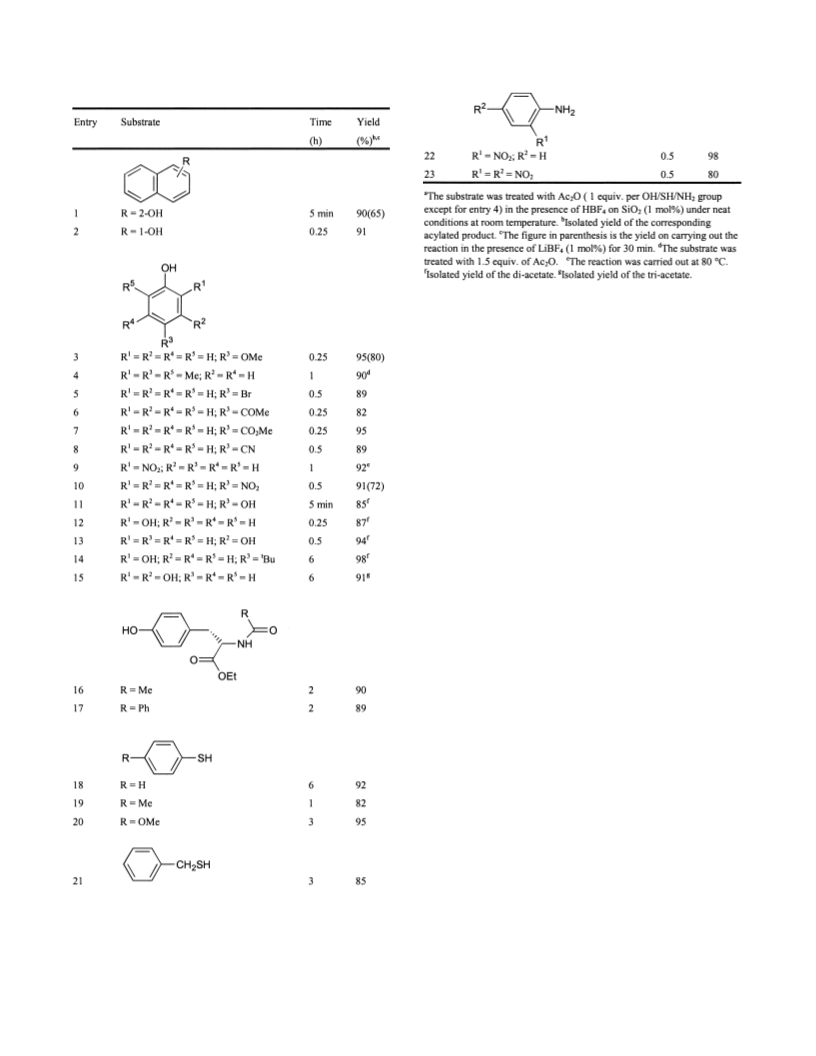
3524
A. K. Chakraborti, R. Gulhane / Tetrahedron Letters 44 (2003) 3521–3525
References
very strong Lewis acids and hence unsuitable for their
use in dealing with acid-sensitive substrates. This is
exemplified by the necessity of a large excess of Ac2O
and/or low temperature (0 to −20°C) to suppress the
competitive side reactions (e.g. dehydration and rear-
rangement) during the acylation of 1-methylcyclohex-
anol, 1-ethynylcyclohexanol and 3-methyl-1-pentyn-
3-ol. The superiority of HBF4–SiO2 over the metal
triflates during acylation of alcohols may be best
demonstrated through comparison of the results of a
few representative examples of acid-sensitive and steri-
cally hindered substrates. Thus, acylation of 1-methyl-
cyclohexanol carried out in the presence of Sc(OTf)3
required 5 equiv. of Ac2O at −20°C for 5 h to afford
a 91% yield.9a,b The TMSOTf catalyzed acylation of
the same substrate was carried out at −10°C in Ac2O
as solvent. The HBF4–SiO2 catalyzed reaction
afforded a 77% yield with stoichiometric amounts of
Ac2O at room temperature for 2 h. The reaction of
1-ethynylcyclohexanol catalyzed by Sc(OTf)3, Bi(OTf)3
and TMSOTf required 10 equiv. of Ac2O and MeCN/
DCM as co-solvents to afford 94, 88, and 68% yields,
respectively, in 2–4 h. Contrary to these, a 96% yield
of the product could be obtained with 1 equiv. of
Ac2O for 6 h using HBF4–SiO2 in the absence of
solvent. Quantitative acylation of 3-methyl-1-pentyn-
3-ol takes place with a stoichiometric amount of
Ac2O in the absence of solvent at room temperature
under the catalytic influence of HBF4–SiO2 but the
corresponding TMSOTf catalyzed reaction required 2
equiv. of Ac2O in DCM at 0°C to afford an 80%
yield. Acylation of the sterically hindered endo-bor-
neol required the use of 10 equiv. of Ac2O in THF
for 7 h in the presence of TMSOTf and the corre-
sponding Bi(OTf)3 catalyzed reaction required 10
equiv. of Ac2O in THF to afford a 99% yield in 7 h.
In contrast, the HBF4–SiO2 catalyzed reaction
afforded comparable results in 45 min with 1 equiv.
of Ac2O in the absence of solvent.
1. (a) Greene, T. W.; Wuts, P. G. M. Protective Groups in
Organic Synthesis; Wiley: New York, 1999; (b) Hanson,
J. R. Protective Groups in Organic Synthesis; Blackwell
Science Inc: Malden, MA, 1999.
2. (a) Chakraborti, A. K.; Nayak, M. K.; Sharma, L. J.
Org. Chem. 2002, 67, 1776–1780; (b) Chakraborti, A. K.;
Nayak, M. K.; Sharma, L. J. Org. Chem. 2002, 67,
2541–2547; (c) Chakraborti, A. K.; Sharma, L.; Sharma,
U. Tetrahedron 2001, 57, 9343–9346; (d) Chakraborti, A.
K.; Nayak, M. K.; Sharma, L. J. Org. Chem. 1999, 64,
8027–8030.
3. Steglich, W.; Hofle, G. Angew. Chem., Int. Ed. Engl.
1969, 8, 981.
4. Vedjes, E.; Diver, T. S. J. Am. Chem. Soc. 1993, 115,
3358–3359.
5. (a) Iqbal, J.; Srivastava, R. J. Org. Chem. 1992, 57,
2001–2007; (b) Ahmed, S.; Iqbal, J. Tetrahedron Lett.
1986, 27, 3791–3794.
6. (a) Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto,
H. J. Am. Chem. Soc. 1995, 117, 4413–4414; (b) Ishihara,
K.; Kubota, M.; Kurihara, H.; Yamamoto, H. J. Org.
Chem. 1996, 61, 4560–4567.
7. Ishihara, K.; Kubota, M.; Yamamoto, H. Synlett 1996,
265–266.
8. (a) Procopiou, P. A.; Baugh, S. P. D.; Flack, S. S.; Inglis,
G. G. A. J. Org. Chem. 1998, 63, 2342–2347; (b) Proco-
piou, P. A.; Baugh, S. P. D.; Flack, S. S.; Inglis, G. G. A.
Chem. Commun. 1996, 2625–2626.
9. (a) Orita, A.; Tanahashi, C.; Kakuda, A.; Otera, J.
Angew. Chem., Int. Ed. Engl. 2000, 39, 2877–2879; (b)
Orita, A.; Tanahashi, C.; Kakuda, A.; Otera, J. J. Org.
Chem. 2001, 66, 8926–8934; (c) Carrigan, M. D.;
Freiberg, D. A.; Smith, R. C.; Zerth, H. M.; Mohan, R.
S. Synthesis 2001, 2091–2093.
10. Saravanan, P.; Singh, V. K. Tetrahedron Lett. 1999, 40,
2611–2614.
11. Chauhan, K. K.; Frost, C. G.; Love, I.; Waite, D. Synlett
1999, 1743–1744.
12. Chandrasekhar, S.; Ramachander, T.; Takhi, M. Tetra-
hedron Lett. 1998, 39, 3263–3266.
While comparing the catalytic efficiencies of HBF4–
SiO2 with other reported catalysts, we found that,
HBF4 on SiO2 is superior as it is less costly21 and the
reactions are, in general, carried out with stoichiomet-
ric amounts of Ac2O,22 at room temperature,23 and in
the absence of solvent.24
13. Ballini, R.; Bosica, G.; Carloni, S.; Ciaralli, L.; Maggi,
R.; Sartori, G. Tetrahedron Lett. 1998, 39, 6049–6052.
14. Li, A.-X.; Li, T.-S.; Ding, T.-H. Chem. Commun. 1997,
1389–1390.
15. Kumareswaran, R.; Pachamuthu, K.; Vankar, Y. D.
Synlett 2000, 1652–1654.
16. Kumar, P.; Pandey, R. K.; Bodas, M. S.; Dongare, M. K.
Synlett 2001, 206–209.
In conclusion, HBF4–SiO2 is a new and efficient cata-
lyst for the acylation of phenols, thiols, alcohols and
amines. The low cost and with increasing environ-
mental concerns;25 the solvent free conditions
employed in the present method will make it ‘envi-
ronmentally friendly’ and potentially useful for indus-
trial applications.
17. Nakae, Y.; Kusaki, I.; Sato, T. Synlett 2001, 1584–1586.
18. Reaction with 4-hydroxybenzaldehyde resulted in a mix-
ture of 4-acetoxybenzaldehyde, 4-hydroxybenzaldehyde-
1,1-diacetate, and 4-acetoxybenzaldehyde-1,1-diacetate.
19. Harrowven, D. C.; Dainty, R. F. Tetrahedron Lett. 1996,
37, 7659–7660.
20. Olah, G. A.; Prakash, G. K. S. Superacids; Wiley: New
York, 1985.
Preparation of HBF4–SiO2: A magnetically stirred
suspension of silica gel (26.7 g, 230–400 mesh) in
diethyl ether (75 ml) was treated with 40% aq. HBF4
(3.3 g) for 3 h. The mixture was concentrated and the
residue dried under vacuum at 100°C for 72 h to
afford HBF4–SiO2 (0.5 mmol of HBF4 g m−1).
21. Aldrich (India) 2000–2001 price (US $): In(OTf)3 (50 g,
362.20); Cu(OTf)2 (50 g, 384.80); Sc(OTf)3 (50 g,
1590.00); estimated price (not available commercially)
(US $): Sc(NTf2)3 (50 g, >4896.00); estimated price (not
available commercially) (US $): Bi(OTf)3·xH2O (50 g,
>315.95); HBF4 (200 g, 3.6).

 Chakraborti, Asit K.
Chakraborti, Asit K.
 Gulhane, Rajesh
Gulhane, Rajesh