, B.;
Steglich, W. Angew. Chem., Int. Ed. Engl. 1978, 17, 522.
(7) For selected examples, see: (a) Ohta, S.; Shimabayashi, A.;
Aono, M.; Okamoto, M. Synthesis 1982, 833. (b) Maseguer,
J. D.; Coll, A. L. P.; Lizarbe, J. R. F.; Bilbao, A. Z. Synthesis
1980, 547. (c) Kim, S.; Lee, J. I.; Ko, Y. K. Tetrahedron
Lett. 1984, 25, 4943. (d) Nahmany, M.; Melman, A. Org.
Lett. 2001, 3, 3733. (e) Saitoh, K.; Shiina, I.; Mukaiyama, T.
Chem. Lett. 1988, 679. (f) Candrasekaran, S.; Tumer, J. V.
Synth. Commun. 1982, 12, 727. (g) Mukaiyama, T.;
Ichikawa, J.; Asami, M. Chem. Lett. 1983, 683. (h) Ramaiah,
M. J. Org. Chem. 1985, 50, 4991. (i) Shiina, I.; Miyoshi, S.;
Miyashita, M.; Mukaiyama, T. Chem. Lett. 1994, 515.
(j) Kunishima, M.; Kawach, C.; Hioki, K.; Terao, K.; Tani,
S. Tetrahedron 2001, 57, 1551. (k) Wakasugi, K.;
Nakamura, A.; Tanabe, Y. Tetrahedron Lett. 2001, 42, 7427.
(l) Shiina, I.; Kubota, M.; Oshiumi, H.; Hashizume, M.
Synlett 2014, 25, 1909–1915
© Georg Thieme Verlag Stuttgart · New York

 Kim, Bo Ram
Kim, Bo Ram
 Sung, Gi Hyeon
Sung, Gi Hyeon
 Ryu, Ki Eun
Ryu, Ki Eun
 Yoon, Hyo Jae
Yoon, Hyo Jae
 Lee, Sang-Gyeong
Lee, Sang-Gyeong
 Yoon, Yong-Jin
Yoon, Yong-Jin